- *Corresponding Author:
- V. P. Vaidya
Department of Chemistry, Sahyadri Science College, Shimoga-577 203, India
E-mail: vaidyavijaya@hotmail.com
| Date of Submission | 16 June 2005 |
| Date of Revision | 07 March 2006 |
| Date of Acceptance | 18 November 2006 |
| Indian J Pharm Sci, 2006, 68 (6): 719-725 |
Abstract
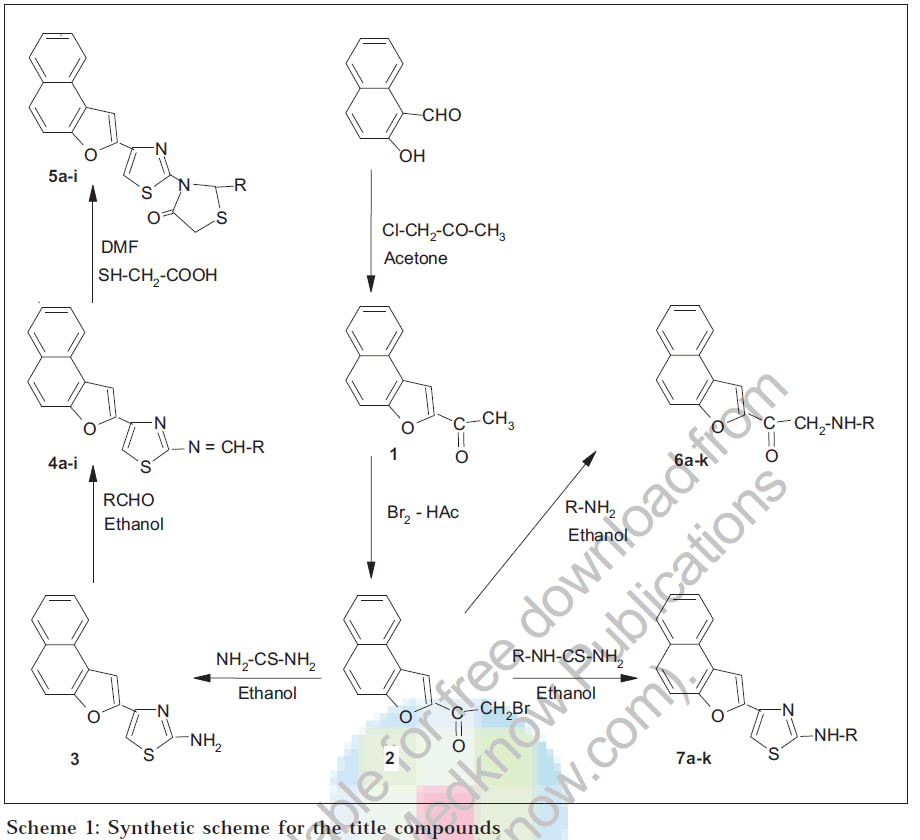
2-Acetylnaphtho[2,1- b ]furan (1), obtained from 2-hydroxy-1-naphthaldehyde was converted into 2-bromoacetylnaphtho[2,1- b ]furan (2), which served as an intermediate for the synthesis of title compounds. The compound (2) was treated with thiourea to get 2-(2-aminothiozol-4-yl)naphtho[2,1- b ]furan (3), which produced 2-(2-arylideneaminothiozol-4-yl)naphtho[2,1- b ]furans (4a-i) on reacting with various aromatic aldehydes. Schiff bases (4a-i) yielded 2-[2-(2-aryl-4-thiazolidinone)thiazol-4-yl]naphtho[2,1- b ]furans (5a-i) on treating with thioglycolic acid. Compound (2) was reacted with different aromatic amines to obtain 2-(N-aryl-2-amino)acetylnaphtho[2,1- b ]furans (6a-k). Similarly reaction with substituted thiourea furnished 2-(2-N-arylaminothiazol-4-yl)naphtho[2,1- b ]furans (7a-k). All the newly synthesized compounds have been characterized by elemental analysis and spectral data, which have been screened for antimicrobial and anthelmintic activities. The selected compounds have been evaluated for antiinflammatory and diuretic activities.
