- *Corresponding Author:
- CH. Erdenechimeg
Department of Chemistry and Technology, Research Canter, Institute of Traditional Medicine and Technology, Ulaanbaatar 17042, Mongolia
E-mail: Erdenechimeg.ch805@gmail.com
| Date of Received | 12 February 2025 |
| Date of Revision | 13 February 2025 |
| Date of Acceptance | 30 March 2025 |
| Indian J Pharm Sci 2025;87(1):14-21 |
This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms
Abstract
Erkhem-8 herbal tablet, named BTSO BO BRGYAD in Tibetan, is prescribed for all types of fever, especially typhus and contagious fever in Mongolian traditional medicine. In the current study, the purpose was to establish the requirements of the standard on the medicine in tablet form and the febrifugal effect of the Erkhem-8 herbal tablet. 712 mg/kg of Erkhem-8 tablet decreased the temperature of the rectum in 1 h-4 h compared to the lipopolysaccharide model group (p<0.01). The temperature of the rectum of animals in the comparative group was statistically significantly reduced at 2 h and 6 h compared to the healthy rats (p<0.01). The group of Erkhem-8 tablet+lipopolysaccharide decr eased the interleukin-1beta cytokines level up to 33.5 % (p=0.05) from the group that treated lipopolysaccharide and the concentration of the interleukin-6 cytokines in the treatment group was statistically significantly reduced by 17.77±2.6 pg/ml or 27.5 % (p=0.05). A slight thickening of the alveolar walls, less blood stasis, and less fluid in the alveolar air spaces were detected in the Erkhem-8 tablet+lipopolysaccharide group. Overall, the criteria of the standardization parameter for the Erkhem-8 herbal tablet were established and the febrifugal effect of the tablet was evaluated.
Keywords
Traditional medicine, febrifugal effect, fever, inflammation, apigenin, luteolin
Inflammation that develops by four characteristics swelling, fever, redness, and hyperalgesia is an immunological protection process elicited in reaction to automatic damage, blazes, allergens, biological infections, and other poisonous stimuli[1]. Fever is a popular symptom of various inflammatory diseases and a process in which the body’s temperature increases above the normal range. Even though fever might be involved as a moderator defense response, it has possibly dangerous effects such as dehydration, swoon, muscular contractions, cardiopulmonary tension, organ injuries (e.g., liver, brain) and other abnormal physiological growth in organisms[2].
Erkhem-8, called BTSO BO BRGYAD in Mongolian and Tibetan, treats fever caused by blood, bile and kills bacteria. It has been used for fever of vital and vessel organs, new and old fever, epidemic, and infectious (typhus fever, contagious fever) disorders. Erkhem-8 has the characteristic remove heat when it is usually used for chronic and acute inflammation, especially pneumonia, hepatitis, jaundice, fever, and contagious disease. This medicine is a general prescription for fever of various causes and internal organs acute, chronic inflammatory disease.
Also, it is mentioned in the “Four Medical Tantras book” and other sutras that Erkhem-8 is called the prescription of noble for treating hot-caused diseases[3,4].
Erkhem-8 contains eight ingredients floss of Carthamus tinctorius L., herb of Odontites vulgaris (O. vulgaris) Moench., Lagotis integrifolia (L. integrifolia) and Gentiana barbata L., wood of Santalum album L., radix of Aconitum Кusnezoffii Reichb., Calcio sinter, and Bostaurus domesticus Gmeline[5].
C. tinctorius has already been investigated to have pain-relieving, antioxidant, antidiabetic, and anti- inflammatory activities, and its common compounds are cartamin, safflower yellow, luteolin, etc.,[6]. Kaempferol glucosides that were isolated from C. tinctorius are shown to affect antinociceptive and anti-inflammatory in vivo studies[7].
O. vulgaris Moench., is applied for treating blood fever, pneumonia, typhoid fever, and alleviating pain. Its chemical compounds are 2.34 % flavonoids (apigenin, luteolin, apigenin 7-O-b-Dglucoside), 2.72 % phenol carboxylic acids, and 1.8 % iridoids (aucubin, and its glycosides, catalpol)[8].
L. integrifolia is a unique medicinal plant in Mongolian Traditional Medicine (MTM) and diosmetin, cynaroside, and mannitol were isolated in the plant[9]. Also, phenylethanoids and iridoids, such as aucubin, gardoside, and mannitol have been detected in L. integrifolia[10].
