- *Corresponding Author:
- G. Mustafa
Department of Biochemistry, Government College University, Faisalabad-38000, Pakistan
E-mail: gmustafa_uaf@yahoo.com
| Date of Submission | 24 January 2017 |
| Date of Revision | 13 May 2017 |
| Date of Acceptance | 02 May 2018 |
| Indian J Pharm Sci 2018;80(4):581-591 |
This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms
Abstract
The pathological emblems of Alzheimer’s disease are the accumulation of amyloid-β plaques and neurofibrillary tangles. The alluvium of toxic amyloid-β-protein in the form of aggregates is central to the pathogenesis of Alzheimer’s disease. The aggregate formation is due to the structural refitting of α-helical sheet of normal, soluble amyloid-β-protein to the β-sheets, which lead to oligomeric, fibrillar, insoluble and disease causing amyloid-β 42. Mounting data suggests that another factor, the tau protein ripens into highly phosphorylated form by several kinases after Aβ-stimulation leads to tangle formation resulting in neuronal bereavement in hippocampus and entorhinal regions as the disease progresses further. An overview has been presented in this review of the role of tau as an important partner of amyloid-β in the pathogenesis of Alzheimer’s disease, both of which could be used as biomarkers for diagnosis and risk assessment with other molecular chaperones which are associated with Alzheimer’s disease. As a part of common pathophysiological mechanism the understanding of amyloid-β and tau toxicities might be helpful for finding molecular targets for the prevention or even cure of Alzheimer’s disease.
Keywords
Alzheimer’s disease, neurofibrillary tangles, amyloid-β-protein, tau protein
Alzheimer’s disease (AD), a neurodegenerative disorder, is the most common form of age-related dementia, first described by a neurologist Alois Alzheimer in 1906. The disease is caused by progressive large scale death of neurons in the brain region, particularly neocortex and hippocampus [1], preceded by a silent preclinical phase that spans over decades [2]. Loss of synaptic function due to neuronal death within hippocampus and entorhinal cortex, development of neuritic plaques and neurofibrillary tangles (NFTs) [3,4], which follow the preclinical phase, constitute the major symptoms of AD.
The main pathological changes that have been observed in the brain tissues of Alzheimer’s patients are amyloid-β (Aβ) peptide and hyperphosphorylated tau (p-tau) protein. The exact mechanisms, which lead to these changes, are still to be determined. Aβ is the major element of neuritic/amyloid plaques and is approximately a 4 kDa polypeptide, formed because of inappropriate cleavage of amyloid precursor protein (APP). NFTs are formed due to abnormal p-tau protein [4,5]. Tau pathology is followed by APP dysfunction within hippocampus and entorhinal cortex. An important risk factor for AD is the age and AD is categorized in to two groups depending on the age of onset. Ninety percent of patients of age above 65 y with disease symptoms are said to be suffering from late inception AD (LOAD) and 10 % patients who are of age below 65 y are said to be suffering from early onset AD [6]. In Pakistan, the proportion of the AD is increasing as a result of changing population demographics [7]. This type of neurodegenerative disorders is characterized by neuronal death. The process of neuronal death may be physiological or pathological. All these mechanisms involve neurofibrillary degeneration, which is categorized by the abnormal aggregation of intraneurons and such changes result in neurodegenerative disorders [8]. Neurodegenerative disorders e.g. AD are the cause for a momentous and increasing proportion of morbidity and fatality. As a result of increased life span and varying population demographics, neurodegenerative disorders and dementias have become more common [9]. By understanding the concept of pathogenesis of a disease, it may be possible to identify a path for developing therapeutic approaches with wide applicability for disease prevention, thus decreasing morbidity and fatality in the elderly population [10,11]. Some of the neurodegenerative disorders with their characteristic symptoms have been summarized in the Table 1.
