- Corresponding Author:
- E. Venkata Rao
Natco Research Centre, B-13, Industrial Estate, Sanathnagar, Hyderabad - 500 018, Andhra Pradesh, India
E-mail: erraguntlavrao@yahoo.co.uk
| Date of Submission | 23 September 2010 |
| Date of Revision | 2 May 2011 |
| Date of Acceptance | 5 May 2011 |
| Indian J Pharm Sci, 2011, 73 (3): 262-270 |
Abstract
A new strategy for the synthesis of curcuminoids is described involving the reaction of acetylacetone difluroboronite with an aromatic aldehyde in the presence of n-butylamine as catalyst. The new intermediate products, curcuminoid difluroboronites, of symmetrically substituted curcuminoids like curcumin and bisdemethoxycurcumin are stable, can be isolated and hydrolysed with aq. methanol at pH 5.8 to get the curcuminoids of high purity. The method is applicable for unsymmetrical curcuminoids like demethoxycurcumin also with some modification involving column chromatography. The intermediate curcuminoid difluroboronites, as also the natural β-diketone pongamol difluroboronite, prepared for the first time were characterized on the basis of physical and chemical properties and spectroscopic data. The advantage of using borontrifluoride to protect the enol group in acetylacetone over the generally used boric oxide is brought out. The importance of conducting biological activity studies using pure curcuminoids is explained.
Keywords
Curcuminoids, pongamol, difluroboronites, synthesis, structure, cleavage, β-Diketones
Turmeric (Curcuma longa Linn., Zingiberaceae) is extensively cultivated in India and other south-east Asian countries. In contrast to turmeric’s limited use in western countries as a coloring agent, it is widely used in the countries of origin as a food additive and medicinal agent. The main components of turmeric are curcuminoids, which in general are 1,7-diaryl-1,6- diene-3,5-dione-heptanoids. Of these, curcumin (CI), demethoxycurcumin (CII) and bisdemethoxycurcumin (CIII) are important.
Curcumin has been reported to have a wide spectrum of biological activities, chiefly antibacterial, antioxidant, antitumor, and carcinogenic [1] and neurological [2]. Clinically curcumin has potential usefulness in treating post-operative inflammation [3], inflammatory bowel disease [4], debilitating illnesses like Crohn’s disease or ulcerative colitis [5] and Alzheimier’s disease [6].
Many of the biological activity studies were carried out on curcumin obtained from commercial sources, which could be mixtures of CI (1), CII (2) and CIII (3). The three components differ in their solubility and methoxy groups, which can influence the biological activity. In contrast to the general belief that CI is the most important active component of turmeric, there are a few biological activity studies carried out on pure components, obtained by isolation [7,8] or synthesis [9], which show that CIII is more potent than CI.
The isolation of pure compounds from turmeric is considered as a difficult and costly procedure. So far the three related compounds were separated by preparative TLC only. In connection with our work (unpublished) on the isolation of pure curcuminoids from turmeric, we turned our attention to synthetic aspects of curcuminoids to obtain pure samples for comparison [10].
The major processes of practical utility for the preparation of the symmetrical curcuminoids, CI and CIII, follow Pabon’s route [11] involving the reaction of vanillin or 4-hydroxybenzaldehyde and acetylacetone in the presence of boric oxide, trialkylborate and a primary amine. The method was extended by Roughley and Whiting [12] using both vanillin and 4-hydroxybenzaldehyde for the synthesis of the unsymmetrical curcuminoid, CII. The synthetic strategy of Pabon was followed by Graf [13] omitting the use of trialkylborate, but heating at 120-1300. A US patent [14] described a process for the synthesis of CI and related compounds making use of boric oxide, trialkylborate along with a chemically inert, highly polar aprotic solvents, especially organic amides. The boron complex of acetylacetone and subsequent condensation with aldehyde produce water in the reaction system reducing the yield of CI and hence the need for trialkylborate as a water scavenger. Preparation of CI on laboratory scale as well as commercial scale invariably follow Pabon’s route [11] with some variation.
As boric oxide is expensive Dinesh Babu and Rajasekharan [15] replaced it with boric acid. Their procedure involves purification by extensive column chromatography. Following Pabon’s method [11], we tried to replace boric oxide with boric acid, but it did not work in our hands. Krackov and Bellis [14] reported an yield of 5% for CI when they replaced boric oxide with boric acid in their work. The purpose of boric oxide is to protect the enol group in acetylacetone to prevent the highly acidic methylene protons from Knoevenegal condensation such that the condensation can occur at the terminal methyl groups of the diketone.
