- *Corresponding Author:
- M. S. Reddy
Department of Pharmaceutics, Manipal College of Pharmaceutical Sciences, Manipal University, Manipal-576 104, India
E-mail: ms.reddy@manipal.edu
| Date of Submission | 23 March 2014 |
| Date of Revision | 23 November 2014 |
| Date of Acceptance | 17 April 2015 |
| Indian J Pharm Sci 2015;77(3):249-257 |
This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.
Abstract
Pharmaceutical research is focused in designing novel drug delivery systems to improve the bioavailability of poorly water soluble drugs. Self-microemulsifying drug delivery systems, one among the lipid-based dosage forms were proven to be promising in improving the oral bioavailability of such drugs by enhancing solubility, permeability and avoiding first-pass metabolism via enhanced lymphatic transport. Further, they have been successful in avoiding both inter and intra individual variations as well as the dose disproportionality. Aqueous insoluble drugs, in general, show greater solubility in lipid based excipients, and hence they are formulated as lipid based drug delivery systems. The extent of solubility of a hydrophobic drug in lipid excipients i.e. oil, surfactant and co-surfactant (components of self-microemulsifying drug delivery systems) greatly affects the drug loading and in producing stable self-microemulsifying drug delivery systems. The present review highlighted the influence of physicochemical factors and structural features of the hydrophobic drug on its solubility in lipid excipients and an attempt was made to explore the role of each component of self-microemulsifying drug delivery systems in the formation of stable microemulsion upon dilution.
Keywords
Lipid based delivery systems, partition coefficient, solubility, electrostatic interaction, interfacial tension
Development of new and effective drug molecules for the treatment of ever increasing ailments of mankind has been the goal of pharmaceutical research. Solubility and permeability of an active ingredient are the two most important parameters determining its absorption and bioavailability. It is estimated that around 40% of the currently available drug molecules in the market are poorly soluble in the aqueous media. Interestingly, most new drug molecules that are being developed due to the advances in in vitro screening methods and the combinatorial chemistry are poorly soluble or insoluble in aqueous media [1,2]. According to the biopharmaceutics classification system (BCS), drug substances are classified into four classes based on their aqueous solubility and permeability (fig. 1). A drug substance is considered highly soluble when the maximum daily dose is soluble in 250 ml of aqueous media over the pH range of 1-7.5. Whereas, a drug substance is considered highly permeable when >85% of the administered dose is absorbed [3]. According to Lipinski’s rule of five, a drug molecule with more than 5 H-bond donors, more than 10 H-bond acceptors, molecular weight >500 and logP >5 is considered poorly permeable [4]. Bioavailability of an active ingredient mainly depends upon its solubility in gastrointestinal tract fluids. Dissolution is the rate limiting step in absorption of poorly water soluble drugs. It is for this reason, the dose of poorly aqueous soluble drugs is increased to attain effective therapeutic drug concentration in the blood [5]. As the hydrophobic nature is inherent for a molecule which can not be altered, various approaches have been made to overcome this drawback. These include particle size reduction, formation of salts, co-crystals, amorphous formulations, pH modification, lipid based formulation and prodrug approach [6]. Each of these methods has some advantages as well as limitations and hence the selection of a particular approach is an important step in increasing their bioavailability. For example, salt formation of a poorly soluble drug which is either neutral, weak acid or weak base may not always be possible. Similarly, reduction of particle size of a drug makes it difficult for handling due to the development of static charges [7]. Further, stress of a mechanical size reduction process may cause deterioration of crystal structure and/or partial or complete amorphisation of the drug with a resultant decrease in the stability as observed with candesartan cilexetil [8]. On the contrary, micronization technique has been successfully used to produce smaller fenofibrate particles (Tricor®) which are dispersed in hydrophilic polyvinylpyrrolidone to reduce its dose [9]. Recently, increased attention is being directed towards the development of lipid based formulations for the delivery of poorly water soluble drugs with an aim to increase their dissolution and bioavailability. The rationale for this is an observable increase in the bioavailability of hydrophobic drugs when administered along with food, especially fatty meal. This is attributed to the fact that fat rich food may lead to one or more of the following such as it stimulates bile flow and pancreatic secretions, delays gastric emptying, change in gastrointestinal pH, enhances lymphatic transport, enhances mesenteric and liver blood flow, increases intestinal wall permeability, and reduces efflux activity. Such an observation led to the development of new guidelines established by the United States Food and Drug Administration (US FDA) entitled “food-effect bioavailability and fed bioequivalence studies” with recommendations for the determination of impact of food in the enhancement of bioavailability (IND) and fed bioequivalence (ANDA) studies [10].