Keywords
The presence of thiazole moiety in the structure of several naturally occurring molecules with important antibiotic, immunosuppressive and antitumor activities have been known for several years [1-4]. The aminothiazole ring system has found application in drug development for the treatment of HIV-infection, hypertension and inflammation [5]. The several thiazole derivatives have been shown to exhibit excellent bactericidal6 fungicidal [7,8] and anthelmintic [9] activities. Recently thiozolidinones have been synthesized and screened for possible antimicrobial activities [10,11].
Encouraged by these reports and in continuation of our search for pharmacologically potent naphtho[2,1-b] furan derivatives, [12-16] we report in this paper the synthesis of some thiazolidine derivatives of naphtho[2,1-b]furan, other related compounds and pharmacological activity of the newly synthesized compounds.
Materials and Methods
Melting points were determined in open capillary tubes and are uncorrected. Purity of the compounds was checked by TLC on silica gel G. IR spectra were recorded in KBr on Unicam FTIR (research spectrophotometer series), NMR spectra on Jeol GSX 270 FT NMR spectrophotometer using TMS as an internal standard (chemical shift in δ, ppm) and mass spectrum on Brucker Apex-II 300 mass spectrophotometer, operating at 70 eV. The pharmacological activities were carried out at National Pharmacy College, Shimoga, (Institutional animals ethics committee, Registration No. 144/1999/CPCSEA/05-07-1999).
2-Acetylnaphtho[2,1-b]furan (1)
2-Hydroxy-1-naphthaldehyde (0.1 mol), chloroacetone (0.1 mol) and anhydrous potassium carbonate (10 g) were refluxed in dry acetone (100 ml) for 12 h. Potassium salts were filtered off, the filtrate on removal of solvent and on trituration with ethanol gave the pale yellow crystals of 2acetylnaphtho[2,1-b]furan (1). The sample was recrystallized from absolute ethanol.
2-Bromoacetylnaphtho[2,1-b]furan (2)
To a solution of 2-acetylnaphtho[2,1-b]furan (1) (0.1 mol) in glacial acetic acid (50 ml) was added a solution of bromine
(0.1 mol) in glacial acetic acid (20 ml) with stirring during 1 h at 10-20° and the stirring was continued for 3 h. The reaction mixture was poured into ice-cold water and the solid obtained was filtered out. It was washed with water, dried and the product was recrystallized from ethanol. IR (KBr) cm-1: 1672 (C=O), 1H NMR (CDCl3) δ ppm: 3.7 (s, 2H, CH2) and 6.7-8.5 (m, 7H, Ar-H).
2-(2-Aminothiazol-4-yl)naphtho[2,1-b]furan (3)
A solution of (2) (0.005 mol) in hot ethanol (50 ml) was treated with thiourea (0.01 mol), to obtain clear solution, which soon deposited some crystals. It was boiled in water (10 ml) containing sodium acetate (2.5 g), which was then filtered, dried and recrystallized from ethanol. IR (KBr) cm-1: 1636 (C=N), 3470, (NH2), 1H NMR(CDCl3) δ ppm : 3.5 (bs, 2H, NH2, D2O exchangeable) (), 6.9-9.1 (m, 8H, Ar-H), MS: m/z 266 [M]+, 250,167,105, 93,83, 77.
2-(2-Arylideneaminothiazol-4-yl)naphtho[2,1-b]furans (4a-i)
Compound (3) (0.01 mol) in ethanol (50 ml) was refluxed with benzaldehyde (0.01 mol) for 6 h in presence of few drops of acetic acid. The solvent was distilled off. The reaction mixture was cooled, poured into ice, filtered, and recrystallized from aqueous dimethyl formamide. The remaining compounds, (4b-i) were synthesized by using appropriate aldehydes similarly. 4f: IR (KBr) cm-1: 1615 (C=N), 1H NMR (CDCl3) δ ppm, 7.2-8.2 (m, 12H, Ar-H), 10.2 (s, 1H, N=CH).