The febrile reaction can be imitated by systemic Lipopolysaccharide (LPS) injection bacterial infection being the most popular cause. In general, the induction of fever releases with pyrogenous cytokines like Interleukin-1 (IL-1), IL-1Beta (β), and Tumor Necrosis Factor-Alpha (TNF-α)[2].
In the research work, the purpose focused on establishing the requirements of the standard on the medicine in tablet form and the trend of the differences in symptoms of inflammation of the Erkhem-8 herbal tablet.
Materials and Methods
Materials:
Erkhem-8 herbal tablet (S.no: 01052024) that developed from Erkhem-8 traditional prescription for the experiment in traditional drug factory, Institute of Traditional Medicine and Technology was employed in the current study.
Standards and chemicals: Reference luteolin, apigenin, and aucubin were used from Sigma Aldrich (United States of America (USA)). All other reagents and solvents were analytical grade.
Animals and groups: Eighty male adult Wistar rats (220-250 g) were used in the current experiment.Animals were placed in the warehouse that was controlled to adjust 20°±1.5° temperature, with a 12 h light/dark cycle and rats were fed rodents water and food ad libitum for 8 d. This study was allowed by the Research Ethics Committee No., 2023/3-04 at the Mongolian National University of Medical Sciences. Healthy rats were measured at rectal temperatures between 35° and 36° using a digital thermometer. Briefly, animals were separated into the following groups, consisting of the vehicle/health control group, LPS treated or model group, LPS+Erkhem-8 tablet 712 mg/kg (LPS+ET 712 mg/kg) treated, and LPS+Paracetamol 50 mg/kg (LPS+P 50 mg/kg) treated groups[16].
Thin-Layer Chromatography (TLC) identification of the flavonoids: A sample of the Erkhem-8 tablet (1 g) was extracted with 50 ml of 40 % Ethanol (EtOH) by reflux for 30 min, and the cooled extracts were added with 10 ml of 10 % Hydrochloric Acid (HCl) and again refluxed for 30 min. The cooled extract was added and shaken with 10 ml chloroform (two times). The chloroform fractions were combined and evaporated and the dry residues were dissolved in methanol and used for the TLC as sample solutions. Reference solutions of luteolin and apigenin were prepared at 1 µl/ml in methanol. 10 µl sample solution and reference solutions were used in TLC plates (Merck Silicagel 60 GF 254). The plate has been placed with a mobile phase of hexane-ethyl acetate-acetic acid (30:15:5, v/v). After drying at 20°-22°, the plate was sprayed with a 5 % solution of aluminum chloride in ethanol and was visualized under 365 nm Ultraviolet (UV) lights[11].
TLC identification of the iridoids: A sample of the Erkhem-8 tablet (1 g) was extracted with 25 ml of methanol by reflux for 30 min, and the extract was cooled and applied for the TLC as a sample solution. The reference solution of aucubin was prepared at 5 µl/ ml in methanol. 10 µl sample solution and reference solutions were used in TLC plates (Merck Silicagel 60 GF 254). The plate has been placed with a mobile phase of ethyl acetate-acetone-formic acid-water (5:5:1:1, v/v). After drying at 20°-22°, the plate was sprayed with a 5 % solution of anisaldehyde-sulfuric acid and heated at 105° for 5-10 min[12].
Determination of total flavonoid content: A sample of the Erkhem-8 tablet (1 g) was extracted with 50 ml of 70 % ethanol by reflux for 30 min, and the extract was cooled and filtered. 3 ml test solution was treated in a 25 ml volumetric flask, added 1 ml of 5 % Sodium nitrite (NaNO2), 1 ml of 10 % Aluminium nitrate (Al(NO3)3), and 10 ml of 4 % Sodium hydroxide (NaOH) solution, and absorbance was measured at 500 nm with a spectrophotometer (UNICO UV-2102 C, China). The amount of flavonoid in extracts is expressed as luteolin equivalent[13].Determination of total iridoid content: The content of iridoids was determined according to the colorimetric method based on a Trim-Hill reaction. 0.4 ml test solution was treated in a 10 ml tube, 4 ml of Trim-Hill reagent, and heated at 70° for 10 min. The solutions were cooled and measured at 609 nm. The total iridoid contents were reported using a calibration curve of aucubin reference[14].
