- *Corresponding Author:
- Saisha Vinjamuri
Bhusanayana Mukundadas Sreenivasaiah College of Engineering, Basavanagudi, Bengaluru 560019, India
E-mail: saishav.bt@bmsce.ac.in
| Date of Received | 09 May 2024 |
| Date of Revision | 29 November 2024 |
| Date of Accepted | 27 February 2025 |
| Indian J Pharm Sci 2024;87(1):44-50 |
This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms
Abstract
Exacum bicolor Roxb., is popularly documented in folklore medicine for treatment of diabetes mellitus besides other ailments such as fever, inflammation, diseases related to skin and eyes. In this present investigation, diet induced obese mice were treated with aqueous extracts of in vitro propagated Exacum bicolor plantlets at three different concentrations of 1, 10, and 100 mg/ kg body-weight of mice/day respectively. Diet induced obese mice treated with Glibenclamide 2 mg/kg body-weight of mice/ day were used as a standard. The results displayed reduced blood glucose, triglycerides, cholesterol and free fatty acids in a dose dependent manner in the diet induced obese mice treated with aqueous extracts of in vitro propagated Exacum bicolor. The results were supported by the histopathological studies of the extracts showing their ability to restore the acini architecture and beta cell number in pancreatic tissues. Further, the study was extended comparing the pharmacological properties such as alpha amylase binding assay between the in vitro and native plant extracts of the Exacum bicolor. The in vivo and in vitro studies of the current research validated and confirmed enhanced antidiabetic properties of the aqueous extracts of in vitro propagated plantlets over the methanolic extracts from native Exacum bicolor plants, corroborating the in vitro propagation and their applications in drug development.
Keywords
Alpha amylase, anti-diabetic, diet induced obese mice, Exacum bicolor, in vitro propagation
Diabetes is a metabolic disorder resulting from altered glycemic conditions acquired either genetically or through induced factors such as obesity, physical inactivity, high glycemic diet, smoking, alcohol consumption and lifestyle changes etc.,[1]. According to the World Health Organization (WHO), November 2021, about 422 million people globally have diabetes, the majority living in lower and middle-income countries. 1.5 million deaths are directly attributed to diabetes each year. It is reported that 95 % of diabetics have Type 2 Diabetes (T2D)/ diabetes mellitus[2]. Hyperglycemic conditions of T2D are highly prevalent and are mainly caused by the deficiency in the pancreatic cells to secrete insulin in adequate quantity or due to insulin resistance[3]. If untreated, this could lead to oxidative stress, inflammation and vascular complications such as nephropathy, neuropathy, cardiac diseases, retinal damage and reduced wound healing abilities. Also, a strong relationship is reported between obesity and diabetes wherein obesity leads to insulin resistance and onset of diabetes making them atopic and vulnerable to other medical complications[4-6].
Ethnobotanical literature reports the use of numerous medicinal plants and their crude extracts in the traditional treatment of diabetes[7,3,8]. Exacum bicolor (E. bicolor) Roxb., a member of the Gentianaceae family is an endemic of Western Ghats, used as a folklore medicine in treatment of diabetes, fever, haemorrhage, colitis, inflammation and skin burns[9-11]. Earlier reports on the phytochemical investigation of the plant revealed the presence of secondary metabolites viz., glycosides, alkaloids, tannins, flavonoids, saponins, triterpenes and steroids[12-15]. Exhaustive usage of this plant in traditional medicine, increased urbanization in addition to the high degree of seed dormancy has led to the development of in vitro propagation techniques. In the present study, the antidiabetic property of the aqueous extracts of in vitro generated E. bicolor plantlets was tested on Diet Induced Obese (DIO) mice validating on par with the medicinal properties of the native plant. These results were compared to correlate with histopathological studies. The effective dosage identified was further used in determining the antioxidant and alpha amylase binding assays of the in vitro and native plant extracts.