| Disease | Pathological Feature | Location | Primary Symptoms |
|---|---|---|---|
| Alzheimer’s disease | β-amyloid plaques NFTs |
Hippocampus and Cortex | Memory loss |
| Parkinson’s disease | Loss of dopaminergic neurons | Substantia nigra and striatum | Motor control problems |
| Huntington’s disease | Cell-death accompanying with movement and cognitive dysfunction | Neostriatum and cortex | Severely reduced life span |
NFTs: neurofibrillary tangles
Table 1: Neurodegenerative disease and their characteristic features and symptoms
Alzheimer’s Disease (AD)
A great proportion of the population is facing agerelated dementia. The individuals are suffering from the loss of mental abilities with their increasing age. While in severe cases, incapacitation and death occurs due to the progressive global loss of normal activities and perceptual abilities. AD is caused by progressive death of neurons on large scale in the brain region particularly neocortex [1]. The clinical symptoms of AD are preceded by a silent preclinical phase, which is characterized by neuropathological change spanning over decades [2].
The most common symptom pattern is gradual difficulty in remembering new information. As the disease progresses, patients also experience confusion, disorganized thinking, impaired judgment, trouble in expressing and disorientation to time, space and location. All these problems could result in unsafe wandering and socially inappropriate behaviour. Patients with advanced AD require help in daily activities of life such as bathing, dressing and eating. In the final stage of the disease patients lose the ability to communicate and even recognize their near and dear ones. Thus, they become confined to their beds and ultimately the disease becomes disastrous [11]. The changes, which occur in the final stage of AD include, dramatic reduction in the width of neocortex [12], senile plaques (SPs) become surrounded by the cellular debris from degenerated neurons and axons, which are then called neuritic plaques [3] and aggregation of fibrils to form NFTs within the neurons of hippocampus [4].
All types of AD possess same pathology [13]. Two hypotheses proposed for AD occurrence [14] were the amyloid cascade hypothesis, according to which the neurodegenerative process in AD is a series of events triggered by abnormal processing of the APP and the neuronal cytoskeleton degeneration hypothesis, which states that abnormal changes in the cytoskeleton stimulate the neurodegenerative process in AD.
Familial AD
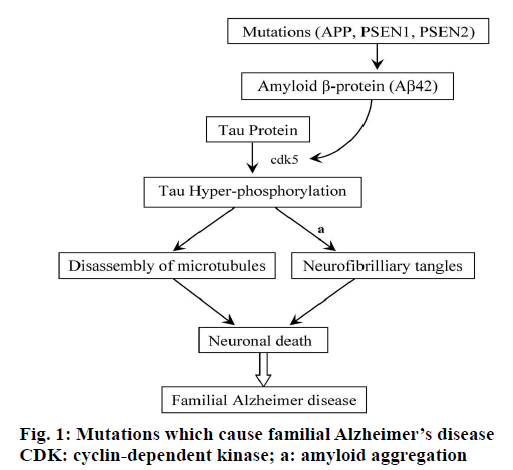
Early onset familial AD is a result of missense mutations. These mutations are caused by changes in amino acid at site 717 and 716 of APP. The activity of either β- or γ-secretase is altered due to mutation, which results in the increase in toxic Aβ42. In addition to APP mutations, the other two genes whose mutations are responsible for early onset of AD are located on the chromosome 1 and 14.
Genes are designated as presenlin-1 (PSEN-1) and presenlin-2 (PSEN-2), respectively. The chromosomal location of PSEN-1 is 14q24.3 and for PSEN-2 is 1q32-q42 [15]. These genes encode transmembrane proteins PSEN-1, which has 467 amino acid residues and PSEN-2, which has 448 amino acid residues. These proteins are synthesized in neurons. Mutations in these genes are responsible for excessive production of Aβ42 that leads to the AD. These PSEN-1 mutations account for 40 % of early onset of familial diseases where clinical signs of the symptoms appear during the age 30 to 55 y. PSEN-2 mutations account for less than 1 % of familial AD in individuals with age of 40 to 75 [16,17].