Lipid-based dosage forms such as solid lipid nanoparticles (SLN), solid dispersions, selfmicroemulsifying drug delivery systems (SMEDDS), liposomes and super saturable self-emulsifying systems can be formulated by different formulation techniques. Both lipids (in the form of solid or liquid) and excipients (surfactants, co-surfactants, cosolvents) used in the formulation not only improve the bioavailability of the drug substance but also help in avoiding the erratic absorption. Such an ability of lipids and excipients is attributed to mechanisms such as maintaining the drug in the solubilized form, promoting lymphatic transport, altering the permeability of epithelia, altering enterocyte based drug transport and inhibiting the p-gp (permeability glycoprotein) efflux [11]. Among the lipid based formulations, there has been a growing interest in developing self-emulsifying systems as they are efficient in hydrophobic drug delivery. Self-emulsifying systems produce either emulsions or microemulsions upon dilution with water depending upon the nature of the oil, surfactant concentration and oil/surfactant ratio [12]. These systems can be in the form of self-emulsifying drug delivery systems (SEDDS), SMEDDS or selfnanoemulsifying drug delivery systems (SNEDDS). Smaller size of the droplets in both SMEDDS and SNEDDS compared to SEDDS offers several advantages such as improved bioavailability and minimized variable bioavailability. SMEDDS, in particular, produces microemulsions with droplet size in the nano range upon dilution. Microemulsions (ME) are solution-like systems with an inner structure of nano droplets stabilized by a set of surfactants and co-surfactants [13]. Often ME system is described as “swollen micelle” as these systems differ with micelles only in the size (micelles are thermodynamically stable consisting of spherical droplets having a radius of less than 20 nm). The basic concept in designing SMEDDS is to present a hydrophobic drug in a solubilized form in vivo that would otherwise exhibit dissolution rate-limited absorption [14]. This review is an overview of various components used in the formulation of SMEDDS and their role in formation of stable microemulsion.
Components
In order to formulate a successful SMEDDS for maximum therapeutic effect, due consideration must be given to various factors such as physicochemical properties of the active moiety as well as excipients, potential for drug excipient interaction (in vitro and in vivo) and physiological factors that promote or inhibit the bioavailability. Further, other important factors such as regulatory status, solubilization capacity, miscibility, physical state of the excipients at room temperature, digestibility and compatibility with capsule shell, chemical stability and cost of the materials should also be considered during the formulation [15]. Such a rationale approach not only helps in reducing the time involved in the formulation development but also reduces the cost of its development [11].
Oil/lipid phase
The function of oil phase in self-microemulsifying system is to solubilize the hydrophobic/lipophilic active moiety in order to improve both drug loading and bioavailability of the hydrophobic active moiety. Selection of oil plays a vital role in the formulation as it determines the amount of drug that can be solubilized in the system [16]. A lipid molecule with a large hydrophobic portion compared to hydrophilic portion is desirable as it maximizes the amount of drug that can be solubilized. Table 1 represents the most commonly used oils in the formulation of SMEDDS.
| Type of oil | Examples |
| Fixed oils (long?chain triglycerides) | Soybean oil, arachis oil, aastor oil, cottonseed oil, maize (corn) oil, hydrolyzed corn oil, olive oil, sesame oil, sunflower oil, palm oil, peanut oil, triolein |
| Medium?chain triglycerides and related esters | Caprylic/capric triglycerides (Akomed E, Akomed R, Miglyol 810, and Captex 355, Neobee M5®, Crodamol GTCC®), fractionated coconut oil (Miglyol 812), Captex 300, Labrafac CC, Triacetin |
| Medium?chain mono and di?glycerides | Mono and diglycerides of capric/caprylic acid. (Capmul MCM and Imwitor) |
| Long?chain mono glycerides | Glycerylmonooleate (Peceol, Capmul GMO), glycerylmonolinoleate (Maisine ?35) |
| Propylene glycol (PG) fatty acid esters | PG Diester of caprylic/capric acid (Labrafac PG), PG monocaprylic ester (Sefsol?218) PG monolaurate (Lauroglyc FCC, Lauroglycol90, Capmul PG?12) PG dicaprylate (Miglyol 840) |
| Caprylic/capric/diglyceryl succinate | Miglyol 829 |
| Fatty acids | Oleic acid (Crossential O94), Caprylic acid |
| Fatty acid esters | Ethyl Oleate (Crodamol EO), Ethyl butyrate, Isopropyl myristate, Isopropyl palmitate |
| Vitamins | Vitamin E |
| Mineral oil | Liquid paraffin |
Table 1: List Of Oils Used In Formulation Of SMEDDS
Long chain triglycerides
Lipids that have fatty acid chains of 14-20 carbons are categorized as LCTs [17]. Fixed oils i.e., vegetable oils contain a mixture of glyceride esters of unsaturated long chain fatty acids. These are considered safe as they are commonly present in daily food and are easily digestible [15]. Large hydrophobic portion of triglycerides is responsible for their high solvent capacity for lipophilic moieties. Though it is difficult to microemulsify, some marketed formulations such as Neoral® (composed of olive oil which, has shown superior oral bioavailability) and Topicaine® gel (composed of Jojoba oil for transdermal application) have been successfully practicing the microemulsification of LCTs [18].