Materials and Methods
E. bicolor plant material was collected from Kaagineeri gudda in Sakaleshpura (Western Ghats-12°, 7968.70 N, 75°, 6548.10 E), India during August to September (flower/fruit development stage). The plant specimen RRCBI-4009 was authenticated and certified by National Ayurveda Dietetics Research Institute (NADRI) in Bengaluru. All the reagents used are of analytical grade and procured from Himedia, SD Fine Chemicals and SRL biochemicals. Glibenclamide used as a standard anti-diabetic drug was obtained from Sigma Aldrich (PHR1287-1G).
In vitro propagation:
The E. bicolor plantlets were developed as described by Sharad et al.[16]. Well-developed plantlets were harvested and used in this study.
Crude extract preparations:
The 3 mo-old in vitro cultured plantlets and the aerial parts of native E. bicolor plant were collected and shade dried for a week and manually powdered. Aqueous and methanolic extracts were prepared by dissolving 100 mg/ml of powdered plant material with respective solvents and placed in a shaker incubator for 72 h. Aqueous and methanolic crude extracts were soxhlet extracted, concentrated under reduced pressure and stored for comparative pharmacological studies[13].
Alpha amylase binding assay:
The aqueous and methanolic extracts of in vitro propagated and native plants were tested for their binding affinity towards alpha amylase by fluorescence spectroscopy[17]. 3 U/ml of alpha amylase solution was prepared freshly and used in this analysis. Acarbose at 1 mg/ml was used as standard. 0.5 ml of standard and the crude extracts were separately mixed with 0.5 ml of 3 U/ml of freshly prepared alpha amylase solution. This mixture was incubated for 5 min in the dark and later, the emission spectra at 282 nm were recorded from the Perkin Elmer ES55 fluorescence spectrometer.
Anti-diabetic activity of crude extracts of in vitro and native plants on DIO mice:
DIO mice C57BL/6J were considered for testing the anti-diabetic property of the aqueous extracts from E. bicolor cultured plantlets. The permission to use C57BL/6J mice as animal models was obtained from the Institutional Animal Ethics Committee (IAEC). A total of thirty six male C57BL/6J mice aged around 7 w to 9 w (20 g-25 g) were procured from Charles River United States of America (USA). All these mice were initially acclimatized for a week at temperature; 20.2 to 22.8 humidity; 46-69 %, air changes; 12-16 changes per hour, light cycle; 12 h light and 12 h dark cycle following the guidelines laid by the Committee for the Purpose of Control and Supervision of Experiments on Animals (CPCSEA).
Grouping and inducing obesity:
After a week of acclimatization, the mice were divided into 6 groups with 6 mice in each group based on body weight stratification and randomization method. To induce the obesity through diet, the mice in the five groups (30) were fed with food rich in carbohydrates and fats (43.77 %, carbohydrates: 22.23 %, proteins: 20.28 %, crude fibres: 6.52 %, ashes: 6.55 %, moisture: 0.65 % and the reverse osmosis purified water was provided ad libitum).
Study design:
Six groups of C57BL/6J mice each having six mice were taken for the study. One group (G0) was labelled as control and the remaining five groups were induced with obesity as mentioned above leading to onset of hyperglycaemic conditions. The blood and lipid parameters were monitored for all the mice initially (0th d) and after inducing obesity through diet (30th d). The obese mice were treated with glibenclamide and aqueous extracts of in vitro propagated E. bicolor plantlets at three different concentrations (1 mg/ kg, 10 mg/kg, and 100 mg/kg body weight of mice) for 4 w as follows: Group 1 (G1) was considered as DIO control with the DIO mice not receiving any treatment for their hyperglycemic conditions; group 2 (G2) the DIO mice treated with glibenclamide at 2 mg/kg of body weight (bw)/day; group 3-5 (G3- G5) DIO mice treated with plant extract at 1 mg/kg body weight (bw), 10 mg/kg bw and 100 mg/kg bw respectively. This treatment routine was continued for 28 d and the body weight and the physiological features were observed on a daily basis.
After 58 d the animals were sacrificed and histopathological studies, blood and lipid parameters/ profiles were tested and analysed.
Blood profiling:
A complete blood count which includes count of Red Blood Cells (RBC), eosinophils, lymphocytes, White Blood Cells (WBC), neutrophils, monocytes, total proteins, haemoglobin, and Thiobarbituric Acid Reactive Substance (TBARS) , lipid profile and blood glucose was carried out. The blood samples were collected by the tail nip method.