Familial AD can also be caused by a third gene, the apolipoprotein E (ApoE). This gene is present on chromosome 19 and exists in polymorphic forms. The three common alleles make six combinations, ∈2/∈2, ∈3/∈3, ∈4/∈4, ∈2/∈3, ∈2/∈4, and ∈3/∈4. Each allele encodes distinctive protein. The protein isoforms formed in brain are ApoE2, ApoE3 and ApoE4 [18]. ApoE4 in combination of environmental factors accounts for 50 % of familial AD, thus enhancing the chances of disease. These mutations lead to overproduction of Aβ42 and p-tau protein [19]. A summary of the mutations which cause familial AD was presented in Figure 1.
Biochemistry of SPs
SPs were first demonstrated by Blocq and Marnesco in 1892 [20]. A closely packed fibrous structure is made up by core of SPs is called as amyloid body. The production of amyloid bodies, associated with AD pathogenesis is called amyloidosis. These amyloid bodies contain fibrillogenic protein (amyloid fibrils that are smooth, straight and about 10 nm in diameter) and nonfibril components (ApoE and serum Aβ component). The SPs, also called neuritic plaques or amyloid plaques, are extracellular accumulation of Aβ protein [21].
Biochemistry of NFTs
The NFTs were first described in 1907 by Alzheimer [20]. NFTs are large and non-membrane bound bundles of abnormal fibres [22]. These are mainly composed of abnormal p-tau protein [5,7] within certain neuronal population. Tau pathology is followed by APP dysfunction and observed in hippocampus and entorhinal cortex [8].
Aβ PROTEIN
This major component of amyloid plaques in AD is ∼4 kDa polypeptide called as Aβ-protein, which is normally secreted and soluble product. Aβ protein exists in different isoforms ranging from amino acid number 39 to 43 [23]. The two commonly occurring isoforms are Aβ1-40 (Aβ40) and Aβ1-42 (Aβ42). Aβ40 has 40 amino acids and is soluble isoform while Aβ42 has 42 amino acids and is insoluble isoform. Comparison of both isoforms that are secreted by cells as well as isolated from AD brain indicates that Aβ40 (>60-70 %) are in higher levels than Aβ42 (~15 %) in normal individual. But the Aβ42 levels become elevated in AD [24]. Abnormal processing of APP leads to excessive insoluble Aβ42 isoform. This accumulates in the form of aggregates, which contain amyloid protein in the form of oligomers, protofibrils or fibrils. Findings of researchers depict that it is due to the conversion of Aβ- monomers to the Aβ-oligomers before aggregation [25] and these oligomers become toxic to neurons [26]. So, elevated levels of insoluble and potentially oligomeric Aβ42 are responsible for synaptic destruction during early phases of AD [27].
Formation of Aβ-peptides
Amyloid β-precursor protein (AβPP) derives Aβ- peptides [23,28] that is an integral type I glycoprotein [29] with large ectodomain, a single membrane spanning domain and short cytosolic domain. The chromosome location for AβPP is 21q21.2. The APP gene has 18 exons that span more than 170 kb, which creates 10 isoforms by alternative splicing. These isoforms range from 563 to 770 amino acid residues. The 695 amino acid APP isoform has been observed in neurons of central nervous system [30].
The region that encodes Aβ-sequence consists of exons 16 and 17, and contains 40 and 43 amino acid residues. This region is extended from the ectodomain into transmembrane domain of the protein [28]. The functional domain of APP has ability of characteristic binding to metal ions copper (Cu), zinc (Zn), heparin, collagen, respectively. The heparin and collagen binding mediate the interaction of APP with extracellular matrix.
A domain that is involved in neuritic outgrowth delineates to a short sequence present after the insertion of exon 7 product [21]. The vital roles of APP isoform are given below [31]. Cell membrane bound 695 amino acid APP isoform may have role in facilitating coordination between cells, connection to the extra neuronal matrix and synaptic stability; intracellular APP might be linked with the cytoskeletal system that transports components within a cell; the other two types are secreted APP fragments, APP1-671 (βAPPs) and APP1-687 (αAPPs) both have role in protection of neurons from damage and modulate events at synapse.