Medium chain triglycerides and related esters
Lipids that have fatty acid chains of 6-12 carbons are categorized as MCTs [17]. MCTs are the most common choice of oil for SMEDDS as they are resistant to oxidation and possess high solvent capacity compared to LCT because of their high effective concentration of ester group. MCTs produced from the distillation of coconut oil are known as glyceryl tricaprylate and comprises of saturated C8 and C10 fatty acids in the liquid state [15]. Labrafac CM 10, a MCT, has shown superior solubility for fenofibrate and produced wider microemulsion region at all surfactant/co-surfactant combinations than Maisine 35, which, is a LCT [19]. Drug substance should possess minimum solubility of 50 mg/ml in LCTs for lymphatic absorption [20]. Upon digestion, products of short and medium chain triglycerides are directed towards portal vein whereas chylomicrons formed from LCTs triggers the lymphatic transport. Further, highly hydrophobic drug substances are easily soluble in vegetable oils and can easily be formulated as simple oil solutions which are readily emulsified in the gut. However, most conventional hydrophobic drug substances do not exhibit superior solubility in LCT such as vegetable oil [21,22].
Moderately hydrophobic drug substances, on the other hand, cannot be formulated into simple oil solutions as their solubility is limited. In such cases, SMEDDS are promising alternative where the drug solubility in the oil will be enhanced due to microemulsification of oil by surfactants. It is well accepted that oils with long hydrocarbon chains (high molecular volume) such as soybean oil, castor oil are difficult to microemulsify compared to MCT (low molecular volume) such as capmul MCM and Miglyol. However, solubilizing capacity of oil for lipophilic moiety increases with chain length (hydrophobic portion) of the oil. Hence the selection of oil is a compromise between the solubilizing potential and ability to facilitate the formation of microemulsion [23]. Malcolmson et al. studied the solubility of testosterone propionate in various oils for the formulation of O/W microemulsion and concluded that oils with larger molecular volume such as triglycerides show superior solubility than the corresponding micellar solution containing only surfactants without oil [24,25]. Enhancement of drug solubility in SMEDDS not only relies on the solubility of the drug in the oil but also on the surfactant(s). For instance, ethyl butyrate, small molecular volume oil, has shown higher solubility for testosterone propionate but its ME formulation has only improved the solubility slightly than the corresponding micellar solution. On the contrary, Miglyol 812 which is a larger molecular volume oil has shown improved solubilization in the ME formulation though the solubility of testosterone propionate is less in the individual components compared to ethyl butyrate [24].
Drug solubility in lipid
Oil component alters the solubility of the drug in SMEDDS by penetrating into the hydrophobic portion of the surfactant monolayer. Extent of oil penetration varies and depends on the molecular volume, polarity, size and shape of the oil molecule. Overall drug solubility in SMEDDS is always Oil component alters the solubility of the drug in SMEDDS by penetrating into the hydrophobic portion of the surfactant monolayer. Extent of oil penetration varies and depends on the molecular volume, polarity, size and shape of the oil molecule. Overall drug solubility in SMEDDS is always higher than the solubility of drug in individual excipients that combine to form SMEDDS. However, such higher solubility considerably depends on the solubility of drug in oil phase, interfacial locus of the drug and drug-surfactant interactions at the interface [26]. In light scattering experiments, it was observed that oils with small molecular volume act like co-surfactants and penetrate into the surfactant monolayer. This forms thinner polyoxyethylene chains near the hydrophobic core of the micelle disrupting the main locus of the drug solubilization due to which, a higher solubility of drug is not observed. Large molecular volume oils, however, forms a distinct core and do not penetrate effectively into the surfactant monolayer. The locus of drug solubilization was found to be effected by the microstructure and solubility of the drug in the excipients. The locus of drug solubilization was found to be at the interface of micelle for phytosterols whereas the same for cholesterol was found to be between the hydrophobic head groups of surfactant molecules. This is attributed to altered side chain flexibility of phytosterol due to the additional substitution of alkyl side chain compared to cholesterol [27].