Hardness: The hardness of the Erkhem-8 herbal tablet was according to Mongolian National Pharmacopoeia (MNP) and used by SY-6D four functions tablet tester (China). The sample for the hardness test was calculated to compare the force applied to the anvils in diameter and height of the tablet (mm)[15].
Friability: According to MNP, the friability is utilized with a SY-6D four-function tablet tester (China). The samples were examined at a speed of 25 rpm/min. To compare the diversity of the weight fore-and- after the turnover the friability value was reported by percentage[15].
Pharmacological methods:
LPS isolated from Escherichia coli type O111:B4 was diluted in saline (Sigma Aldrich). To induce the LPS model, LPS solution (0.5 mg/kg body weight) was injected intravenously through the tail vein, and 60 min later, experimental and comparative drugs were administered orally. Following the injected LPS, each dose of Erkhem-8 tablet (LPS+ET 712 mg/kg) and Paracetamol (LPS+P 50 mg/kg) was orally administrated to the animals. The cytokine level assay was measured after 10 h. Each group was included ten animals (n=10). In another examination temperature, and histological investigation were performed following the injection of erkhem-8 tablet and paracetamol to a predetermined group of rats that had been injected with LPS. The erkhem-8 tablet and paracetamol were administered orally an hour and 6 h later than the LPS injection. The healthy rats were injected with 0.9 % Sodium chloride (NaCl) intravenously[16,17].
Temperature measurement assay: In every group, the temperature of the rectum of each animal was measured periodically with a digital thermometer. The initial temperature (T0: 35°-36°) was the temperature of rats following adaptation for 30 min at 20°. The monitoring of temperature changes was performed every 60 min[16, 17].
Cytokine IL-1β and IL-6 levels assay: Plasma for cytokine testing was acquired from the blood via cardiocentesis and blood 2-3 ml was collected on tubes containing Ethylenediaminetetraacetic Acid (EDTA) and the mixture was centrifuged for 15 min at 1500 rpm. The supernatant containing blood plasma was gathered in a tube for further cytokine testing. IL-6 and IL-1β cytokines were tested using the Enzyme- linked immunosorbent assay kit (Shanghai MLBIO Biotechnology Co. Ltd.,)[16,17].
Histological examination:
Histological specimens were fixed in a 10 % neutral buffered formalin solution for above 24 h, washed with running water for 24 h, dehydrated with graded ethanol, embedded in melted paraffin wax, sectioned (3-5 μm thick) utilizing a microtome (Yamato Kohki, Japan), and stained with Hematoxylin and Eosin (HE) (Sigma Aldrich) and Masson’s trichrome (IHC World stain kit, USA). Sections were examined and photographed by light microscopy (Nikon Eclipse Ci, Japan) inflammatory cells of lung alveoli were observed by a light microscope with a calibrated ocular scale[18].
Statistical analysis:
The average value and Standard Deviation (SD) were evaluated. The data of the cytokines assay and rectal temperature were calculated using a one-way Analysis Of Variance (ANOVA) that includes p values less than 0.05 and the Kruskal-Wallis test.
Results and Discussion
The main ingredients of Erkhem-8 herbal tablets such as O. vulgaris, Carthamus tinctorius, L. integrifolia, and Gentiana barbata L. contain flavonoids, especially luteolin, apigenin, quercetin, and their glycosides have been studied[7,19,20].
Also, O. vulgaris and L. integrifolia are included iridoids and phenylethanoids. The most widely occurring iridoid is Aucubin which is studied for antioxidant, anti-inflammatory, anti-fibrotic, anti- cancer, hepatoprotective, and anti-aging activities[21].