Proteolytic processing of APP
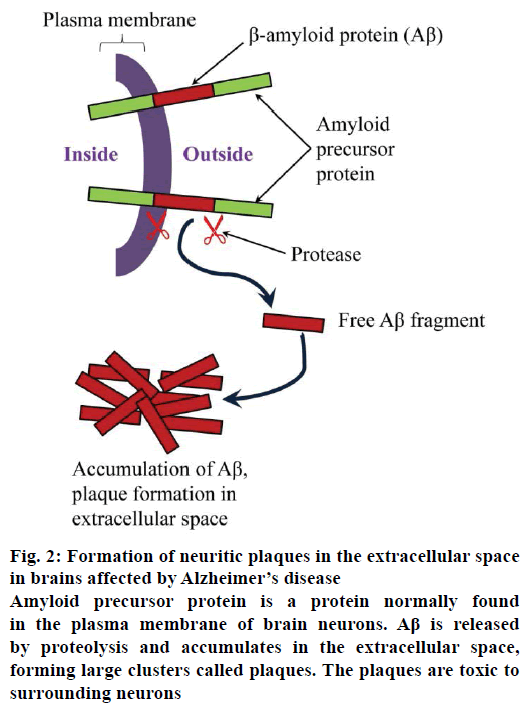
Proteolytic processing events occurring near and within APP transmembrane domain provide best demonstration for the formation of toxic Aβ42 and ultimately for the pathogenesis of AD. Three alternative cleavages shed ectodomain of APP form the membrane. These cleavages are α-secretase cleavage (it splits Aβ-domain and prevents Aβ-formation by producing non-amyloidogenic product because it is incapable of forming pathogenic Aβ [32]), β-secretase cleavage (the amino terminal of the Aβ-peptide is exposed by this cleavage [33]) and γ-secretase cleavage (the resulting fragment formed from α-secretase and β-secretase cleavage remains associated in the membrane and upon further processing by γ-secretase it gets degraded). The third cleavage creates Aβ-protein (Aβ42) whose accumulation causes AD because of its toxic and fibril aggregates [23,24,28]. The mechanism of the formation of these toxic Aβ aggregates is represented in Figure 2.
Figure 2: Formation of neuritic plaques in the extracellular space
in brains affected by Alzheimer’s disease
Amyloid precursor protein is a protein normally found
in the plasma membrane of brain neurons. Aβ is released
by proteolysis and accumulates in the extracellular space,
forming large clusters called plaques. The plaques are toxic to
surrounding neurons
Tau Protein
Tau protein is the member of microtubule-associated proteins family and found in cytosol and in axons in neurons. This is a cell membrane-associated protein, which exists in many animal species including Caenorhabditis elegans, Drosophila, goldfish, bullfrog, rodents, bovines, goat, monkeys, and human [34]. In human tau protein is present in neurons but is in trace amounts in non-neuronal cells [35]. It has been suggested that tau proteins might be synthesized in glial cells, mostly in pathological situations [36] and it is possible to identify tau mRNA and proteins in numerous peripheral tissues such as heart, kidney, lung, muscle, pancreas as well as in fibroblasts [37].