The total RBC count was estimated using the formula (Total RBC=N×dilution factor×volume correction factor) Where N=Total number of RBC cells counted in the chamber, Dilution=1: 200, Dilution factor=200. The WBC count was estimated by the formula (Total WBC count=N×50) N=Total number of WBC cells in 4 chambers i.e. 0.4 cu.mm of diluted blood. The total protein content in the serum was estimated using the Bradford method with Bovine Serum Albumin (BSA) as standard.
The total haemoglobin content was estimated using the High-Performance Liquid Chromatography (HPLC) method[18]. The total glucose content, triglycerides and cholesterol was estimated by using AGAPPE kits.
Histopathological studies:
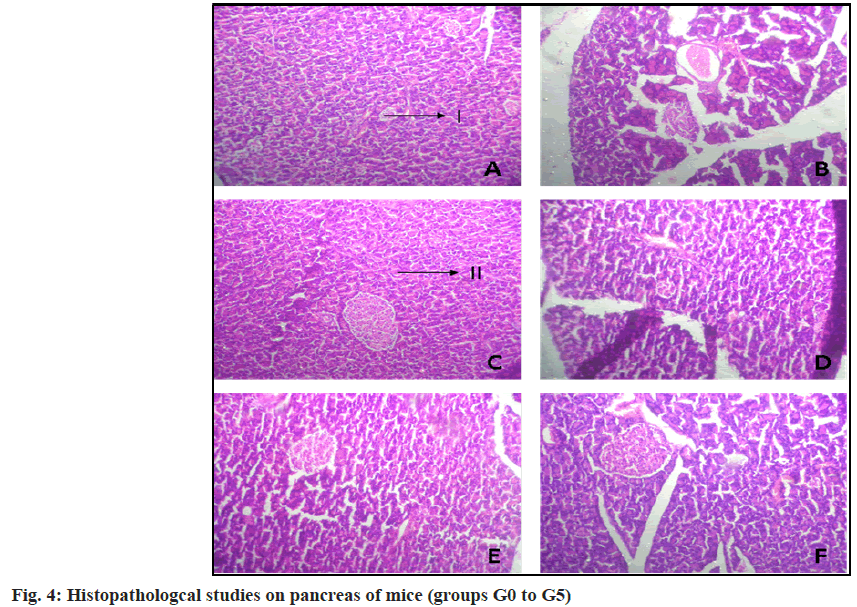
Histopathological studies were performed to study the morphological features and to observe the changes in the islets of Langerhans cells (beta cells and acinar cells) in pancreas. The mice were euthanized with a high dose of anaesthetic agent[19] and a histopathological study of the pancreas was carried out. The specimen slides were prepared using the Hematoxylin and Eosin (H&E) staining method[20]. The images were captured using an inverted microscope.
Antioxidant assay:
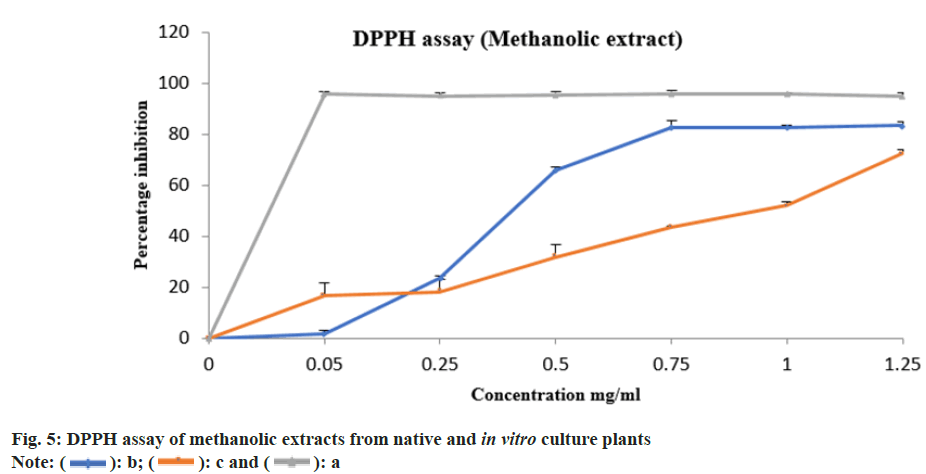
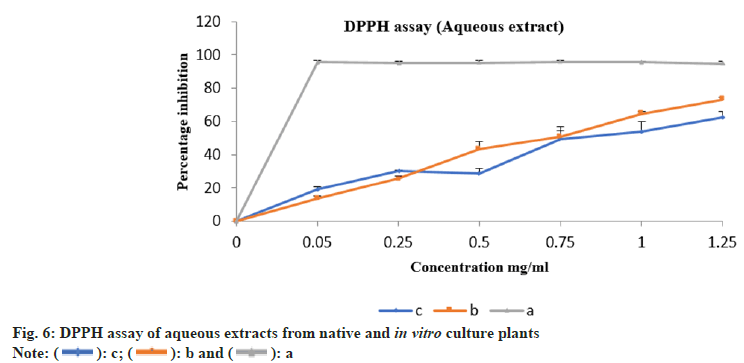
The aqueous and methanolic extracts from native and in vitro propagated E. bicolor were evaluated for their antioxidant property by 2, 2-Diphenyl-1- Picrylhydrazyl (DPPH) assay as described by Brand et al.