Against this background we have tried an alternative procedure for protecting the enol group in acetylacetone for the synthesis of CI. Our patented process [10], involving the use of borontrifluoride in place of boric oxide, is elaborated and discussed in this paper covering the chemical and spectral aspects. Further the study was extended to the synthesis of CII and CIII.
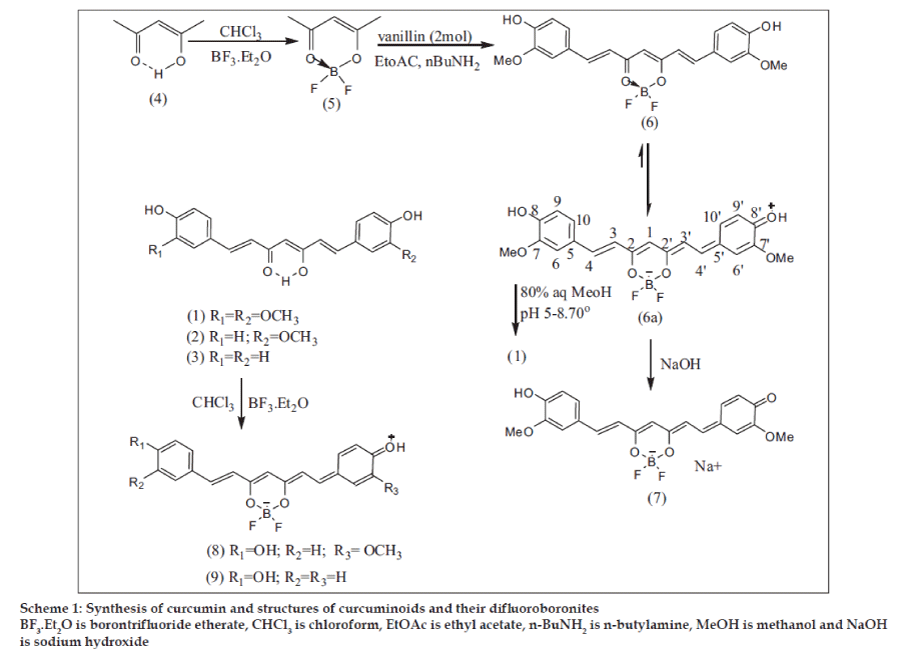
While studying the chemical properties of CI (1), we had occasion to try borontrifluoride etherate (BF3.Et2O) in CHCl3 medium at 0-50 to effect demethylation [16]. No demethylation occurred but a hitherto unreported CI derivative was formed. When the reaction was carried out in Ac2O-Et2O medium, the corresponding diacetyl compound was formed with amazing ease. This was also the case with CII (2) and CIII (3). CI was chosen for a more detailed study.
A search in literature revealed that β-diketones (acetylacetone, benzoylacetone and dibenzoylmethane) react with BF3.Et2O in benzene solution affording chelated difluoroboronites [17], initially termed as boron-β-diketofluorides [18]. No spectral data is available in literature for comparative study. We have prepared for the first time such a derivative for pongamol [19,20], a natural β-diketone, besides the known acetylacetonedifluroboronite [18] (AADFB) (5) for our study. Since curcuminoids and acetylacetone are all β-diketones and react with BF3.Et2O in the same way, we envisaged that AADFB in which the enol group is already protected can be used in place of acetylacetone and boric oxide for curcuminoids synthesis.
Materials and Methods
AADFB was prepared as per the procedure in literature [18]. TLC was carried out on silica gel 60 F254 Merck precoated sheets in solvent system CHCl3- MeOH (9.9:0.1) and (9.6:0.4) for curcuminoids and derivatives and Hexane-EtOAc (7:3) for pongamol and derivative. Column chromatography (CC) was carried out on silica gel 100-200 mesh. Aqueous methanol used for hydrolysis is methanol-water (80:20). Stirring during reaction was mechanical using magnetic stirrer or paddle stirrer depending on the volume of reaction mixture. HPLC was carried out on Novopak (150×3.9 mm) column using wavelength 248 nm for detection. Mobile phase was a mixture of acetonitrile and phosphate buffer (0.05M, pH 3) (500:500). RT stands for room temperature. Water used is demineralised water.