2-[2-(2-Aryl-4-thiazolidinono)thiazol-4-yl]naphtho [2,1-b]furans (5a-i)
A mixture of 2-(2-arylideneaminothiazol-4- yl)naphtho[2,1-b]furan, (4a), (0.01 mol) and mercaptoacetic acid (0.01 mol) in DMF (25 ml) containing pinch of anhydrous zinc chloride was heated under reflux for 5 h. The reaction mixture was cooled and poured onto crushed ice. The solid thus separated was filtered, washed with water and recrystallized from methanol to obtain 5a: IR (KBr) cm-1: 1675 (C=O), 1H NMR:(CDCl3) δ ppm, 3.3 (s, 1H, CH), 4.2 (s, 2H, CH2), 7.3-8.2 (m, 13H, Ar-H). Compounds 5b-i) were prepared similarly from (4b-i) by using mercaptoacetic acid.
2-(N-Aryl-2-amino)acetylnaphtho[2,1-b]furans (6a-k)
An equimolar mixture of appropriate aromatic amines and 2-bromoacetylnaphtho[2,1-b]furan (2) in ethanol (50 ml) was refluxed for 5 h, cooled and poured onto ice cold water. The solid mass thus obtained was filtered and recrystallized from absolute ethanol. 6e: IR (KBr) cm-1: 1669 (C=O), 3260 (NH), 1H NMR: (CDCl3) δ ppm, 2.9 (s, 2H, CH2). 3.9 (s, 3H, OCH3), 6.8-8.5 (m, 11H, ArH), and
9.2 (bs, 1H, NH, exchangeable with D2O).
2-[2-Arylaminothiazol-4-yl]naphtho[2,1-b]furans (7a-k)
Arylthiourea (0.01 mol) and 2-bromoacetylnaphtho[2,1-b]furan
(2) (0.01 mol) in ethanol (50 ml) were mixed and the mixture was
heated under reflux for 3 h. The reaction mixture was then cooled, poured onto water, and neutralized with aqueous 5% sodium acetate solution. The solid mass thus obtained was collected by filtration, washed with water and recrystallized from aqueous dimethyl formamide. 7a: IR (KBr) cm-1: 1630 (C=N), 3330 (NH). 1H NMR δ ppm: 7.1-8.4 (m, 14H, ArH). 7h, IR: 1622 (C=N), 3395 (NH2), 1H NMR: 1.5 (s, 3H, CH3), 7.4-8.2 (m, 12H, ArH), 9.1 (bs, 1H, NH, exchangeable with D2O). The physical data of the synthesized compounds is presented in Table 1.