[21]. Different concentrations of the extracts ranging from 0.05 to 1.25 mg/ml were used in the study. The values obtained from results of our study on DIO mice were used in calculating the concentration of the aqueous extract. The calculations were carried out described by Earnest et al.[22]. Ascorbic acid and gallic acid solutions were considered as standard and positive control respectively. Respective solvent alone was considered as blank whereas the solvent with DPPH was used as negative control. All the experiments were carried out in triplicate. The absorbance was read at 517 nm. (Thermo Fisher: GENESYS 10S Ultra Violet (UV-Vis) v4.003 2L9Q082005 spectrophotometer). Percentage inhibition was calculated using the formula mentioned below. Percentage inhibition=((Absorbance control- Absorbance sample)/Absorbance control)×100. Percentage inhibition against concentration were plotted and half-maximal Inhibitory Concentration (IC50) values were calculated using nonlinear regression.
Results and Discussion
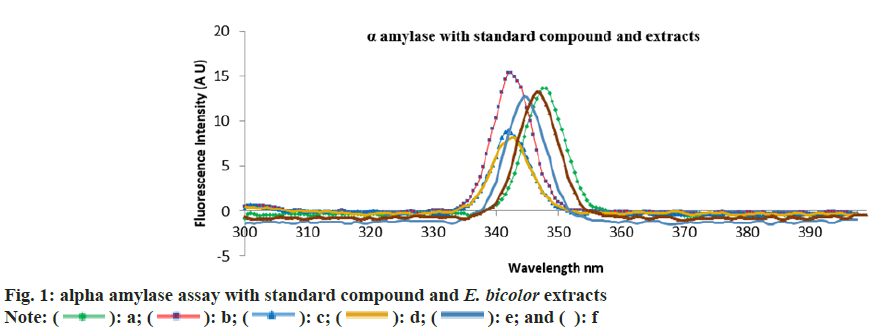
The emission maxima of alpha amylase was 342.5 nm, whereas alpha amylase with acarbose was at 347.5 nm. The emission spectra of native plant extracts with alpha amylase was 342 nm and that of in vitro plantlets was 347.5 nm (fig. 1). The results indicate that there is 1.5 % shift in absorbance in the case of in vitro plant extracts indicating increased binding affinity compared to native plant extracts.
Anti-diabetic activity of crude extracts of tissue culture and native plants on DIO mice including the C57BL/6J mice in group G1 to group G5 fed with carbohydrate rich diets for 30 d were obese and showed a significant increase in their body weight.