General procedure for the synthesis of CI and CIII (Scheme 1)
The aromatic aldehyde, vanillin or 4-hydroxybenzaldehyde (0.05 mol), was dissolved in EtOAc (40 ml) and AADFB (0.025 mol) added in small lots under stirring at RT. n-Butylamine (n-BuNH2) (0.0045 mol) was added in small lots with a microsyringe over 40 min. After the addition, stirring was continued for 3-4 h. The solvent was distilled off from the reaction mixture.
Isolation of curcuminoid difluoroboronite
To the reaction mixture, water (60 ml) was added followed by 1N HCl drop wise under heating at 600 and stirring until a pH of 2-3 was reached. The separated red solid was filtered, washed free from acid with water and dried. A small portion was purified for spectral analysis by passing the acetone solution through a small column of silica gel and evaporating the acetone eluate. The above reaction can be conducted in CHCl3 medium in place of EtOAc, and morpholine can replace n-BuNH2 as base catalyst. However, we occasionally observed unreacted aromatic aldehyde when morpholine was used.
Hydrolysis of curcuminoid difluoroboronites
The difluoroboronite solid of CI (CIDFB) or CIII (CIIIDFB) was dissolved in aqueous MeOH (ca. 200 ml for 1 g of solid), the pH adjusted to ca. 5.8 by drop wise addition of 1% aqueous NaOH and heated at 70º under vigorous stirring for 3-4 h, when the deep red solution turned orange. Water was added and distilled under mild vacuum to remove most of the methanol. The resulting suspension was cooled to 150 under stirring and the solid formed (CI or CIII) was filtered, washed with water and dried. In cases where the product contained a small quantity of the unhydrolyzed product (colour and TLC), the product was dissolved in 5% aqueous NaOH and extracted with CHCl3 to remove the impurity. The CHCl3 layer was rejected. The alkali layer is immediately cooled to 50 and acidified to pH 3 with conc. HCl under stirring. The precipitated solid (CI or CIII) was filtered, washed free from acid with water and dried.
Following the above procedure CI and CIII were prepared using the quantities mentioned here (as an example). CI from vanillin (9.5 g, 0.062 mol), AADFB (4.65 g, 0.031 mol), n-BuNH2 (0.625 ml, 0.0056 mol), yellow crystalline powder from aqueous MeOH, m.p. 181-830, yield 72-77%, HPLC purity 98.2 to 99.6%. UV, IR provided in (Table 1), and 1H NMR in (Table 2).
CIDFB (6a): Red powder, m.p. 230-320, yield 88- 95%, UV, IR (Table 1), 1H NMR (Table 2), MS (20 ev) m/z (rel.int.), 416 (M+, 25.4), 350 (M+-BF2OH, 100), 281 (3.4), 273 (8.5), 177 (C10H9O3 +, 62.7), 138 (83), 124 (53.4).
CIII (3) from 4-hydroxybenzaldehyde (2.463 g, 0.02 mol), AADFB (1.48 g, 0.013 mol), n-ButNH2 (0.180 ml, 0.0016 mol), orange plates from CHCl3-CH3CN, m.p. 231-33°, yield 60-65%, HPLC purity 98.5- 99.5%, UV, IR (Table 1). CIIIDFB (9): Red powder, m.p. 221-24°, yield 80-90%, UV (Table 1).
Synthesis of CII
Vanillin (1.531 g, 0.01 mol), 4-hydroxybenzaldehyde (1.231 g, 0.01 mol), AADFB (1.5 g, 0.113 mol) and n-BuNH2 (0.18 ml, 0.0016 mol) were used to carry out the reaction in EtOAc as described under CI and CIII. Difluoroboronites of CI, CII and CIII were formed (TLC). By the usual workup no solid separated. The product was extracted with EtOAc and washed with water, dried (Na2SO4) and evaporated. The resulting solid (3.9 g) contained CII difluoroboronite (CIIDFB) (8) as the major component and appeared as the middle spot in TLC, The mixture of difluoroboronites could not be separated by CC.
The mixture (2 g) was hydrolyzed with aq. MeOH as described under CI and CIII resulting in the formation of CI, CII and CIII (1.77 g). The mixture (1 g) was subjected to CC and eluted with CHCl3 and CHCl3- MeOH mixtures. CHCl3 eluates gave CI. The initial eluates of 1% MeOH in CHCl3 gave a mixture of CI and CII. The later eluates gave pure CII (0.32 g). The 2% MeOH in CHCl3 gave CII and CIII mixture. Roughly and Whiting [12] used preparative TLC for separation of the three components.