| Compd.Mol.formula | Mol.formula | R | Yield (%) | m.p.(0) | Found % calculated | |
|---|---|---|---|---|---|---|
| N | ||||||
| 1 | C14H10O2 | ? | 66 | 97 | ? | ? |
| 2 | C14H9O2Br | ? | 80 | 180 | ? | ? |
| 3 | C15H10 N2SO | ? | 55 | 210 | ? | ? |
| 4a | C22H14N2SO | C6H5 | 45 | 230 | 7.80 | 7.91 |
| 4b | C22H14N2SO2 | 2-OH C6H4 | 52 | 188 | 7.46 | 7.57 |
| 4c | C23H16N2SO2 | 2-OCH3 C6H4 | 48 | 198 | 7.10 | 7.29 |
| 4d | C23H16N2SO2 | 4-OCH3 C6H4 | 42 | 235 | 7.20 | 7.29 |
| 4e | C23H16N2SC3 | 4-OH3- OCH3C6H3 | 62 | 245 | 6.50 | 7.00 |
| 4f | C22H13N2SOCl | 4-Cl C6H4 | 45 | 210 | 6.95 | 7.21 |
| 4g | C22H13N3SC3 | 3-NO2 C6H4 | 65 | 265 | 10.40 | 10.53 |
| 4h | C24H16N2SO | CH=CH C6H5 | 46 | 220 | 7.20 | 7.37 |
| 4i | C20H12N2SO2 | Furyl | 68 | 215 | 7.94 | 8.14 |
| 5a | 16N2S2O2 | C6H5 | 40 | 240 | 6.20 | 6.54 |
| 5b | 16N2S2O3 | 2-OH C6H4 | 50 | 220 | 6.20 | 6.31 |
| 5c | 18N2S2O3 | 2-OCH3 C6H4 | 48 | 238 | 5.88 | 6.11 |
| 5d | 18N2S2O3 | 4-OCH3 C6H4 | 48 | 240 | 5.95 | 6.11 |
| 5e | 18N2S2O4 | 4-OH 3-OCH3C6H3 | 42 | 255 | 5.79 | 5.91 |
| 5f | C24H15N2S2O2Cl | 4-Cl C6H4 | 50 | 220 | 5.80 | 6.05 |
| 5g | N3S2O4 | 3-NO2 C6H4 | 70 | 280 | 8.40 | 8.88 |
| 5h | N2S2O2 | CH=CH C6H5 | 42 | 245 | 6.10 | 6.17 |
| 5i | N2S2O3 | Furyl | 68 | 265 | 6.54 | 6.70 |
| 6a | 15NO2 | C6H5 | 50 | 210 | 4.45 | 4.65 |
| 6b | 15NO4 | 4-COOH C6H4 | 60 | 220 | 3.95 | 4.06 |
| 6c | BrNO2 | 4-Br C6H4 | 50 | 185 | 3.18 | 3.68 |
| 6d | 15NO3 | 4-OH C6H4 | 55 | 190 | 4.40 | 4.42 |
| 6e | 17NO3 | 4-OCH3 C6H4 | 65 | 180 | 4.02 | 4.23 |
| 6f | 17NO2 | 2-CH3 C6H4 | 40 | 220 | 4.25 | 4.44 |
| 6g | 17NO2 | 3-CH3 C6H4 | 50 | 195 | 4.20 | 4.44 |
| 6h | 17NO2 | 4-CH3 C6H4 | 60 | 180 | 4.30 | 4.44 |
| 6i | 14N2O4 | 2-NO2C6H4 | 60 | 225 | 7.75 | 8.09 |
| 7a | 14N2O4 | 3-NO2C6H4 | 60 | 180 | 7.84 | 8.09 |
| 7b | 14N2O4 | 4-NO2C6H4 | 60 | 220 | 7.88 | 8.09 |
| 7c | 14N2OS | C6H5 | 50 | 225 | 7.95 | 8.19 |
| 7d | C22H14N2O3S | 4-COOH C6H4 | 40 | 250 | 7.20 | 7.25 |
| 7e | C21H13BrN2OS | 4-Br C6H4 | 50 | 260 | 6.20 | 6.65 |
| 7i | C21H14N2O2S | 4-OH C6H4 | 55 | 245 | 7.50 | 7.82 |
| 7j | C22H16N2O2S | 4-OCH3 C6H4 | 65 | 225 | 7.05 | 7.53 |
| 7k | 16N2OS | 2-CH3 C6H4 | 50 | 220 | 7.40 | 7.87 |
| 16N2OS | 3-CH3 C6H4 | 40 | 240 | 7.52 | 7.87 | |
| 16N2OS | 4-CH3 C6H4 | 55 | 230 | 7.72 | 7.87 | |
| 7i | C21H13N3O3S | 2-NO2C6H4 | 50 | 250 | 10.42 | 10.85 |
| 7j | C21H13N3O3S | 3-NO2C6H4 | 50 | 250 | 10.42 | 10.85 |
| 7k | C21H13N3O3S | 4-NO2C6H4 | 60 | 220 | 10.50 | 10.85 |
Table 1: Physical Constants of the Synthesized Compounds
Antimicrobial activity
The antimicrobial activity was determined by cup-plate method. [17,18] Pure cultures of the test microorganisms were procured from the cultures maintained at National College of Pharmacy, Shimoga. The in vitro antimicrobial activity was carried out against 24 h culture of two bacteria and two fungi. The bacteria used were Staphylococcus aureus and Klebsiella pneumoniae, while the fungi used were Aspergillus niger and Candida albicans. The compounds were tested at a concentration of 0.001 mol/ml in dimethyl formamide against both the organisms. Ciprofloxacin (0.001 mol/ml) and ciclopirox olamine (0.001 mol/ml) were used as standards for comparison of antibacterial and antifungal activities, respectively. The zone of inhibition was compared with the standard drug after 24 h of incubation at 37° for antibacterial activity and 72 h at 25° for antifungal activity. The results are reported in Table 2.