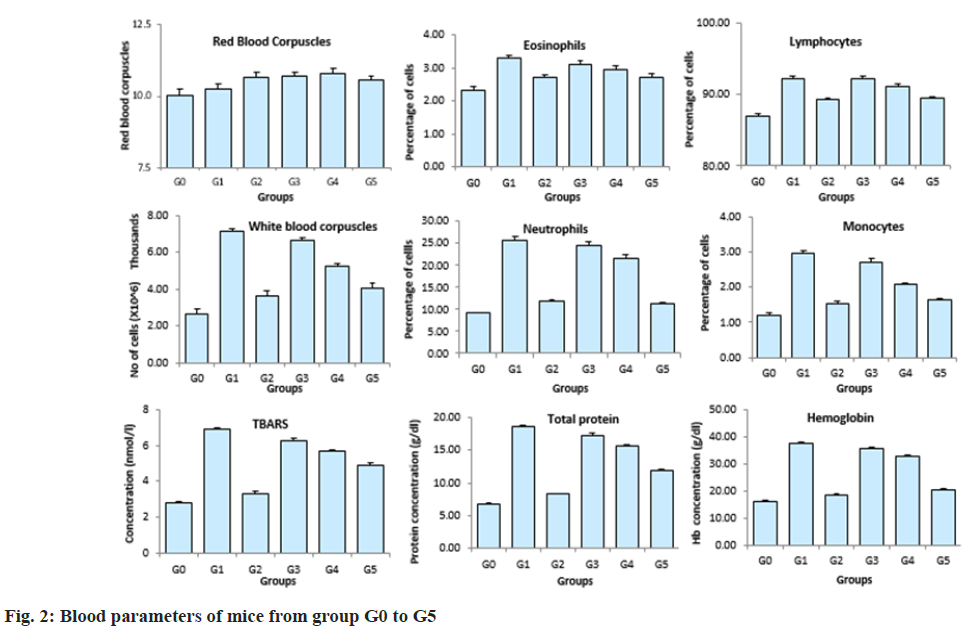
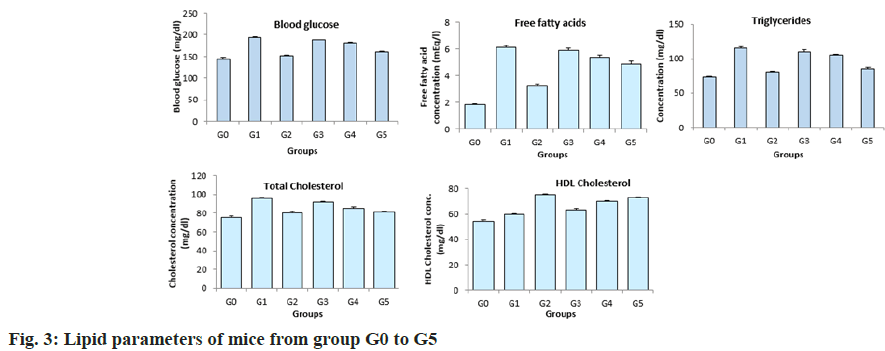
Complete blood count, lipid profile and blood glucose was carried out after 58 d (fig. 2). It was observed that the RBC count was similar in all the groups. Eosinophils and lymphocytes were significantly higher in the untreated mice compared to the mice treated with plant extracts and glibenclamide. The lymphocytes levels in mice across group G3 to G5 had no significant variations. A dose dependent reduction in the WBC levels in the mice across group 3 to group 5 was observed after the treatment period. The WBC levels were high in the DIO mice without any treatment and were significantly low in the DIO mice treated with glibenclamide. Similar results were observed with respect to neutrophil levels, monocyte levels, TBARS, total protein and hemoglobin levels. Similarly, the blood glucose and lipid profile showed a dose dependent reduction in mice treated with aqueous extract of in vitro cultured plants across the group G3, G4 and G5 respectively (fig. 3). The blood glucose levels, free fatty acids, triglycerides and cholesterol were high in the group of untreated DIO mice whereas the DIO mice treated with glibenclamide had significantly reduced levels of blood glucose, free fatty acids, triglycerides and cholesterol. A dose dependent increase in the High-Density Lipoproteins (HDL) levels were observed in mice from group 3 to 5 groups. The HDL levels were low in the DIO mice without any treatment and the DIO mice treated with glibenclamide had higher levels of HDL.