Flavonoid glycosides need to hydrolase into separate aglycon and glycoside. The result has shown that apigenin and luteolin were available for the identification of the prescription (Table 1). These aglycons of flavonoids were simultaneously visualized with good specificity in the TLC condition (Table 1). The intensity of the apigenin spot was deeper than the luteolin spot intensity was similar to the result of TLC in Artemisia annua L. but a slight modification was made in the methodology of the experiment[11]. Aucubin was available for identification in the methanol extract of the Erkhem-8 herbal tablet directly and the Ration factor (Rf) value was 0.51.
| Bio-active compound | Mobile phase | Spray reagents | Visible color | Rf value |
|---|---|---|---|---|
| Luteolin | Hexane-ethyl acetate-acetic acid (30:15:5, v/v) | 5 % solution of aluminum chloride in ethanol. At 365 nm UV lights. | Light yellow fluorescence | 0.31 |
| Apigenin | Light yellow fluorescence | 0.43 | ||
| Aucubin | Ethyl acetate-acetone-formic acid-water (5:5:1:1, v/v) | 5 % solution of anisaldehyde-sulfuric acid. To heat at 105° for 5-10 min. | Brown | 0.51 |
Table 1: Identification of Bio-Active Compounds in The Erkhem-8 Herbal Tablet
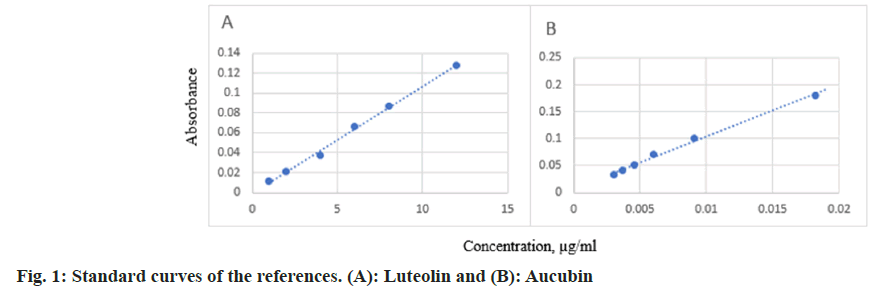
The colorimetric method of aluminum chloride is a widely used method based on the reaction of a complex agent, Al (III) with flavonoids in phytochemistry studies[22]. The flavonoid contents of the extract in terms of luteolin equivalent (standard curve equation: y=0.0108x-0.0012, r2=0.9963) were from 4.0 to 40.0 (fig. 1A). The content of iridoids in terms of aucubin equivalent (standard curve equation: y=9.6425x+0.0074, r2=0.9952) were between 3-18 µg/ ml (fig. 1B). The amount of flavonoid was more than the iridoid content in the Erkhem-8 herbal tablet (Table 2).
| Biological active substance | Amount |
|---|---|
| Total flavonoids | 0.61±0.04 % |
| Total iridoids | 0.39±0.06 % |
Note: Each amount is the average of three measurements
Table 2: Total Amount of Flavonoids and Iridoids
Erkhem-8 herbal tablet had a yellow colour, bitter taste, no special odour, and a convex round shape. General requirements for Erkhem-8 herbal tablet were estimated as the average weight 355.1±5.9 mg, hardness 0.71±0.07, friability 98.35±0.87 %, and moisture 1.3±0.1 % (Table 3).
| General requirements | Method | Result |
|---|---|---|
| Color | Yellow | |
| Taste | Sensual | Bitter |
| Odor | No special odor | |
| Shape | Convex round | |
| Diameter | Digital caliper | 9.1±0.02 mm |
| Height | 5.3±0.03 mm | |
| Average weight | Electronic weight | 355.1±5.9 mg |
| Moisture | 1.3±0.1 % | |
| Hardness | Tablet tester | 0.71±0.07 |
| Friability | 98.35±0.87 % |
Table 3: General Requirements of Erkhem-8 Herbal Tablet
The following results illustrated the effect of the Erkhem-8 tablet on the LPS-induced inflammatory model in rats (Table 4).