CII (2): Orange brown crystalline powder from CHCl3-CH3CN, m.p. 170-72°, yield 32-35%, HPLC purity 97.5-98.5% UV, IR (Table 1).
Preparation of CIDFB from CI
To the CHCl3 solution of CI (23 mg in 1 ml) BF3. Et2O (0.5 ml) was added and kept in cold for 5 h. It was then poured into cold water, extracted with CHCl3, the extract washed with water, dried (Na2SO4), evaporated and weighed (23.8 mg). The product was identical in all respects with the synthetic product mentioned above (Scheme 1).
Preparation of CIIDFB for UV spectra
CII (3 mg) was treated with CHCl3 (10 ml) and BF3. Et2O (0.2 ml) and kept at RT for 1 h. CHCl3 was evaporated and the residue washed with water by decantation a few times, dissolved in acetone and evaporated to dryness. The residue was dissolved in MeOH for UV spectral measurement (Table 1).
| Compound | UV λmax nm (log ε) | IR Flat υmax cm-1 | |
|---|---|---|---|
| MeOH | MeOH+alkali | ||
| 13 | 349 (3.94) | - | 2923,1600,1567,1537,804 |
| 14 | 389 (4.08) | - | 1590,1538,1491,812 |
| 1 | 430 (4.71) | 486 (4.68) | 3386,2875,1628,1603,1510,962,836 |
| 6a | 501 (4.82) | 629 (4.78) | 3448,2924,1592,1541,1508,975,817 |
| 10 | 462 (4.36) | - | 1764,1618,1599,1588,1544,1508,965,846 |
| 2 | 424 (4.68) | 470 (4.59) | 3423,2922,1625,1602,1585,1511,960,826 |
| 8 | 493 | 611 | - |
| 11 | 458 (4.32) | - | 1762,1624,1599,1545,1507,981,836 |
| 3 | 418 (4.77) | 465 (4.69) | 3432,1629,1601,1583,1576,1511,976,833 |
| 9 | 486 | 594 | - |
| 12 | 454 (4.31) | - | 1756,1621,1598,1582,1542,1276,978,836 |
Table 1: Uv And Ir Data Of Compounds
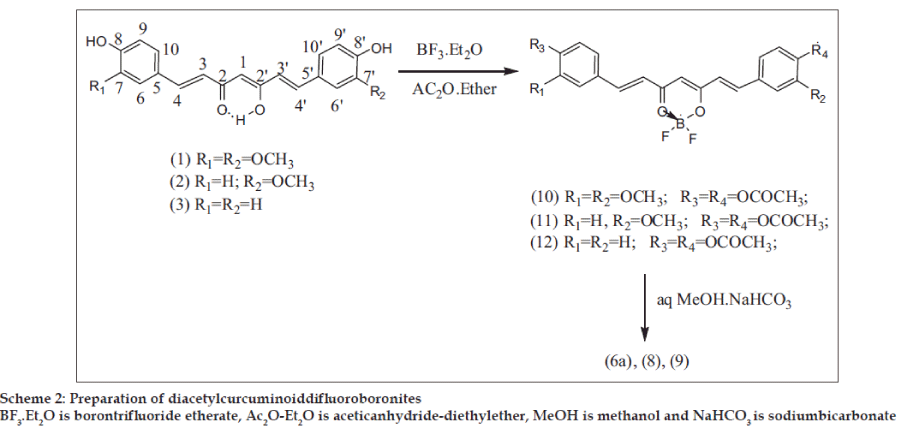
Preparation of diacetyldifluoroboronites of CI, CII and CIII
The curcuminoid (50 mg) was dissolved in acetic anhydride (3 ml) and ether (2 ml) and treated with BF3.Et2O (1 ml), stoppered and kept in the cold for 24 h. It was then poured into cold water under stirring and kept at RT for 2 h. The light yellow solid that separated was filtered, washed with cold aqueous NaHCO3, followed by water and dried. The product was purified by passing acetone solution through a small column of silica gel and eluting with acetone (Scheme 2).