| Zone of inhibition in mm | ||||||
|---|---|---|---|---|---|---|
| Compound | Antibacterial activity* | Antifungal activity* | Anthelmintic activity (in min) | |||
| S. aureus | K. pneumoniae | A. niger | C. albicans | Paralysis | Death | |
| 4a | 8 | 10 | 12 | 14 | 110 | 150 |
| 4b | - | - | 10 | 12 | 120 | 130 |
| 4c | - | - | 8 | 10 | 110 | 140 |
| 4d | 12 | 14 | 8 | 14 | 120 | 180 |
| 4e | - | - | - | - | - | - |
| 4f | 10 | 12 | 14 | 16 | 120 | 170 |
| 4g | 16 | 12 | 24 | 22 | 110 | 140 |
| 4h | 12 | - | - | 12 | 140 | - |
| 4i | - | - | - | 6 | 110 | 160 |
| 5a | 14 | 10 | 16 | 18 | 110 | 150 |
| 5b | - | - | 15 | - | 120 | 130 |
| 5c | 10 | 14 | 26 | 28 | 120 | 170 |
| 5d | - | 18 | 10 | - | 110 | 160 |
| 5e | 15 | 18 | 20 | 24 | 140 | - |
| 5f | 18 | 17 | 14 | 20 | 120 | 150 |
| 5g | - | 22 | - | - | 130 | - |
| 5h | - | 14 | - | 15 | 120 | 150 |
| 5i | 18 | - | 20 | 24 | 110 | 130 |
| 6a | 26 | 28 | 26 | 24 | 90 | 110 |
| 6b | - | 16 | - | - | 100 | 130 |
| 6c | 10 | 12 | 25 | 24 | 80 | 120 |
| 6d | 20 | 16 | 12 | - | - | - |
| 6e | - | - | 20 | 22 | 120 | 170 |
| 6f | 14 | 16 | - | 22 | 110 | 150 |
| 6g | - | - | 20 | 18 | 120 | 160 |
| 6h | 24 | 26 | 26 | 26 | 100 | 130 |
| 6i | 15 | 19 | 20 | 24 | 120 | 150 |
| 6j | 12 | 10 | 14 | 16 | 120 | 160 |
| 6k | - | - | - | - | 150 | - |
| 7a | 24 | 28 | 26 | 24 | 125 | 155 |
| 7b | - | - | 18 | 16 | 130 | 160 |
| 7c | 26 | 26 | 25 | 24 | - | - |
| 7d | 20 | 16 | 12 | 10 | - | - |
| 7e | 13 | 10 | 10 | 12 | 110 | 120 |
| 7f | 6 | 12 | 12 | - | 80 | 100 |
| 7g | 15 | 18 | 20 | 18 | - | - |
| 7h | 24 | 26 | 26 | 26 | 125 | 165 |
| 7i | 15 | 12 | 10 | 14 | 90 | 120 |
| 7j | 10 | 12 | 14 | 16 | 100 | 135 |
| 7k | - | 10 | 16 | 140 | - | |
| Std | 24 | 26 | 24 | 22 | 100 | 125 |
| Saline | - | - | - | - | Nil | Nil |
Table 2: Antimicrobial and Anthelmintic Activity of Synthesized Compounds
Anthelmintic activity
The earthworms (Pheritima posthuma, Order-annelida, Class-oligichaeta)) were used to evaluate anthelmintic activity [19]. The test compounds 0.001 mol/ml in 25 ml suspension of 0.1% Tween-80 solution, 25 ml of 6% dextrose, 25 ml 0.1% Tween-80 solution and 25 ml of piperazine citrate solution (Standard) were poured into separate petridishes as test sample, control, blank, and standard respectively. Three worms were placed in each petridish and the time taken by worm to become motionless was noted as paralysis time and the time taken for complete death of worms was also recorded. The results are reported in Table 2.