| Time (hour) | Groups | |||
|---|---|---|---|---|
| Vehicle/control | LPS/model | LPS+ET 712 mg/kg | LPS+P 50 mg/kg | |
| 0 | 35.0±1.1 | 35.73±0.61 | 35.18±1.35 | 36.06±0.99 |
| 1 | 35.0±1.1 | 37.43±0.99** | 35.42±0.39## | 36.60±0.50 |
| 2 | 35.0±1.1 | 38.55±0.78*** | 34.90±0.72### | 35.66±1.10### |
| 3 | 35.0±1.1 | 37.50±0.50** | 35.22±0.91### | 36.56±0.70 |
| 4 | 35.0±1.1 | 38.10±0.43*** | 35.56±0.59### | 37.86±0.51 |
| 5 | 35.0±1.1 | 37.52±0.39** | 36.80±0.69 | 37.86±0.35 |
| 6 | 35.0±1.1 | 38.01±0.51*** | 37.30±0.47 | 36.32±1.08### |
| 7 | 35.0±1.1 | 37.35±0.59** | 37.23±0.45 | 37.42±0.46 |
| 8 | 35.0±1.1 | 38.70±0.48*** | 37.50±1.23 | 37.06±0.87 |
| 9 | 35.0±1.1 | 37.80±0.63** | 36.90±1.07 | 37.15±0.49 |
| 10 | 35.0±1.1 | 37.84±0.79** | 36.00±0.61 | 37.05±0.21 |
Note: Compared to vehicle controls ***p<0.001, **p<0.05 and compared to LPS models ###p<0.01, ##p<0.05
Table 4: Differences in Rectal Temperature Between Groups During The LPS-Induced Inflammatory Model
Under the analysis results, when LPS was injected into the tail vein of rats at 0.5 mg/kg dose, the temperature of the rectum increased more than the control group at 0-10 h. But, in the 712 mg/kg dose of Erkhem-8 tablet, 1 the temperature of the rectum decreased in 1 h-4 h contrasted to the group that treated LPS (p<0.01). The temperature of the rectum of animals in the comparative group was statistically significantly decreased at 2 h and 6 h compared to the control group (p<0.01) (Table 4).
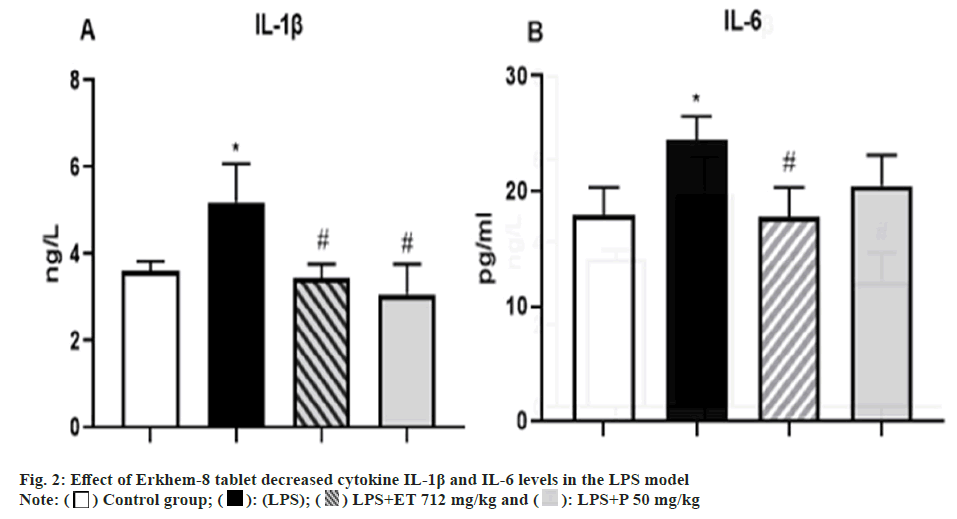
Effect of ET on plasma IL-1β (Group. 1A) and IL-6 (Group. 1B) levels in LPS-induced rats (n=10), showing a significant reduction of both cytokine’s levels compared with the LPS. The result showed IL-1β level was 3.6±0.23 µg/l in control rats and increased by 44.4 % to 5.2±0.89 ng/l in LPS (p=0.01). However, the 712 mg/kg group of ET+LPS touched up to 3.45±0.3 ng/l or else, it was decreased by 33.5 % (p=0.05) from the LPS, while in the paracetamol group, the IL-1β amount decreased by 41 % up to 3.076±0.68 ng/l (p=0.05).
However, IL-6 cytokine concentration was 17.9±2.5 pg/ml in controls and 24.5±2.0 pg/ml in LPS groups increased by 40 % (p=0.01). A statistically significant difference was observed in the 712 mg/kg group of LPS+ET by 17.77±2.6 pg/ml or 27.5 % (p=0.05). In the comparative group, it was determined to be reduced by 20.5±2.6 pg/ml (p=0.994) (Group 1).