Diacetyl CIDFB (10) (53.4 mg): m.p. 226-29°, UV, IR (Table 1), 1H NMR (Table 2), MS (20 ev) m/z (rel.int), 500 (M+, 6) 458 (M+-C2H2O, 39), 416 (M+-C4H4O2, 32.2), 350 (M+-C4H4O2-BF2OH,100), 282 (4.2), 238 (7.2), 177 (C10H9O3 +, 43.2), 124 (4.7). Diacetyl CIIDFB (11) (54.2 mg): m.p. 219-21°, UV, IR (Table 1). Diacetyl CIIIDFB (12) (56.3 mg): m.p. 236-39°, UV, IR (Table 1).
| H | (1)a,b | (6a)c | (12)a | |
|---|---|---|---|---|
| 4,4' | 7.59, 2 H, d (15.8) | 7.89, 2 H, d (15.6) | 8.03, 2 H, d (15.6) | |
| 6,6' | 7.15, 2 H, d (2) | 7.41, 2 H, d (1.6) | 7.20 to 7.25, 4 H, m | |
| 10,10' | 7.08, 2 H, dd (1.8, 9) | 7.30, 2 H, dd (1.9,8.6) | ||
| 9,9' | 6.93, 2 H, d (8.2) | 6.91, 2 H, d (8) | 7.11, 2 H, d (8) | |
| 3,3' | 6.48, 2 H, d (15.8) | 6.93, 2 H, d (14.6) | 6.67, 2 H, d (15.6) | |
| 1 | 5.81, 1 H, s | 6.38, 1 H, s | 6.11, 1 H, s | |
| 7,7'-2 OMe | 3.97, 3.95, 6 H, s, s | 3.94, 3.87, 6 H,s,s | 3.90, 6 H, s | |
| 8,8'-2 OAc | - | - | 2.34, 6 H, s | |
Table 2: 1h Nmr Data Of Compounds (1), (6a) And (12) (200 Mhz) Ppm From Tms As Internal Standard, J (Hz)
Deacetylation of (10): The substance (10 mg) was treated with NaHCO3 in MeOH (0.8 ml) (prepared by mixing 0.1 ml of saturated aq NaHCO3 with 1 ml of MeOH) and kept at RT for 90 min with occasional shaking. Water (2 ml) was added and extracted with CHCl3. The CHCl3 layer was washed with water, filtered, dried (Na2SO4) and the solvent evaporated. The product was identical with CIDFB obtained by synthesis.
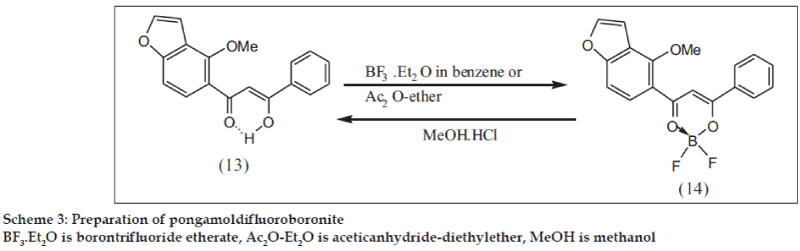
Preparation of Pongamol difluoroboronite (PDFB) (14)
(a) Pongamol [20] (13) (30 mg) was treated with acetic anhydride (1.5 ml) and ether (1 ml) and the solution kept at 0°. BF3.Et2O (0.3 ml) was added drop wise. The flask was stoppered and kept in refrigerator for 5 h. Immediately after the addition of BF3.Et2O, a deep red color appeared which slowly changed to deep yellow with separation of crystals. The crystalline product was filtered, washed with cold aq NaHCO3 and water and dried (Scheme 3). PDFB (14) (23.4 mg): m.p. 215-18°, negative Fe+++ reaction and insoluble in 5% aq. NaOH.
(b) Pongamol (10 mg) was treated with benzene (3 ml) and BF3.Et2O (0.2 ml) and kept at 60° for 3 h. Benzene was evaporated and to the residue MeOH was added drop wise when a yellow crystalline solid separated (7.6 mg), m.p. 216-218° identical with the product under (a), UV, IR (Table 1). PDFB moved slower than pongamol in TLC (viewed in ordinary light and UV chamber)
Hydrolysis of (14)
The substance (10 mg) was treated with MeOH (2 ml) and 1 drop of 6N HCl and heated under reflux at 70° for 3 h. Cold water (10 ml) was added to the reaction mixture and extracted with CHCl3. The CHCl3 layer was washed free from acid with water, dried (Na2SO4) and evaporated to get a crystalline solid (8.2 mg), identical with pongamol in all respects.