In addition to molecular volume and polarity of the oil, drug solubility in oil is affected by physicochemical properties of drug molecule itself. Consideration of BCS classification and Lipinski’s rule of 5 for the selection of drug is only useful during initial screening stages. As per BCS classification, some of the acidic drugs are listed in Class II despite having good absorption and disposition as they do not satisfy the requirement of higher solubility at low pH values. Lipinski’s rule of 5, on the other hand, holds good only when the drug is not a substrate for the active transporter [4]. This suggests that aqueous solubility and log P alone are not sufficient to predict the solubility of drug in the oil. This further indicates that the solubility of any two drugs with similar log P would not be the same due to their different physicochemical properties.
To demonstrate this, a study was conducted in our laboratory with two antihypertensive drugs having close partition coefficient (log P) values, different aqueous solubility and varying physicochemical properties. Candesartan cilexetil is hydrophobic and has log P value of 7.3, molecular weight 610.66 g/mol with a polar surface area 135.77 whereas, valsartan is slightly soluble in aqueous phase with log P value of 5.3, molecular weight 434.53 g/mol with a polar surface area 103.48 (clogP and polar surface area were calculated using chembiodraw ultra 11.0). Unlike candesartan cilexetil, valsartan exhibits pH dependent solubility [28].
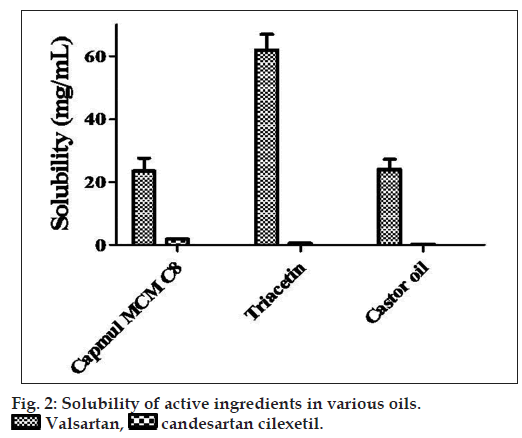
If only log P and aqueous solubility of these two drugs are considered, it is only natural to assume that candesartan cilexetil would be highly soluble in lipid phase whereas valsartan would be less soluble. A specific and sensitive HPLC-UV method was developed and validated to measure the super saturation solubility of these two drugs in various oils and the results showed a completely different solubility profiles. Solubility profile of these two drugs in different oil phase is given in fig. 2.
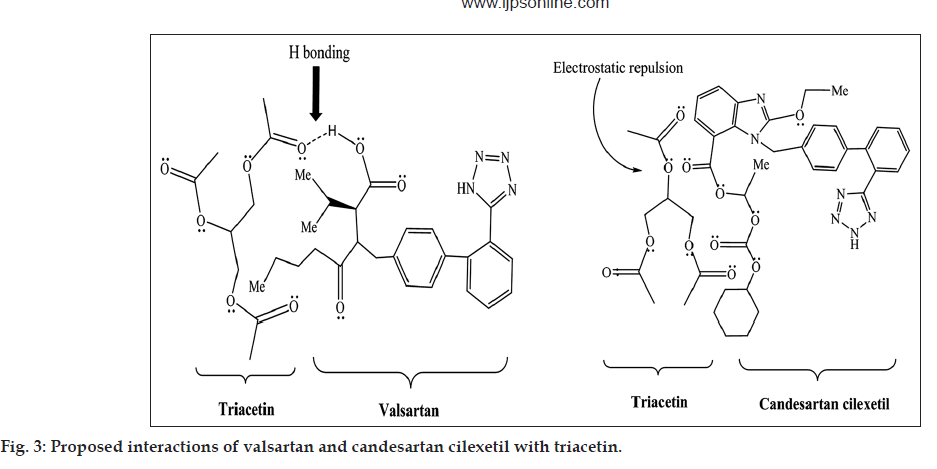
Although log P and polar surface area of valsartan and candesartan cilexetil are closer, their solubility with triacetin, castor oil and capmul MCM C8 differs significantly. This may be attributed to the hydrogen bonding capacity and electrostatic interaction of both the scaffold with the oils. Nevertheless, valsartan is having aliphatic carboxylic group which is expected to be involved in hydrogen bond interaction with the hydrogen acceptor functionality of the triacetin as well as castor oil. We assume that the branched chain aliphatic ester moiety of triacetin, capmul MCM C8 and castor oil gets involved in the electrostatic repulsion with cilexetil part of candesartan. In case of valsartan, such electrostatic interactions are not possible. Furthermore, aliphatic ester chain of triacetin and castor oil may solvate the lipophilic chain of valsartan more favorably than candesartan in the absence of any electrostatic repulsion (proposed interaction is shown in fig. 3). However, significant difference was not observed with other oils such as olive oil, peanut oil, corn oil, miglyol 810, sunflower oil and soybean oil (data not shown).