H&E of the pancreatic tissue was carried out to study the histopathological differences amongst the six groups of mice taken (fig. 4). The DIO mice from group G1 showed considerable cell damage and degenerated acini cells compared to the DIO mice treated with glibenclamide. The DIO mice treated with plant extracts (G3-G5) showed a gradual increase in the number of beta cells and improved architecture of the acini cells over the control group (G1).
The percentage inhibition of methanolic and aqueous extracts from native plants were 83.21 % and 62.34 %, whereas from the in vitro culture plantlets was 72.38 % and 73.26 % respectively. The percentage inhibition vs. concentration for methanolic and aqueous extracts are shown in fig. 5 and fig. 6 respectively. The ascorbic acid (standard) had an IC50 value of 0.41 mg/ml. The IC50 values from methanolic and aqueous extract of native plants was 0.33 mg/ml and 0.62 mg/ml, whereas from the in vitro culture plants it was 0.73 mg/ml and 0.50 mg/ml respectively.
DIO mice have been used in many studies to evaluate the therapeutic properties of various plant extracts. The methanolic extract of Salvia oficinalis was tested for its anti- diabetic properties by Kheder et al.[23]. The authors had administered the extracts to DIO mice at two doses of 100 and 400 mg/kg/ day for 5 w. They have observed improvement in glucose tolerance and decrease in insulin resistance that was similar to the effects produced by standard anti-diabetic drugs. Attakpa et al.[24] have reported using DIO mice groups to study the anti-diabetic effect of Moringa oliferia at three different concentrations (200, 400 and 600 mg/kg body weight of mice) and compared the results with glibenclamide treatment group. The authors have reported reduced blood glucose levels and an increased expression of Peroxisome Proliferator-Activated Receptor Alpha (PPARα) content (by western blotting) in DIO mice. Sunil et al.[25] have reported the anti-diabetic and anti- obesity effect of Memeceylon umbellatum using DIO mice. The authors have performed the histological examinations and analysed the serum parameters and the gene expression patterns. They have observed a significant reduction in the body weight, fasting glucose and triglyceride levels of mice treated with 250 mg/kg body weight of extracts. They have mentioned an improvement in glucose tolerance and reduced levels of serum Interleukin-6 (IL-6) and Low-Density Lipoprotein (LDL) levels along with down regulation of IL-6, Plasminogen Activator Inhibitor-1 (PAI- 1) and Apolipoprotein B (ApoB) gene from the gene expression studies. Ashwini et al.[26] has reported testing the anti-inflammatory effects of E. bicolor plant extracts on Swiss albino mice. They reported a dose dependent reduction in edema of paws. However, there have not been any reports of using DIO mice as animal models to study the anti-diabetic properties of
E. bicolor plant extracts. In this study, DIO mice were treated with aqueous extracts of E. bicolor plant extracts at three different concentrations. The main factors in the onset of diabetes viz., blood glucose, body weight, triglycerides and total cholesterol levels were reduced in a dose dependent manner in the mice treated with plant extracts. Our results correlate with the reported literature validating the anti-diabetic potential in E. bicolor plants and to the best of our knowledge is a first time report.
Upon analysing the IC50 values from the results, it was observed that the tissue culture plants had better antioxidant property than the native plants. Ashwini et al.[9] have observed a maximum inhibition percentage of 93.58 % and an IC50 value of 14.84 µg/ml from the crude methanolic extract of E. bicolor whereas Sharad et al.[27] have reported the antioxidant property from the methanol and hexane extracts of E. bicolor to be modest. In our present study aqueous extracts of E. bicolor was used in this study, it was safe and non-toxic on the model organisms. The effective dosage was identified and used to validate the antioxidant property of the plant extracts of native and tissue engineered plantlets. Sharad et al.[27] have investigated the Absorption, Distribution, Metabolism, and Excretion (ADME) properties of 32 phytochemicals using SwissADME and have identified two molecules i.e. phthalic acid butyl dodecyl ester and pyrimidine-2,4,6 (1H,3H,5H)-trione-5-(3-(2-(4- tertbutylphenoxy) ethoxy) benzylidene as possible drug candidates. The authors have performed antimicrobial assays using crude extracts of E. bicolor and have reported prominent antibacterial property against Bacillus pumilus, Bacillus subtilis, Staphylococcus aureus, Escherichia coli and moderate antifungal property towards Aspergillus niger and Aspergillus fumigatus, they have also reported moderate cytotoxic activity in reducing the cell viability of Michigan Cancer Foundation-7 (MCF-7) breast cancer cell lines.