Gene organization and isoforms
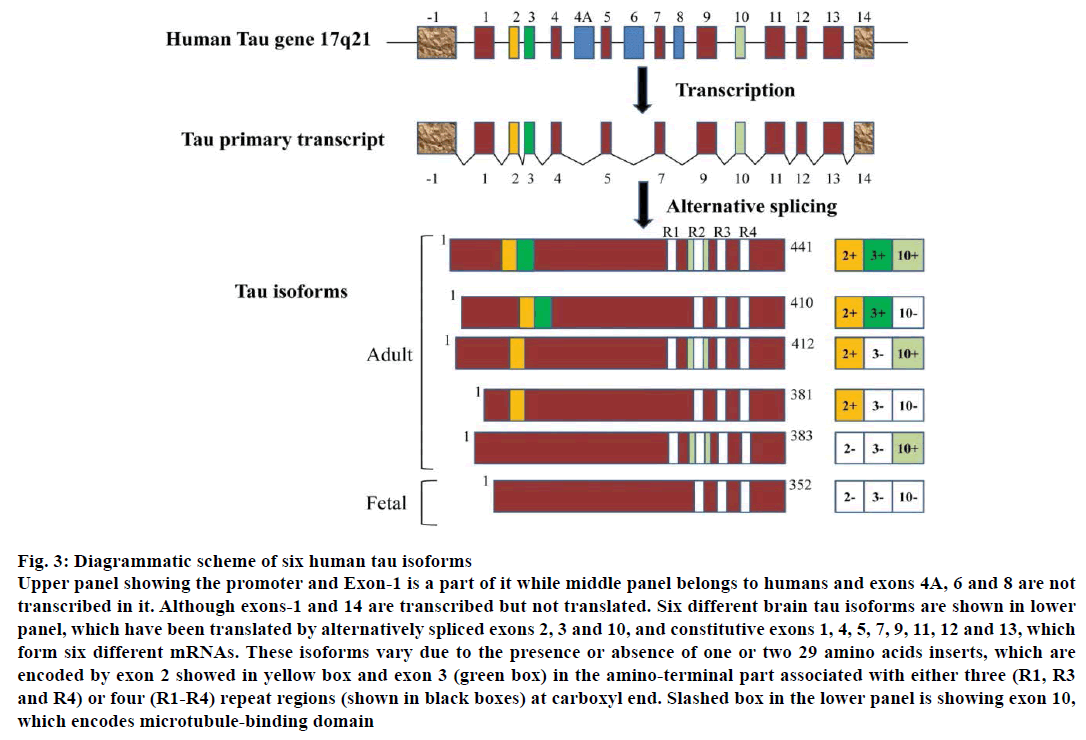
The human tau gene is distinctive and found over 100 kb on the long arm of chromosome 17 at locus position 17q21 and has 16 exons. The restriction analysis and sequencing of the gene reveals that it comprises two CpG islands, one related to the promoter region and other with exon 9 [38]. A TATA-less sequence was also revealed by the sequence of the promoter, which is related to the presence of multiple initiation sites. At the carboxyl terminal region of tau-protein, a microtubule binding domain is present. This binds to other proteins such as protein phosphatase 2A (PP2A). The N-terminal of tau-protein binds to actin proteins. The primary transcript of tau has 16 exons. Exons 2, 3 and 10 are alternatively spliced and are specific for adult brain. In the human brain the tau primary transcript gives rise to six mRNAs shown in Figure 3.
Figure 3: Diagrammatic scheme of six human tau isoforms
Upper panel showing the promoter and Exon-1 is a part of it while middle panel belongs to humans and exons 4A, 6 and 8 are not
transcribed in it. Although exons-1 and 14 are transcribed but not translated. Six different brain tau isoforms are shown in lower
panel, which have been translated by alternatively spliced exons 2, 3 and 10, and constitutive exons 1, 4, 5, 7, 9, 11, 12 and 13, which
form six different mRNAs. These isoforms vary due to the presence or absence of one or two 29 amino acids inserts, which are
encoded by exon 2 showed in yellow box and exon 3 (green box) in the amino-terminal part associated with either three (R1, R3
and R4) or four (R1-R4) repeat regions (shown in black boxes) at carboxyl end. Slashed box in the lower panel is showing exon 10,
which encodes microtubule-binding domain
All the six isoforms differ from each other by the presence of four repeats of 31 or 33 amino acids at the C-terminus. The presence of none or one or two inserts in the N-terminal region has been reported [39] and molecular weights of these inserts were found in the range of 45 to 65 kDa. These isoforms range in length from 352-441 amino acids in central nervous system. The adult tau protein isoforms are of 441, 412, 410, 383, 381 amino acids and fetal tau-protein isoform is of 352 amino acids [40].
Tau protein functions in AD pathogenesis
Cytoskeleton consists of microtubules and maintains morphology and structural integrity. Microtubuleassociated proteins maintain the assembly and stability of microtubules by promoting their polymerization and bind with tubulin, thus suppressing their dissociation and all this happens when tau is in dephosphorylated form and its dephosphorylation occurs at serine and threonine site [5]. Tau has at least 30 phosphorylation sites with Ser-Pro and Thr-Pro motives [8]. Tauprotein hyper-phosphorylation enhances its ability to self-assemble into tangles of paired helical filament (PHF) and this hyperphosphorylation occurs at the C-terminus. These tangles become accumulated in the form of aggregates that results in the loss of axonal or dendritic transport as well as disassembly of the microtubules in the disease [5,39].