Surfactants
An amphiphilic molecule (surfactant), as the name indicates, is composed of polar group (hydrophilic) and nonpolar (lipophilic) group. The prime requirement in achieving ultra low interfacial tension at the oil water interface can be attained by selecting suitable amphiphile. Selection of surfactant primarily depends on the factors such as the efficiency and rapidity to microemulsify the selected oil, solubilizing capacity for the drug, safety (depending on the route of administration), type of emulsion to be formulated, cloud point of surfactant and ability to inhibit p-gp (if the active ingredient is the p-gp substrate) [29].
In SMEDDS, oil phase containing the drug is emulsified into nano sized droplets. As the size of oil phase containing drug decreases to nano size, the effective surface volume increases by several folds and therefore, it requires large concentration of surfactant for its stabilization [30]. However, too large a concentration of surfactant was found to increase the droplet size of the emulsion as it enhances the penetration of water into oil droplet with interfacial film getting disrupted leading to expulsion/bulging of the film into the aqueous phase [16]. Further, the concentration of surfactant also depends on molecular volume of the oil. In general, large molecular volume oils such as LCTs require higher surfactant than short chain mono/diglycerides.
Based on the hydrophilic–lipophilic balance (HLB) value of the surfactant, it may form O/W (water is the continuous phase), W/O (oil is the continuous phase) or bicontinuous (comprises equal amount water and oil) microemulsion. Upon dispersion of surfactant in water or oil and water mixture, depending on intermolecular forces, it self-associates, into a variety of equilibrium phases in the form of spherical, hexagonal, rod shaped, lamellar phase micelles whereas reverse micelles may form in apolar liquids such as alkanes. However, they will form interface when dispersed in the mixture of oil and water to make the system thermodynamically stable [31].
During the formulation of SMEDDS, nonionic surfactants are preferred over ionic surfactants as they are less toxic. Further, it was observed that for the same alkyl chain length of ionic and nonionic surfactants, critical micelle concentration [CMC] value (mol/l) is less for nonionic surfactants. Hence, they are only required in less concentration for effective reduction in interfacial tension. Polyoxyethylene surfactants, nonionic, are widely used in most formulations today [32]. Some of the commonly used polyoxyethylene surfactants with HLB values are represented in Table 2.
| Chemical name | Commercial name | HLB |
|---|---|---|
| POE Sorbitanmonolaurate | Tween 20 | 17 |
| POE Sorbitanmonopalmitate | Tween 40 | 15.6 |
| POE Sorbitanmonostearate | Tween 60 | 15.0 |
| POE Sorbitanmonooleate | Tween 80 | 15.0 |
| POE Sorbitantristearate | Tween 65 | 10.5 |
| POE Sorbitantrioleate | Tween 85 | 11.0 |
| POE glycerol trioleate | Tagat TO | 11.5 |
| POE?40?Hydrogenated | Cremophore RH 40 | 14.0 to 16.0 |
| castor oil | (solid) | |
| POE?35?Castor oil | Cremophore EL (liquid) | 12.0?14.0 |
| POE (10) Oleyl ether | Brij 96 | 12.4 |
| POE (23) Lauryl ether | Brij 35 | 16.9 |
| POE?Vitamin E | Alpha?tocopherol TPGS | 13.0 |
Table 2: Commonly Used Polyoxyethylene Surfactants
Co-surfactants/co-solvents
ME formation from SMEDDS is dynamic and its equilibrium is maintained by dynamic exchange of substances/molecules between dispersed phases that occur continuously. Dynamic process involves the exchange of water between bound and free states, exchange of co-surfactant from the interfacial film to continuous phase and dispersed phase and exchange of surfactant between the interfacial film and water [30]. This dynamic behavior of ME is due to the flexibility of the interfacial film which is imparted mainly by the presence of co-surfactant. Both type and concentration of co-surfactant play a role in the formulation of SMEDDS. For example, SMEDDS with low molecular weight co-surfactants are not advisable to fill into gelatin capsules since they may get absorbed into capsule shells which may lead to drug precipitation [15]. Similarly, higher co-surfactant concentration also results in precipitation of drug upon dilution of SMEDDS due to partitioning of co-surfactant into aqueous phase [31]. Ethanol, propylene glycol, PEG and Transcutol P are some of the widely used co-surfactants.
Formulation of SMEDDS
Solubility studies of the active ingredient in the components of SMEDDS are the primary preformulation tests which help in the selection of suitable excipients for the initial formulation optimization. Excipients i.e., oil, surfactant and co surfactant in which drug show superior solubility are selected to achieve optimum drug loading and to minimize the final volume [33,34].