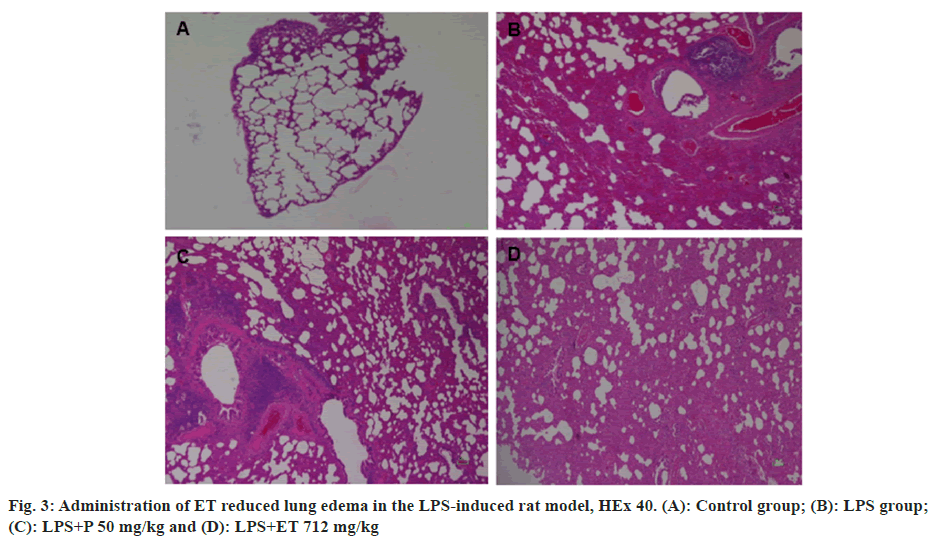
Following the results of the histological analysis, the alveolar walls of the lung lobes of LPS group rats were significantly thickened and hemostasis occurred. Changes such as foci of infiltration of inflammatory cells in the alveolar wall, proliferation of Bronchial Epithelial Cells (BEC), and enlargement of lymphoid follicles around the bronchioles indicate the occurrence of Acute Lung Injury (ALI). Lymphoid follicles around the bronchioles in the lungs of the group of LPS+ET 712 mg/kg were slightly enlarged, and BEC proliferated. There was infiltration of inflammatory cells and the alveolar wall became thick in a slice of the lung lobe (fig. 2).
The lymphoid follicles around the bronchioles in the lungs of the rats were enlarged, and a few BEC proliferated in the LPS+P 50 mg/kg group. A slight thickening of the alveolar walls, less amount of blood stasis, and less amount of fluid in the alveolar air spaces were observed (fig. 3).
By the results of activity inflammatory cytokine levels and histological analysis, the therapeutic effect of Erkhem-8 and the comparative group is similar (Table 5).
| Groups | Pathological changes | |||||
|---|---|---|---|---|---|---|
| Inflammatory cells | Blood stasis | Alveolar wall edema | Intra-alveolar edema | Proliferation of bronchiolar epithelial cells | Alveolar wall thickening | |
| LPS | +++ | ++ | - | - | +++ | +++ |
| LPS+P 50 mg/kg | ++ | + | - | + | ++ | + |
| LPS+ET 712 mg/kg | ++ | + | - | - | ++ | + |
Note: None (-); less (+), moderately (++) and severe (+++)
Table 5: Assessment of The Effects of Erkhem-8 on The Transformation in Lung Micro Preparation During The LPS Model by Pathological Scores
The LPS mechanism of ALI is primarily contributed in the circumstance of sepsis and pneumonia, where bacteria or endotoxins provoke an inflammatory reaction in the lungs. In brief, LPS is recognized by immunological protection through Pattern Recognition Receptors (PRRs), including Toll-Like Receptor 4 (TLR4). The connecting of LPS to TLR4 activates various signalling pathways[17,18].
The activity of TLR4 triggers the Nuclear Factor Kappa-B (NF-κB) and other signalling pathways developing to the expression of Pro-Inflammatory Cytokines (PIC) such as TNF-alpha, IL-1, and IL-6. According to recent studies inflammatory cytokines play a relevant role in the pathogenesis of ALI. IL-1β causes the failure of the lung’s epithelial and endothelial cellular layer. Also, IL-6 is elaborated pulmonary edema and protein-rich hyaline membranes. Synthesis of these cytokines plays a critical role in the pathogenesis of ALI and is also involved in the production of ROS. Therefore, inhibition of PIC could improve ALI. LPS is injected, and it binds to TLRs on immune cells, leading to the release of PICs such as IL-1, TNF-α, and IL-6. These cytokines play an important role in the onset of fever[17,23,24].