Results and Discussion
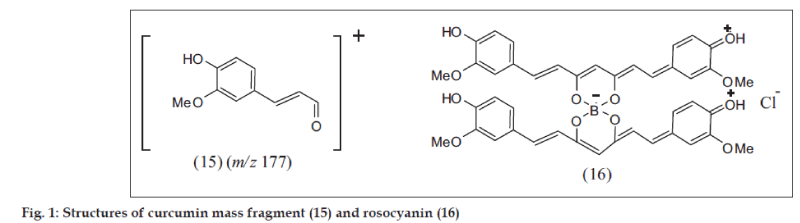
That CIDFB (6) or (6a) was an intermediate in the synthesis of CI (1) (Scheme 1), also obtained directly from CI by reaction with BF3.Et2O, is evident from its negative ferric reaction and lower mobility in TLC with the spot turning blue on exposure to ammonia vapor while CI turns orange. In the MS of CIDFB the M+ was observed at m/z 416 consistent with the formula C21H19O6BF2 with the base peak at 350 (M+-66) and the characteristic CI fragment [21] (15) at m/z 177. The MS of the diacetyl derivative (10) was similar with M+ at m/z 500, consistent with the formula C25H23O8BF2. However the prior loss of acetyl groups precedes the formation of the base peak at m/z 350 and the CI fragment at m/z 177 (fig. 1)
In accordance with the reported structures of difluoroboronites of β-diketones [17], CIDFB will be expected to have structure (6). While PDFB structure (14) conforms to this view, CIDFB does not as per the UV and IR spectral data (Table 1). In pongamol (13) the IR band at 1600 cm-1 represents both the chelated carbonyl and the –C=C– group. In the corresponding difluoroboronite (14), the –C=C– band got shifted to 1538 cm-1 with greater intensity and the chelated carbonyl appeared at 1590 cm-1. In acetylacetone (4) and its difluoroboronite (5), the shift in IR of –C=C– band is 63 cm-1 (from 1622 to 1559 cm-1) as observed by us. In CI (1) the chelated carbonyl and –C=C– bands appear at 1628 and 1603 cm-1 respectively. While the –C=C– band of CIDFB appears at 1541 cm-1, the chelated carbonyl band is absent. However in the diacetyl CIDFB (10), the –C=C– band appeared at 1544 cm-1 and the chelated carbonyl band at 1618 cm-1 (Table 1). This shows that CIDFB structure differs from that of CI and diacetyl CIDFB. This is also evident from the UV spectra.
The UV data (Table 1) shows that the λmax of PDFB (14) exhibits a bathochromic shift of 40 nm from that of pongamol (13). A similar difference (32 nm) was observed for CI (1) and diacetyl CIDFB (10), whereas the difference in λmax of CI and CIDFB is large (71 nm). This suggests a difference in the structure pattern with additional conjugation in CIDFB. This was also the case with CII (2), CIII (3) and their corresponding difluoroboronites (8) and (9) and their diacetyl difluoroboronites (11) and (12). Based on these facts, the structure for CIDFB is more in favour of the quinonoid form (6a) than (6). This is further indicated by the observation that the red color of CIDFB in MeOH with λmax 501 nm acquires a deep blue color on the addition of alkali with λmax 629 nm. CI itself shows a bathochromic shift of 56 nm on the addition of alkali. However, the shift is far greater (128 nm) on the addition of alkali to CIDFB. The deep blue colored product (in solution, pH 11.7) is tentatively assigned the structure (7). Similar was the case with CII and CIII and their corresponding difluoroboronites.
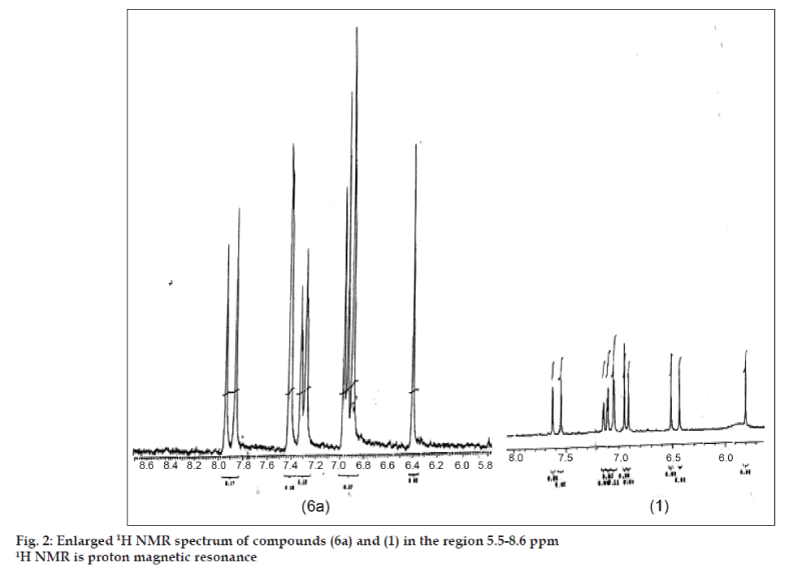
The 1H NMR spectral data of CI and its derivatives (Table 2) also seem to confirm the structure assigned to CIDFB (6a) and its diacetyl derivative (10). The data show downfield shift of all protons (other than methoxyls) in (6a) and (10). The 6,6’ and 10,10’ protons in (10) appear as a multiplet as in diacetyl CI [12]. The 1H NMR of (6a) differs from that of (1) and (10) in respect of 3,3’ and 9,9’ proton peaks. These four protons appear like three singlets accounting for 1+1+2 protons. We consider them as double doublets (dd) with the peak at 6.89 ppm integrating for two protons being common for both the doublets (fig. 2) and represent both 3’ and 9’ protons. Also the splitting of methoxyl protons in (6a) is significantly more than in (1).