Antiinflammatory activity
Winter’s hind paw method [20] was used for the evaluation of the antiinflammatory activity of compounds. Wistar rats of either sex weighing 140-200 g were divided into six groups of four animals each. Carrageenan, an irritant, was injected sub-cutaneously into the hind paw of the albino rats at a concentration of 1 mg/ml to produce the edema. Acacia mucilage (2%) was given to one set of animals as control. Another set received the standard drug, ibuprofen (40 mg/kg body weight) and rest of the animals received the test samples (30 mg/kg body weight) orally. The initial volume of the paw was measured plethysmographically within 30 sec of the injection. The relative increase in the paw volume after 3 h of the carrageenan injection was noted. The percentage inhibition of the inflammation was calculated and the results were reported in Table 3.
| Compound after 3 h | Paw volume ± SEM | % protection |
|---|---|---|
| Control | 1.24 ± 0.152 | - |
| Ibuprofen | 0.30 ± 0.037 | 75.8 |
| 5a | 0.44 ± 0.060 | 64.5 |
| 5b | 0.62 ± 0.105 | 50.0 |
| 5d | 0.46 ± 0.047 | 62.9 |
| 5f | 0.55 ± 0.085 | 55.6 |
| 5f | 0.70 ± 0.106 | 43.5 |
Table 3: Antiinflammatory Activity of Some Selected Compounds
Diuretic activity
The activity was evaluated on albino rats by reported method [21]. The Wistar strain rats weighing 140-200 g of either sex were divided into eight groups of three animals each and placed in metabolic cages, which were provided with a wire mesh at the bottom and a funnel to collect urine. Sieves made up of stainless steel are placed in the funnel to retain feces. The rats were fed with standard diet and water ad libitum. Food and water were withdrawn 24 h prior to the experiment. For screening procedure six groups of animals were served test compound and received 30 mg/kg body weight were administrated orally, which was suspended in 0.1% Tween-80. Another set of animals served standard and received 40 mg/kg body weight of frusemide (aqueous solution) orally. The remaining group of animals served as control and received 0.1% Tween-80 solution. Sodium chloride solution (0.9%) at a dose of 5 ml/100 g body weight was given to all the animals by gavage before the experiment. Urine excretion was recorded after 5 h and the values were tabulated in Table 4.