In the current study, the treatment effects of the erkhem-8 tablet, which proceeds from erkhem-8 prescription, LPS-induced ALI in experimental animals were inquired about rectal temperature, PIC, and changes of the histological structure. As evidenced in our work, when compared with the LPS group, erkhem-8 treatment was illustrated to notably decrease the concentration of IL-1β and reduce the concentration of IL-6 cytokine in rats. The observation was detected that was similar to the paracetamol treatment.
In LPS-induced ALI, the rectal temperature increased significantly during the 0 h-10 h of observation and decreased statistically significantly during the 0 h-4 h in the erkhem-8 group.
Histological analysis demonstrated the alveolar walls of the lung lobes of LPS group rats were significantly thickened and hemostasis occurred. Changes such as foci of infiltration of inflammatory cells in the alveolar wall, proliferation of BEC, and enlargement of lymphoid follicles around the bronchioles indicate the occurrence of ALI. However, this change was less evidenced in the erhem-8 group, the expansion of rat BEC, infiltration of inflammatory cells, and the thickening of the alveolar wall were slightly defined. The histological experiment disclosed that erkhem-8 decreased histopathological changes, and neutrophilic infiltration, then improved lung damage. The results of our study are directly related to the biologically active substance contained in erkhem-8. One example of this is the flavonoid family Apigenin, which is a main component of the erkhem-8 tablet, which reduces the concentration of the proinflammatory cytokine Cyclooxygenase-2 (COX- 2) and NF-kB activation during LPS-induced ALI. The results illustrated that apigenin is helpful in the treatment of ALI[25].
In conclusion, the draft of the monograph for the erkhem-8 herbal tablet was developed and criteria of the quality control requirements for the herbal remedy were established. The LPS-induced ALI in the treatment group significantly reduced compared with the control group. The results illustrated that the value of the inflammatory primarily cytokines decreased in the Erkhem-8 group.
Acknowledgments:
We want to thank members of our institute for their unique support of the research project.
Funding source:
The research project was financed by the Mongolian Foundation for Science and Technology and the agreement number of the project was ShUTBIKhKhZG-2022/136.
Conflict of interest:
The authors declared no conflict of interests.
References
- Yoon JH, Baek SJ. Molecular targets of dietary polyphenols with anti-inflammatory properties. Yonsei Med J 2005;46(5):585-96.
[Crossref] [Google Scholar] [PubMed]
- Yao XJ, Yin JA, Xia YF, Wei ZF, Luo YB, Liu M, et al. Puerarin exerts antipyretic effect on lipopolysaccharide-induced fever in rats involving inhibition of pyrogen production from macrophages. J Ethnopharmacol 2012;141(1):322-30.
[Crossref] [Google Scholar] [PubMed]
- Ganbayar Y, Tumurbaatar N. Ayurveda in Mongolia, history of Ayurveda in Mongolia, Lunrig Dandar, “History of Four Medical Tantras”. Mongolian J Health Sci 2009;1(6).
- Yuthok Yönten Gönpo. Four medical tantra. Ulaanbaatar 1991;306.
- Jambalchoijidanzanperenlei. Manag Rinchin Junai. Traditional Medical Source Book. China: Inner Mongolian medical treasurers: Printing house; 1978.
- Asgarpanah J, Kazemivash N. Phytochemistry, pharmacology and medicinal properties of Carthamus tinctorius L. Chinese J Integrat Med 2013;19:153-9.
[Crossref] [Google Scholar] [PubMed]
- Wang Y, Chen P, Tang C, Wang Y, Li Y, Zhang H. Antinociceptive and anti-inflammatory activities of extract and two isolated flavonoids of Carthamus tinctorius L. J Ethnopharmacol 2014;151(2):944-50.
[Crossref] [Google Scholar] [PubMed]
- WHO Western Pacific Region, medicinal plants in Mongolia; 2013;124.
- Chuluunbaatar E, Sodnomtseren P, Chimedtseren C, Batsuren G, Dulamjav B. Isolation of two flavonoids and mannitol from Lagotis integrifolia (Willd.) Schischk (Scrophulariaceae). Central Asian J Med Sci 2017;3(2):167-72.