Pseudoternary phase diagrams are constructed with the selected excipients by titration method and by keeping the ratio of any two of the four components as constant (usually surfactant and co surfactant). This is mixed with required volume of the third phase (usually oil) in different ratios. Each of individual mixture is titrated with fourth component usually water in incremental amounts and tested for the clarity, flowability, time for self emulsification and dispersibility [35,36]. Compositions, which form clear solutions, are denoted by suitable symbols in ternary diagram, joined and the area indicate the microemulsion existing area. Wider area indicate the good self-emulsification capacity [37].
From pseudoternary phase diagram, formulation is optimized mainly based on globule size, selfemulsification time. SMEDDS can be easily prepared in single step by dissolving the drug in the selected composition of excipients by simple mixing with no critical steps.
Microemulsion formation
Prior knowledge of interfacial tension between the oil and water, changes in surface area upon emulsification of the oil and entropy changes in the system helps in understanding the theory involved in the microemulsion formation. [31,38]
Though, the interfacial tension γ between the oil and water remains constant, the area of the bulk oil (A1) droplets will be enormously enhanced upon emulsification due to reduced size (A2) therefore surface free energy (ΔA γ) and entropy of the system will be increased. The formation of microemulsion from SMEDDS according to the second law of thermodynamics can be expressed as, ΔG=γ ΔA–T ΔS, where, ΔG is the free energy of formation, ΔS is the change in the entropy of the system, γ is the interfacial tension and T is the temperature of the system.
A positive ΔG value is considered to be unstable and the system formation is non-spontaneous. In the above mathematical expression, change in surface area (ΔA) and entropy (TΔS) are constant for a particular combination of system. Hence, reducing the interfacial tension (γ) to zero results in negative free energy that makes the system thermodynamically stable and favors the spontaneous emulsification. The formulation of SMEDDS should aim to reduce the interfacial tension to minimum possible. The ultralow interfacial tension can be achieved by adding a third component in the form of co-surfactant which will aid the surfactant activity in reducing the interfacial tension. If surfactant concentration alone is increased to reduce the interfacial tension, CMC reaches before the γ is reduced to zero.
Biopharmaceutical aspects
SMEDDS produces O/W microemulsion upon contact with GI fluids with the aid of gastrointestinal motility. Drug release from the microemulsion takes place by partition from droplet to into intestinal fluids [16]. Easy partitioning of drug from the droplets is made possible due to the formation of small droplets with enormous interfacial area [39].
As the dissolution is rate-limiting step for the absorption of poor soluble drugs, SMEDDS can bypass dissolution which in turn enhances bioavailability [26]. Components of SMEDDS contribute in the improvement of bioavailability of drug. Lipid/ oil alter the in vivo fate of drug/dosage form by increasing the solubility in turn dissolution rate and by promoting lymphatic transport. Drug can be protected by chemical and enzymatic means as it is presented in the form of oil droplets [16,40]. Surfactants increase the permeability of drug by disturbing the structural organization of membrane components thus facilitating passive diffusion [16]. Surfactants of high HLB like Tween 80 are reported to increase the permeability of the drug by their loosening effect on tight junctions [41]. Many surfactants like Tween 80, Spans, Cremophors (EL and RH40), Pluronics are reported to have inhibitory action on efflux transporters which helps in improving the bioavailability of the drugs (substrates to the efflux pumps) [42]. The same was proved incase of paclitaxel, with the formulation comprising polysorbate 80 due to its inhibitory effect on to p-glycoprotein transporter [43].
Recent study proved that bioavailability enhancement of fenofibare, a BSC class II drug was significantly higher using SMEDDS compared to solid dispersion pellets [44]. Another study conducted using cyclosporine, a BCS class IV drug, concluded significant bioavailability enhancement by SMEDDS compared to polymeric nanoparticles, which is due to multiple absorption mechanisms, like enhanced dissolution rate and improved permeability [45].
Conclusions
SMEDDS, one of the lipid based dosage forms, are easy to manufacture and have the advantage of improving the solubility of hydrophobic drug with the aid of surfactant and co-surfactant which inturn improves the bioavailability thereby reduces the dosage requirement and avoids erratic absorption profiles (food effect). However, careful selection of oil, surfactant and co-surfactant plays an important role in the successful formulation of SMEDDS such as higher drug loading with suitable oil in required/ minimum amounts. During the selection of each of these ingredients, physicochemical properties of the active ingredient must also be given due consideration as it greatly affects solubilization capacity of the system. With better understanding of the relationship between the log P value, aqueous solubility and physicochemical properties of the active ingredients and their effect on solubility would help in formulating more hydrophobic drugs in the form of SMEDDS.
Financial support and sponsorship
Authors are thankful to Department of Science and Technology (DST, Nanomission, Project S.No: SR/ NM/NS-47/2008) Government of India for providing financial.