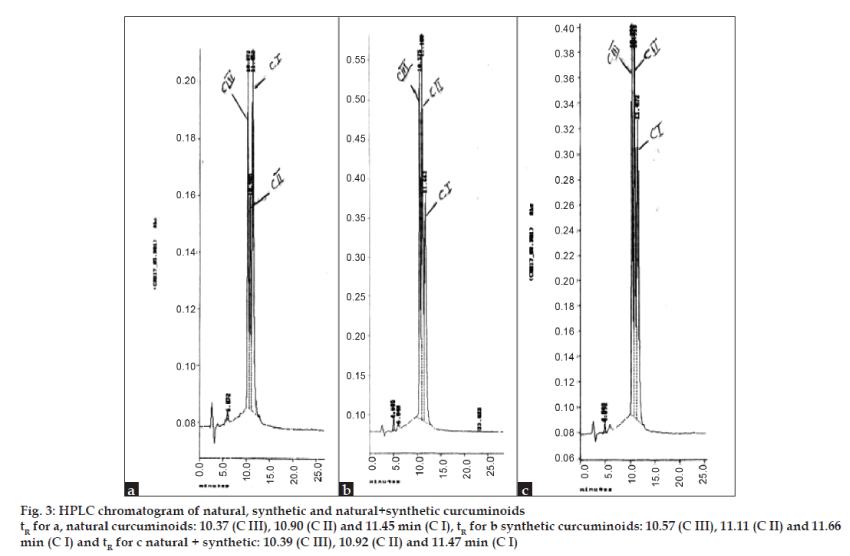
Rosocyanin (fig. 1), the compound formed when two molecules of CI (1) react with boric acid in the presence of mineral acid (concentarted H2SO4 or HCl), had been assigned the quinonoid structure [22] (16), supported by IR data [23]. This compound, isolated following a devious procedure [22], was a dark green powder with metallic luster decomposing at ca. 250° without melting.The next important stage in the synthesis is cleavage of the difluoroboronites to generate the diketones. The difluoroboronites of acetylacetone, benzoylacetone and dibenzoylmethane were reported [18] to decompose in moist air or warm alcohol liberating HF and orthoboric acid. PDFB (14) could be hydrolyzed with MeOH containing a little HCl, which hastened the reaction. CIDFB (6a) (as also CIIIDFB) is stable in acid medium and indeed it was isolated at pH 3. The red colored difluoroboronite solution in aq. MeOH showed a pH of 5.63 initially, but an hour after heating at 70° the pH changed to 3 due to release of HF with the solution turning deep red and further hydrolysis slowing down considerably. It was necessary to adjust the pH to ca. 5.8 and maintain it for complete hydrolysis. At pH 7 and above a deep blue solution is obtained resulting in intractable products. The 4/4’ free OH in the aryl groups is necessary for quinonoid formation. Dimethyl CIDFB underwent cleavage with relative ease and did not give blue colour with alkali. The synthetic curcuminoids obtained by our method are identical with the natural samples in all respects including HPLC. The HPLC chromatograms of natural, synthetic and mixed natural and synthetic curcuminoids are presented in fig. 3.