| Compound | Volume of urine collected after 5 h | T/S (Lipschitz values) |
|---|---|---|
| Std | 29 | 1.00 |
| 6a | 20 | 0.69 |
| 6h | 15 | 0.52 |
| 6k | 09 | 0.31 |
| 7a | 16 | 0.55 |
| 7h | 17 | 0.58 |
| 7k | 12 | 0.41 |
| Control | 08 | 0.27 |
Table 4: Diuretic Activity of Some Selected Compounds
Results and Discussion
The starting compound 2-acetylnaphtho[2,1-b]furan (1) was synthesized from 2-hydroxy-1-naphthaldehyde by reacting with chloroacetone, anhydrous potassium carbonate in dry acetone. The structure of compound
(1) was well established by IR, 1H NMR, 13C NMR and mass spectral studies. The IR spectrum exhibited a peak at 1678 cm-1 which has been attributed to -C=O group. 1H NMR spectrum showed a singlet at δ 2.6 integrating for 3 protons of -CH3 group and a multiplet between δ
7.5 and 8.2 integrating for 7 aromatic protons. The decoupled 13C NMR spectrum of (1) further confirmed the structure. It revealed peaks at δ 27 due to -CH3 carbon, δ 111.9 and 112.8 due to carbon atoms of furan ring and a bunch of singlets at, δ 123-131 due to ten carbon atoms of naphthalene ring and at δ 188 due to - C=O carbon. Mass spectrum of (1), showed molecular ion peak at m/z 210, consistent with the molecular weight with high intensity. Peaks observed at m/z 195, 139, and 43 are in well agreement with expected fragmentation pattern.
The compound (1) was converted into 2-bromoacetylnaphtho[2,1-b]furan (2) by bromination in presence of acetic acid at 10-20°, which served as an excellent intermediate for the synthesis of various naphtho[2,1-b]furan derivatives. The compound (2) was refluxed with thiourea and the product obtained was identified as 2-(2-aminothiazol-4-yl)naphtho[2,1-b]furan (3), on the basis of spectral studies. The compound (3) was made to undergo condensation with various aromatic aldehydes to obtain corresponding Schiff bases, 2-(2arylideneaminothiazol-4-yl)naphtho[2,1-b]furans, (4a-i). On refluxing Schiff bases (4a-i), with thioglycolic acid in dimethyl formamide in the presence of catalytic amount of anhydrous zinc chloride, cyclocondensation took place very smoothly and resulted in the formation of new triheterocyclic compounds, 2-[2-(2-aryl-4-thiazolidinono) thiazol-4-yl]naphtho[2,1-b]furans (5a-i).
The compound (2) on reaction with various aromatic amines in ethanol produced 2-(N-aryl-2amino)acetylnaphtho [2,1-b]furans (6a-k). Various substituted thiourea were prepared by the reaction between potassium thiocyanate and appropriate amines, these substituted thiourea on refluxing with compound (2), produced 2-(2-N-arylaminothiazol-4-yl)naphtho[2,1-b]furans (7a-k) in good yield (Scheme-1). The structures assigned to the compounds have been established by IR and 1H NMR spectral studies.
Investigation of antimicrobial activity revealed that the compounds 6a, 6h, 7a, 7c, and 7h exhibited promising activity against all the four organisms, whereas compounds 4g, 5c, and 6c were active against A. niger and C. albicans. Amongst the compounds tested for anthelmintic activity on Pheritima posthuma 6a, 6c, 7f, and 7i were found to be active and it was comparable with the piparazine citrate at the tested dose of 0.001 mol/ml concentration.
Anthelmintic drugs are those which expel the worms. These drugs cause paralysis of worms by penetrating the mucopolysaccharide layer to prevent the movement of worms. This leads to either paralysis or death of the worms. The tested compounds have exhibited considerable anthelmintic activity.
Compound 5a exhibited comparable (64.5%) antiinflammatory percent protection at the dose of 30 mg/kg, while the standard drug ibuprofen showed inhibition of 75.8% after 3 h of drug treatment. Screening results of diuretic activity revealed that the compound 6a exhibited moderate activity. From these results one could conclude that, all the activities are independent of electron withdrawing and electron donating groups in the newly synthesized molecules.
Pain and inflammation is caused due to injury and tissue damage. Carrageenan is an inflammagen and upon administration, it causes inflammation due to the release of histamine, serotinin and kinin, followed by bradykinin, protease, prostaglandin, and lysozyme. Most of the clinically effective non-steroidal antiinflammatory drugs (NSAIDS) are responded to prevent the release of bradykinin, protease, prostaglandin, and lysozyme. Also NSAIDS are effective and can act centrally on CNS or peripherally on ANS. The compounds, which are reported to have antiinflammatory activity are NSAIDS and probably act similar to the existing clinically effective agents available now.