Proteomics of Aβ and Tau Protein
Advances in the field of proteomic analysis have significant implications for understanding the huge number of pathways that manage behaviour and cognition and nervous system response to disease or injury [41]. The analysis of Aβ and tau protein from the patients of AD helps to sought out the model and formation of toxic Aβ protein and p-tau [42].
Aβ protein in AD
Amyloid fibrils in AD are insoluble and orderly aggregates of toxic Aβ proteins. These fibrils have diameter from 70-100 A° and are consisted of different protofilaments. Each protofilament contains an organization of β-sheet in polypeptide chains that are vertical to the extended axis of the fibre while protofibrils contain semifibrillar aggregates, which are found in early phases of the disease [43].
The demonstration of organization of the fibril has been aided by the structural studies of ex vivo amyloid fibrils taken from the brain of an AD patient but due to high insolubility of the plagues the data of high resolution has been limited. The synthetic fragments that correspond to Aβ peptide regions have been shown to generate amyloid fibrils in vitro. Electron microscopy showed indistinguishable fibrils from tissue amyloid fibrils with similar characteristics to in vivo amyloid fibrils e.g. when stained with Congo red they showed the same green birefringence under cross-polarized light [43].
Amyloid-forming peptide
The structural integrity of the fibre is maintained by the key sequence, residues 11-25 of Aβ. The peptide corresponding to this key sequence was carefully formed by Serpell et al. [43] on the basis of similar properties to full-length Aβ. This was resulted in vitro formation of amyloid fibrils, which resembled to full-length Aβ (1-40) and Aβ (1-42) as well as ex vivo amyloid. Highly oriented diffraction patterns have been shown by these well-ordered fibrils. These peptide regions have been considered to be involved in the confirmation switching from α- to β- structures particularly 13 and 14 residues of histidine and 11 and 22 of glutamate. The electrostatic character of these residues has been hypothesized for directing the formation of salt-bridge and facilitation of β-sheet folding and fibrillogenesis [44]. The replacement of single amino acid such as residues 16, 18, 19 or 20 leads to a decreased tendency for the formation of a fibril. These residues are considered to be very important for the formation of amyloid fibril. Glu22Gln called the Dutch-type mutation, has been recognized to increase the formation of fibril and peptide stability [45]. A hydrophobic cluster of residues is found in the peptide at 17-21 and it has been proposed that the cluster is involved in the interaction of side-chain. The shortest peptide, which is of 14-23 residues has been reported for its tendency for making fibrils correspond to the core of structural model. The residues from 16-20 i.e. KLVFF is a peptide sequence, which inhibits the formation of an amyloid fibril therefore, these residues are important in the assembly process [46].
Levels of Aβ-protein
Inappropriate cleavage of APP leads to toxic Aβ formation due to the accumulation and aggregation of APP fragments. A membrane bound peptide fragment released from APP by β-secretase cleavage is known as C99 and this is a smaller aggregation-prone peptide. Its formation is described as rate-limiting step in the production of Aβ-peptide. This has been proved by using the C99 yeast expression system that this fragment is formed due to mutation in APP or the defective proteasomal function [47].
In vivo gene expression studies suggest that Aβ accumulation leads to the increased microtubuleassociated protein tau mRNA, which then results an increased level of tau protein in cerebrospinal fluid. The levels of both Aβ40 and Aβ42 are higher in brain regions, which include hippocampus, olfactory bulbs and cortex than in the cerebellum, septum and striatum. As the disease progresses, Aβ40 accumulate into plaques and this recruitment leads to decreased Aβ42/Aβ40 ratio. But Aβ40 insoluble to soluble ratio increases with the passage of time while Aβ42 insoluble to soluble ratio decreases [48].