- Jensen SR, Gotfredsen CH, Zidorn C. Iridoids and phenylethanoids in Lagotis integrifolia and Wulfeniopsis amherstiana (Plantaginaceae). Biochem Syst Ecol 2009;37(4):421-5.
- Phadungrakwittaya R, Chotewuttakorn S, Piwtong P, Thamsermsang O, Laohapand T, Akarasereenont P. Identification of apigenin and luteolin in Artemisia annua L. for the quality control. Siriraj Med J 2019;71(3):240-5.
- Wagner H, Bladt S. Plant drug analysis, a thin layer chromatography atlas. 2nd ed. Berlin: Springer; 1996.
- Du Q, Li W, Yang S, Duo J, Li Q, Xue Y. Detection and development on total flavonoids by the way of Al(NO3)3 ultraviolet colorimetry. RISM 2020;7(2):604-7.
- Erdenechimeg C, Guiqide A, Dejidmaa B, Chimedragchaa C, Purevsuren S. Total phenolic, flavonoid, alkaloid and iridoid content and preventive effect of Lider-7-tang on lipopolysaccharide-induced acute lung injury in rats. Brazil J Med Biol Res 2017;50(12):e5916.
[Crossref] [Google Scholar] [PubMed]
- Mongolian national first pharmacopeia. Ulaanbaatar (MGL); 2011.
- Tjahjono Y, Karnati S, Foe K, Anggara E, Gunawan YN, Wijaya H, et al. Anti-inflammatory activity of 2-((3-(chloromethyl) benzoyl) oxy) benzoic acid in LPS-induced rat model. Prostaglandins Other Lipid Mediators 2021;154:106549.
[Crossref] [Google Scholar] [PubMed]
- Ali H, Khan A, Ali J, Ullah H, Khan A, Ali H, et al. Attenuation of LPS-induced acute lung injury by continentalic acid in rodents through inhibition of inflammatory mediators correlates with increased Nrf2 protein expression. BMC Pharmacol Toxicol 2020;21:1-4.
[Crossref] [Google Scholar] [PubMed]
- Zhang Z, Jiang S, Tian H, Zeng Y, He K, Lin L, et al. Ethyl acetate fraction from Nymphaea hybrida Peck modulates inflammatory responses in LPS-stimulated RAW 264.7 cells and acute inflammation murine models. J Ethnopharmacol 2021;269:113698.
[Crossref] [Google Scholar] [PubMed]
- Ji M, Wang C, Yang T, Meng X, Wang X, Li M. Integrated phytochemical analysis based on UPLC–MS/MS and network pharmacology approaches to explore the effect of Odontites vulgaris Moench on rheumatoid arthritis. Front Pharmacol 2021;12:707687.
[Crossref] [Google Scholar] [PubMed]
- Liu Y, Zong T, Wang M, Yang D, Sun J, Zhou W, Li G. Chemical constituents from the whole plant of Odontites vulgaris Moench and their chemotaxonomic significance. Biochem Syst Ecol 2024;112:104764.
- Zeng X, Guo F, Ouyang D. A review of the pharmacology and toxicology of aucubin. Fitoterapia. 2020;140:104443.
[Crossref] [Google Scholar] [PubMed]
- Ying C, Wan D. Quantitative determination of total and individual flavonoids in stems and leaves of Buddleja davidii and Buddleja albiflora. Pharmacogn Mag 2012;8(32):273.
[Google Scholar] [PubMed]
- Garami A, Steiner AA, Romanovsky AA. Fever and hypothermia in systemic inflammation. Handbook Clin Neurol 2018;157:565-97.
[Crossref] [Google Scholar] [PubMed]
- Domscheit H, Hegeman MA, Carvalho N, Spieth PM. Molecular dynamics of lipopolysaccharide-induced lung injury in rodents. Front Physiol 2020;11:36.
[Crossref] [Google Scholar] [PubMed]
- Wang J, Liu YT, Xiao L, Zhu L, Wang Q, Yan T. Anti-inflammatory effects of apigenin in lipopolysaccharide-induced inflammatory in acute lung injury by suppressing COX-2 and NF-kB pathway. Inflammation 2014;37:2085-90.
[Crossref] [Google Scholar] [PubMed]