Diuretics are agents, which promote the excretion of urine and also increase the rate of excretion of Na+ and Cl–. Extra cellular fluid volume is determined by sodium chloride in the body. Diuretics reduce the extra cellular fluid volume by decreasing total body sodium chloride content. Some of the compounds, which are shown to exhibit diuretic activity, are presumed to influence the excretion of sodium and it is well-supported by their antiinflammatory activity, as the inflammation can also be caused due to accumulation of fluid in the joints. However, the mechanisms of action of these compounds are yet to be established.
Acknowledgements
The authors are grateful to UGC for awarding teacher fellowship to HMV. The authors are also thankful to CDRI, Lucknow, for providing spectral data for the synthesized compounds and to Mr. M. L. Vijay Kumar, National College of Pharmacy, Shimoga, for his help in carrying out pharmacological activities.
References
- Mulzer, J., Mantoulider, A. and Ohler, E., Tetrahedron Lett., 1997, 38, 7725.
- Badorc, A., Bordes, M.F., deCointel, P., Savi, P., Lale, A., Petitou, M., Maffrand, J. P. and Herbert, J.M., J. Med. Chem., 1997, 40, 3393.
- Feng, S. and Schreiber, L.S. , J. Med. Chem., 1997, 119, 10873.
- Rzasa, M., Shea, H.A. and Romo, D., J. Amer. Chem. Soc., 1998, 120, 591.
- Kearney, P.C., Fernandz, M. and Flygare, J.A., J. Org. Chem., 1988, 63, 196.
- Holla, B.S., Kalluraya, B. and Ambekar, S.Y., J. Indian Chem. Soc., 1987, 64, 494
- Gawande, N.G. and Shingare, M.S., Indian J. Chem., 1981, 20B, 387.
- Lodhi, R.S., Srivastava, S.D. and Srivastava, S.K., Indian J. Chem, 1998, 37B, 899.
- Kalluraya, B., Rahiman, A.M., David Banji, Isloor, A.M. and Ganesh Rai, Indian J. Heterocycl. Chem., 2000, 9, 217.
- Parmar, J.M., Modha, J.J. and Parikh, A.R., Indian J. Chem., 1998, 38B, 440.
- Galietta, J.V. and Verkman, A.S., J. Biol. Chem., 2002, 277.
- Vagdevi, H.M. and Vaidya, V.P., Indian J. Heterocycl. Chem., 2001, 10, 253.
- Vagdevi, H.M., Latha, K.P., Vaidya, V.P., Vijaya Kumar, M.L. and Pai K.S.R., Indian J. Pharm. Sci., 2001, 63, 286.
- Mahadevan, K.M., Vaidya, V.P. and Vagdevi, H.M., Indian J. Chem., 2003, 42B, 1931.
- Padmashali, B., Vaidya, V.P. and Vijaya Kumar, M.L., Indian J. Heterocycl.Chem., 2001, 11, 15.
- Latha, K.P., Vaidya, V.P. and Keshavayya, J., Synth. React. Inorg. Met-Org. Chem., 2004, 34, 667.
- Mukhanova, T.I., Lykova, O.A., Alekseeva, L.M. and Granik, V.G., Chem. Heterocycl. Compd., 1998, 34(6), 651.
- “Indian Pharmacopoeia”, Controller of Publication Delhi, India II, A 9.1, 1996. 100.
- Giand, K.N., Dar R.N., Chopra, B.N. and Kaul, K.N., Indian J. Pharm. Sci., 1963, 27, 196.
- Hsieh, H.K., Tsao L.T., Wang, J.P. and Lin, C.N., J. Pharm.Pharmacol., 2000, 52(2), 163.
- Lipschitz, W.L., Hadidian, Z. and Kerpesar, A., J. Pharmacol. Exp. Ther., 1943, 79, 97