Conflict of interest
There are no conflicts of interest.
References
- Dahan A, Hoffman A. Enhanced Gastrointestinal Absorption of Lipophilic Drugs. In: Barry WB, Touitou E, editors. Enhancement in Drug Delivery. New York: CRC Press; 2007, p. 111-32.
- Takagi T, Ramachandran C, Bermejo M, Yamashita S, Yu LX, Amidon GL. A provisional biopharmaceutical classification of the top 200 oral drug products in the United States, Great Britain, Spain, and Japan. Mol Pharm 2006;3:631-43.
- Shargel L, Wu-pong S, Yu AB. Applied Biopharmaceutics and pharmacokinetics. USA: McGraw-Hill Companies; 2005.
- O’Driscoll CM, Griffin BT. Biopharmaceutical challenges associated with drugs with low aqueous solubility- The potential impact of lipid-based formulations. Adv Drug Deliv Rev 2008;60:617-24.
- Kawabata Y, Wada K, Nakatani M, Yamada S, Onoue S. Formulation design for poorly water-soluble drugs based on biopharmaceuticsclassification system: Basic approaches and practical applications. Int J Pharm 2011;420:1-10.
- Shah HN, Phuapradit W, Zhang Y, Sandhu H, Linzhang W, Malick W. Approaches for improving bioavailability of poorly soluble drugs. In: Augsburger LL, editor. Pharmaceutical Dosage Form: Tablets. New York: Informa Healthcare; 2008. p. 51-104.
- Serajuddin AT. Solid dispersion of poorly water-soluble drugs: Early promises, subsequent problems and recent breakthroughs. J Pharm Sci 1999;88:1058-66.
- Legen I. Self-microemulsifying drug delivery systems. 2008. European Patent 1 961 412.
- Wei JD, Ho HO, Chen CH, Ke WT, Chen ET, Sheu MT. Characterisation of fenofibrate dissolution delivered by a self-microemulsifying drug-delivery system. J Pharm Pharmacol 2010;62:1685-96.
- Food and Drug Administration, Guidance for Industry: Food-Effect Bioavailability and Fed Bioequivalence Studies. Rockville, USA: Guidance for Industry; 2002.
- Jannin V, Musakhanian J, Marchaud D. Approaches for the development of solid and semi-solid lipid-based formulations. Adv Drug Deliv Rev 2008;60:734-46.
- Gershanik T, Benita S. Self -dispersing lipid formulations for improving oral absorption of lipophilic drugs. Eur J Pharm Biopharm 2000;50:179-88.
- Spernath A, Aserin A. Microemulsions as carriers for drugs and nutraceuticals. Adv Colloid Interface Sci 2006;128-130:47-64.
- Cannon BJ, Long AM. Emulsions, Microemulsions, and Lipid-Based Drug Delivery Systems for Drug Solubilization and Delivery-Part II: Oral Applications. In: Liu R, editors. Water-Insoluble Drug Formulation. New York: CRC Press; 2008. p. 228-50.
- Pouton CW, Porter CJ. Formulation of lipid-based delivery systems for oral administration: Materials, methods and strategies. Adv DrugDeliv Rev 2008;60:625-37.
- Gursoy RN, Benita S. Self-emulsifying drug delivery systems (SEDDS) for improved oral delivery of lipophilic drugs. Biomed Pharmacother 2004;58:173-82.
- Prajapati NH, Patel DP, Patel NG, Dalrymple DM, Serajuddin AT. Effect of difference in fatty acid chain lengths of medium chain lipids on lipid/surfactant/water phase diagrams and drug Solubility. J Excipients Food Chem 2011;2:73.
- El Laithy HM, El-Shaboury KM. The development of cutinalipogels and gel micromeulsion for topical administration of fluconazole. AAPS PharmSciTech 2002;3:E35.
- Patel AR, Vavia PR. Preparation and in vivo evaluation of SMEDDS (Self-Microemulsifying Drug Delivery System) containing fenofibrate. AAPS J 2007;9:E344-52.
- Yanez JA, Wang SW, Knemeyer IW, Wirth MA, Alton KB. Intestinal lymphatic transport for drug delivery. Adv Drug Deliv Rev 2011;63:923-42.
- Stegemann S, Leveiller F, Franchi D, de Jong H, Lindén H. When poor solubility becomes an issue: From early stage to proof of concept. Eur J Pharm Sci 2007;31:249-61.
- Pouton CW. Formulation of self-emulsifying drug delivery systems. Adv Drug Deliv Rev 1997;25:47-58.
- Patravale VB, Date AA. Microemulsions: Pharmaceutical Applications. In: Stubenrauch C. editor. Microemulsions: Background, New Concepts, Applications, Perspectives, United Kingdom: Blackwell Publishing Ltd; 2009. p. 259-93.