Further a preliminary screening on the binding affinity of the plant extracts to alpha amylase was also performed. The present research investigation represents the first reports on antioxidant and amylase assays on the tissue cultured plantlets of E. bicolor and compared pharmacologically with the native plant extracts.
The plant extracts of E. bicolor have scientifically proven to contain anti-diabetic properties, hence there is a need to conserve and utilize its medicinal properties to the fullest. The current research has led to the development of a rapid and efficient in vitro propagation technique for the endemic medicinal plant E. bicolor Roxb. Testing of the in vitro generated plantlet extracts on model organisms (DIO mice) has shown positive results indicating the presence of anti- diabetic and antioxidant properties. In vitro generated plantlet extracts showed better activity when compared to native plant Exacum crude extracts. The present study not only bioengineered the plant through in vitro propagation, but also gave insights to the bioactive metabolites that can be served as lead molecules for treating type II diabetes.
Acknowledgments:
The authors were thankful to Skanda Life Sciences Pvt. Ltd. Bengaluru for helping with the animal models and histopathological studies. The authors also thank Technical Education Quality Improvement Programme (TEQIP-2 and TEQIP-3) for providing the financial assistance during this research work.
Conflict of interests:
The authors declared no conflict of interests.
References
- Lee J, Noh S, Lim S, Kim B. Plant extracts for type 2 diabetes: From traditional medicine to modern drug discovery. Antioxidants (Basel) 2021;10(1):81-123.
[Crossref] [Google Scholar] [PubMed]
- Islam MS, Wilson RD. Experimentally induced rodent models of type 2 diabetes. Methods Mol Biol 2012;933:161-74.
[Crossref] [Google Scholar] [PubMed]
- Govindappa MA. Review on role of plant(s) extracts and its phytochemicals for the management of diabetes. J Diabetes Metab2015;6(7):1-38.
- Wondmkun YT. Obesity, insulin resistance, and type 2 diabetes: Associations and therapeutic implications. Diabetes Metab Syndr Obes 2020;13:3611-6.
[Crossref] [Google Scholar] [PubMed]
- Siddiqui S. Obesity and diabetes: Interrelationship. Adv Obes Weight Manag Control 2018;8(2):155-8.
[Crossref]
- Pilar S. Obesity and diabetes. Nutr Hosp 2019;28(Suppl_5):138-43.
- Singh M, Nayak P, Mishra V. Herbal approaches for the treatment of diabetes mellitus. Plant Arch 2020;20(Suppl_2):3221-8.
- Kooti W, Farokhipour M, Asadzadeh Z, Ashtary-Larky D, Asadi-Samani M. The role of medicinal plants in the treatment of diabetes: A systematic review. Electron Phys 2016;8(1):1832-42.
[Crossref] [Google Scholar] [PubMed]
- Ashwini AM, Ramakrishnaiah H, Manohar SH, Majumdar M. An efficient multiple shoot induction and genetic fidelity assessment of Exacum bicolor roxb., an endemic and endangered medicinal plant. In Vitro Cell Dev Biol Plant 2015;51:659-68.
- Sreelatha U, Baburaj TS, Kutty CN, Nazeem PA, Bhaskar J. Cultivation prospects of Exacum bicolor roxb. an endangered, ornamental and anti-diabetic herb. Indian J Nat Prod Res 2007;6(5):402-4.
- Vinayaka KS, Ashwini HS, Kekuda TRP, Krishanamurthy YL. Traditional utilization and phytochemical analysis of medicinal herb Exacum Linn. from central Western Ghats. Asia Pac J Health Sci 2016;3(1):161-4.
- Jeeshna MV, Paulsamy S. Phenology of Exaum bicolor roxb., an endangered medicinal herb of Kannur and Wayanad districts, Kerala, India.Maejo Int J Sci Technol 2011;5(3):358-64.