Tau in phosphorylation in AD
The important modification of tau protein in AD is its phosphorylation. A triplet of proteins is revealed by biochemical characterization using immunoblotting, which consists of tau55, tau64 and tau69 (tau isoforms) and it is called PHF-tau [49]. Development of the typical AD-triplet along with tau74 is due to the involvement of tau isoforms where tau74 is a tau-variant. Tau74 leads to abnormally increased phosphorylation of tau and this leads to decreased eletrophoretic mobility of the protein, which ultimately results NFTs formation [50].
Kinases and sites for tau phosphorylation
Most of the kinases responsible for tau phosphorylation are part of the proline-directed protein kinases, which includes mitogen-activated protein kinase, glycogen synthase kinase 3 (GSK3), tau-tubulin kinase and cyclin-dependent kinases including cyclin-dependent kinase 5 (CDK 1/CDC 2) and CDK 5. Furthermore, stress-activated protein kinases (SAP kinases) have been recently implicated in tau phosphorylation [51]. A number of different protein kinases can phosphorylate non-Ser/Thr-pro sites, which include cyclic-AMPdependent kinase [52], Ca+2/calmodulin-dependent protein kinase II [53], microtubule-affinity regulating kinase and casein kinase II [54].
Tau protein as biological marker
In AD patients, the accumulated tau proteins are the principal elements of NFT and a major indication for immunohistochemical or biochemical revealing in the central nervous system because it is associated with the complexity of dementia [55]. The presence of these tau proteins therefore, has been examined in biological fluids and peripheral tissues in order to identify an ideal AD marker to use it as an ideal diagnostic tool. These biomarkers should ideally possess the ability to help diagnose the disease in its early asymptomatic phase and therefore, increase the success of a hypothetical preventive treatment [56].
Molecular Chaperones
Transcription and translation processes are required for expression of polypeptide gene products that generate mRNA and synthesize polypeptide chains, respectively. It is known that some important amino acid residues at the right positions are necessary for protein folding into a 3D functional and exclusive conformation. It is amazing that out of millions of conformational forms only one form is associated with a functional protein. In some instances the process of directing and targeting intermediate polypeptide folding to a fully folded confirmation is aided by proteins known as molecular chaperones (sometimes also known as chaperonins). Ellis was the first person who gave the concept of chaperones [57]. Since then, to explore the functional and physiological implications of these molecular chaperones a very active field of research has been initiated. By reviewing major aspects of this field many details have been demonstrated in a more authentic manner [58].
Initially the proteins were defined as being heat inducible and now defined as having heat shock elements [59]. The major Hsp families along with their normal functions are given in Table 2. A number of genetic and acquired diseases result from the abnormal protein activity because mutations cause the up- or down-regulation of the gene product and its function. On the other hand some diseases are due to the synthesis of a mutant or misfolded protein, which gets a novel activity (such as the tendency to from aggregates) with pathological characteristics. Different human diseases, which involve protein misfolding have been studied [60,61].
| Family | Size (kDa) | Major Functions |
|---|---|---|
| Hsp100 | ≥100 | Protein disaggregation, thermotolerance |
| Hsp90 | 81-99 | Regulatory interactions with signaling proteins, stabilization of misfolded proteins |
| Hsp70 | 65-80 | Protein folding, membrane transport of proteins |
| Hsp60 | 55-64 | Protein folding (limited substrates in eukaryotic cytoplasm) |
| Hsp40 | 35-54 | Protein folding, co-chaperons for Hsp 70 |
| Small Hsp | ≤34 | Stabilization of misfolded proteins, thermotolerance |
Table 2: Size and functions of HSP families [64]
Protein folding diseases or conformational diseases e.g. AD have various etiologies, which include changes in the primary structure of proteins, defects in chaperones and the incongruous presence or effect of other proteins [62]. Misfolding of proteins and protein aggregation are controlled by molecular chaperones such as heat shock proteins (Hsps). It has been illustrated with the great understanding of folding in vivo as well as in vitro that many diseases arise as a result of protein misfolding [63].