- Malcolmson C, Satra C, Kantaria S, Sidhu A, Lawrence MJ. Effect of oil on the level of solubilizationof testosterone propionate into nonionic oil-in-water microemulsions. J Pharm Sci 1998;87:109-16.
- Bandivadekar MM, Pancholi SS, Kaul-Ghanekar R, Choudhari A, Koppikar S. Self-microemulsifying smaller molecular volume oil (Capmul MCM) using non-ionic surfactants: A delivery system for poorly water-soluble drug. Drug DevInd Pharm 2012;38:883-92.
- Narang AS, Delmarre D, Gao D. Stable drug encapsulation in micelles and microemulsions. Int J Pharm 2007;345:9-25.
- Rane SS, Anderson BD. What determines drug solubility in lipid vehicles: Is it predictable?.Adv Drug Deliv Rev 2008;60:638-56.
- Joshi Y, Kennedy JJ, Li P, Royce AE, Wagner RF. Microemulsion dosage forms of Valsartan and Methods of Making the Same. 2008, WO/2008/073731.
- Date AA, Nagarsenker MS. Parenteral microemulsions: An overview. Int J Pharm 2008;355:19-30.
- Bagwe RP, Kanicky JR, Palla BJ, Patanjali PK, Shah DO. Improved drug delivery using microemulsions: Rationale, recent progress, and new horizons. Crit Rev Ther Drug Carrier Syst 2001;18:77-140.
- Lawrence MJ, Rees GD. Microemulsion-based media as novel drug delivery systems. Adv Drug Deliv Rev 2009;45:89-121.
- Jiao J. Polyoxyethylated nonionic surfactants and their applications in topical ocular drug delivery. AdvDrug Deliv Rev 2008;60:1663-73.
- Wang Y, Sun J, Zhang T, Liu H, He F, He Z. Enhanced oralbioavailability of tacrolimus in rats by self micro emulsifying drug delivery system. Drug DevInd Pharm 2011;37:1225-30.
- Singh AK, Chaurasiya A, Awasthi A, Mishra G, Asati D, Khar RK, et al. Oral Bioavailability enhancement of exemestanefromself-microemulsifying drug delivery system (SMEDDS). AAPS Pharm SciTech 2009;10:906-16.
- Hu L, Wu H, Niu F, Yan C, Yang X, Jia Y. Design of fenofibratemicroemulsion for improved bioavailability. Int J Pharm 2011;420:251-5.
- Singh AK, Chaurasiya A, Singh M, Upadhyay SC, Mukherjee R,
- Zhang H, Cui Y, Zhu S, Feng F, Zheng X. Characterization and antimicrobial activity of a pharmaceutical microemulsion. Int J Pharm 2010;395:154-60.
- Tadros FT. Applied surfactants principles and applications. Weinheim: Wiley-VCH Verlag GmbH & Co. KGaA; 2005.
- Tang J, Sun J, He Z. Self-emulsifying drug delivery systems: Strategy for improving oral delivery of poorly soluble drugs. Curr Drug Ther 2007;2:85-93.
- Akula S, Gurram AK, Devireddy SR. Self-Microemulsifying Drug Delivery Systems: An Attractive Strategy for Enhanced Therapeutic Profile. IntSch Res Notices 2014;2014:Article ID 964051.
- Buyukozturk F, Benneyan JC, Carrier. Impact of emulsion-based drug delivery systems on intestinal permeability and drug release kinetics. J Control Release 2010;142:22-30.
- Bansal T, Akhtar N, Jaggi M, Khar RK, Talegaonkar S. Novel formulation approaches for optimizing the delivery of anti cancer drugs based on p-glycoprotein modulation. Drug Discov Today 2009;14:1067-74.
- Nornoo AO, Zheng H, Lopes LB, Johnson-Restrepo B, Kannan K, Reed R. Oral microemulsions of paclitaxel: In situ and pharmacokinetic studies. Eur J Pharm Biopharm 2009;71:310-7.
- Weng T, Qi J, Lu Y, Wang K, Tian Z, Hu K, et al. The role of lipid-based nano delivery systems on oral bioavailability enhancement of fenofibrate, a BCS II drug: Comparison with fast-release formulations. J Nanobiotechnol 2014;12:39-46.
- Wang K, Qi J, Weng T, Tian Z, Lu Y, Hu K, et al. Enhancement of oral bioavailability of cyclosporine A: Comparison of various nanoscale drug-delivery systems. Int J Nanomedicine 2014;28:4991-9.

 . Valsartan,
. Valsartan,  candesartan cilexetil.
candesartan cilexetil.