- Jeeshna MV, Paulsamy S. Evaluation of certain flavonoids of medicinal importance in the wild and micropropagated plants of the endangered medicinal species, Exacum bicolorroxb. J Appl Pharm Sci 2011;1(5):99-102.
- Appaji MA, Puttarudrappa L, Ravi BV, Majumdar M. GC-MS analysis, evaluation of phytochemicals, antioxidant, thrombolytic and anti-inflammatory activities of Exacum bicolor. Bangladesh J Pharmacol 2015;10(4):745-52.
- Vinjamuri S, Sharad S. Comparison of phytochemical components in leaves and stems of Exacum bicolorRoxb. by GCMS. World J Pharm Pharm Sci 2017;6(7):2134-8.
- Sharad SA, Saisha V. A simple and efficient micropropagation protocol for developing plantlets of Exacum bicolorroxb.-An endangered, ornamental and antidiabetic herb. Asian J Pharm Clin Res2021;14(5):36-40.
- Li Y, Gao F, Gao F, Shan F, Bian J, Zhao C. Study on the interaction between 3 flavonoid compounds and alpha-amylase by fluorescence spectroscopy and enzymatic kinetics. J Food Sci 2009;74(3):199-203.
[Crossref] [Google Scholar] [PubMed]
- Han BG, Hao CM, Tchekneva EE, Wang YY, Lee CA, Ebrahim B, et al. Markers of glycemic control in the mouse: comparisons of 6-h- and overnight-fasted blood glucoses to Hb A1c. Am J Physiol Endocrinol Metab 2008;295(4):981-6.
[Crossref] [Google Scholar] [PubMed]
- Cicero L, Fazzotta S, Palumbo VD, Cassata G, Lo Monte AI. Anesthesia protocols in laboratory animals used for scientific purposes. Acta Biomed 2018;89(3):337-42.
[Crossref] [Google Scholar] [PubMed]
- Campbell-Thompson ML, Heiple T, Montgomery E, Zhang L, Schneider L. Staining protocols for human pancreatic islets. J Vis Exp 2012;63:4068-72.
[Crossref] [Google Scholar] [PubMed]
- Brand-Williams W, Cuvelier ME, Berset C. Use of a free radical method to evaluate antioxidant activity. LWT-Food SCI Technol 1995;28(1):25-30.
- Earnest E, Ajaghaku D. Guidelines on dosage calculation and stock solution preparation in experimental animals' studies. J Nat Sci Res 2014;4(18):100-6.
- Khedher MR, Hammami M, Arch JR, Hislop DC, Eze D, Wargent ET, et al. Preventive effects of Salvia officinalis leaf extract on insulin resistance and inflammation in a model of high fat diet-induced obesity in mice that responds to rosiglitazone. Peer J 2018;6:4166-88.
[Crossref] [Google Scholar] [PubMed]
- Attakpa ES, Sangaré MM, Béhanzin GJ, Ategbo JM, Seri B, Khan NA. Moringa olifeira Lam. stimulates activation of the insulin-dependent Akt pathway antidiabetic effect in a Diet-Induced Obesity (DIO) mouse model. Folia Biol (Praha) 2017;63(2):42-51.
[Crossref] [Google Scholar] [PubMed]
- Sunila V, Nitya S, Venkatarangannac MV, Ramesh RB, Mala M. The anti-diabetic and anti-obesity effect of Memecylon umbellatum extract in high fat diet induced obese mice.Biomed Pharmacother 2017;89:880-6.
[Crossref] [Google Scholar] [PubMed]
- Ashwini M, Gowda SV, Mala M. In vivo anti-inflammatory activity of Exacum bicolorRoxb. leaves. Indian J Pharm Sci 2018;80(6):1155-9.
- Sharad SA, Lekshmi P, Akbar AP, Narasimha RP, Saisha V. ADME analysis of crude extracts of Exacum bicolor roxb. and in vitro screening for antioxidant, antimicrobial and cytotoxicity assays. Asian J Chem 2022;34(10):2633-8.
[Crossref]