Figure 3: HPLC chromatogram of natural, synthetic and natural+synthetic curcuminoids
tR for a, natural curcuminoids: 10.37 (C III), 10.90 (C II) and 11.45 min (C I), tR for b synthetic curcuminoids: 10.57 (C III), 11.11 (C II) and 11.66
min (C I) and tR for c natural + synthetic: 10.39 (C III), 10.92 (C II) and 11.47 min (C I)
Our studies show that borontrifluoride is an effective and less costly reagent than boric oxide to protect the enol group in acetyl acetone. The use of expensive trialkylborate can be dispensed with. The intermediate difluoroboronites are stable and can be cleaved whenever required. The synthetic strategy will enable the preparation of a variety of curcuminoids in pure state and good yield for appropriate biological activity and clinical studies.
Acknowledgements
The authors thank Sri V. C. Nannapaneni, Chairman and Managing Director, Natco Pharma Ltd. for encouragement and his keen interest in this work. Our grateful thanks are due to Dr. G. Sudershan Reddy, Department of Chemistry, National Taiwan Normal University, Taipei, Taiwan (now in USA) for the NMR and Mass spectra. We also thank Sri Srikanth for experimental assistance and Ms. G. Vijayalakshmi for HPLC results.
References
- Chattopadhyay I, Biswas K, Bandyopadhyay U, Banerjee, RK. Turmeric and curcumin: Biological activity and medicinal applications. CurrSci (India) 2004;87:44-53.
- Kulkarni SK, Dhir A. An overview of curcumin in neurological disorders. Indian J Pharm Sci 2010;72:149-54.
- Satokar RR, Shah SJ, Shenoy SG. Evaluation of anti-inflammatory property of curcumin (deferuloylmethane) in patients with post-operative inflammation. Int J ClinPharmacolTherToxicol 1986;24:
- 651-4.
- Rao EV. Turmeric and curcumin. CurrSci (India) 2004;87:1324.
- Internet News: Turmeric- an effective therapy for Crohn's disease. Natural Product Radiance (CSIR) 2004;3:115.
- Rovner SL. Untangling Alzheimer’s. ChemEng News 2005;83:38-45.
- Roth GN, Chandra A, Nair MG. Novel bioactivities of Curcuma longa constituents. J Nat Prod 1998;61:542-5.
- Masuda J, Jitoe A, Isobe J, Nakatani N, Yonemori S. Anti-oxidative and anti-inflammatory curcumin-related phenolics from rhizomes of
- Curcuma domestica.Phytochemistry 1993;32:1557-60.
- Mazumdar A, Neamati N, Sunder S, Schulz J, Pertz H, Eich E, et al. Curcumin analogs with altered potencies against HIV-I integraseas probes for biochemical mechanisms of drug action. J Med Chem 1997;40:3057-63.
- Erraguntla VR, Paladugu S. An improved process for the preparation of curcumin. Indian Patent 192887; April 21, 2005 (Assignee: NatcoPharma Ltd.)
- Pabon HJ. A synthesis of curcumin and related compounds. Rec TravChim 1964;83:379-86.
- Roughley PJ, Whiting DA. Experiments in the biosynthesis of curcumin. J ChemSoc Perkin I 1973;2379-88.
- Graf E. Curcumin from vanillin and acetyl acetone. German. Patent 1,280,849; Oct. 24.1968.
- Krackov MH, Bellis HE. Process for the synthesis of curcumin related compounds. US Patent 5,679,864; Oct. 21.1997.
- Babu KV, Rajasekharan KN. A convenient synthesis of curcumin-I and its analogues. Org Prep ProcInt 1994;26:674-7.
- Bhat MV, Kulkarni SU. Cleavage of ethers (Review). Synthesis 1983:249-82.
- Lappert MF. Organic compounds of boron. Chem Rev 1956;56:961-1064.
- Morgan GT, Turnstall RB. Boron β-diketonedifluorides. J ChemSoc 1924;125:1963-7.
- Narayanaswamy S, Rangaswami S, Seshadri TR. Chemistry of pongamol part II. J ChemSoc 1954;1871-3.
- Pelter A, Ward RS, Rao EV, Raju NR. 8-Substituted flavonoids and 3’-substituted-7-oxygenated chalcones from Tephrosiapurpurea. J ChemSoc Perkin I 1981;2491-8.
- Matthes HW, Luu B, Ourisson G. Cytotoxic components of Zingiberzerumbet, Curcuma zeodariaand C. domestica. Phytochemistry1980;19:2643-50.
- Spicer GS, Strickland JD. Compounds of curcumin and boric acid, part I, the structure of rosocyanin. J ChemSoc 1952;4644-50.
- Bellamy LJ, Spicer GS, Strickland JD. Compounds of curcumin and boric acid, part III, infra-red studies of rosocyanin and allied compounds. J ChemSoc 1952;4653-6.