Molecular chaperone in AD
Molecular chaperones have been found to be linked to a great extent with Aβ-plaques and neuronal NFTs in astrocytes and oligodendrocytes in the brains of AD patients in comparison to control brain [64]. Molecular chaperones Hsp20, 27, 72 and 90 are linked with Aβ plaques, while Hsp72 and the small Hsp αB-crystallin (20 kDa) are linked with glial but not neuronal NFTs in AD brains. Hsp72 and Hsp73 are both induced in the brains of AD versus control brains [65], whereas Hsp27 is induced in cortical astrocytes and αB-crystalline is found in temporal and frontal lobe astrocytes and oligodendrocytes [66]. But the mechanism of functioning of molecular chaperones is not clear in the pathogenesis of AD.
Acquired chaperonopathies are associated with post-translational modifications of chaperones and usually, become clinically evident in late-life and cause neurodegenerative disorders as in AD. The modifications lead to the disruption of chaperones and they become unable to perform their duty normally. Thus, these incompetent chaperones fail to meet the excessive need of proteins repair [67]. As the disease progresses, chaperones levels become increased or decreased. However, the mechanism of these to occur is still not fully understood. Recent studies have revealed that these quantitative alterations in the levels may be due to the modifications in heat-shock factors, which regulates chaperones gene transcription. These changes lead to up or down regulation of chaperone genes, mutations of these genes or post-translational modifications of the chaperone itself [68]. The effects on levels of chaperones, which are involved in AD are given in Table 3 [69].
| Chaperones | Effect on levels | Method |
|---|---|---|
| Hsp72 | Elevates in tau and NFTs | Immunohistochemistry, WB |
| Grp78 | Elevates in neurons | Immunohistochemistry, WB |
| Hsp72 | Elevates in brain cortical tissue | WB, synthesis in vitro |
| Hsp60, Hsp70RY, Hsc70, αB-crystalline, Grp75, Grp94 | Unusual spreading in various brain areas | 2-D gels plus MALDI and mass spectrometry |
NFTs: neurofibrillary tangles
Table 3: Chaperones associated with Alzheimer’s disease [69]
Hsp as therapeutic agents for AD
Molecular chaperones also reveal in vitro protective effects in AD models. The endoplasmic reticulum Hsp32, Hsp70, Hsp90 has all been found to increase the clearance of Aβ after Aβ40- or Aβ42-induced toxicity in vitro [70]. In addition, Hsp20, 27 and αB-crystallin all reduce the aggregation of Aβ40, but not Aβ42 in to mature fibrils [71]. Hsp inducers clear p-tau from cells in vitro. These Hsp-mediated pharmacological intervention would be helpful in AD [72].
Non-Genetic Risk Factors of Ad
Cerebrovascular changes
Hemorrhagic infarcts, vasculopathies, small and large ischemic cortical infarcts and changes in white matter enhance the risks of dementia. White matter hyperintensies or infarcts can increase Aβ deposition that in turn leads towards cognitive decline. Also, hypoperfusion can cause overexpression of CDK 5 that is a serine-threonine kinase and responsible for synapse formation and synaptic plasticity [73]. This kinase is also involved in abnormal phosphorylation of tau and contributes towards NFTs formation therefore, a key protein linking NFT pathology to amyloid plaques.
Type 2 diabetes (T2D)
It has been reported that T2D nearly doubles the risk of AD [74]. Cerebrovascular and non-cerebrovascular mechanisms could be involved in linking T2D and LOAD. Hyperinsulinemia and advanced products of glycosylation are non-cerebrovascular mechanisms, which link T2D with LOAD. The clearance of Aβ in brain is linked with insulin-degrading enzyme (IDE). Insulin and Aβ have been considered as competing substrates for IDE. In brain, insulin is involved in the deposition of Aβ and tau protein phosphorylation, which cause pathogenesis of LOAD [75].
Metabolic syndrome
Yaffe et al. [76] have assessed the relationship between risk of cognitive decline or AD and metabolic syndrome as a whole. They reported that there is a positive correlation between cognitive dysfunction and syndrome presence.
Traumatic brain injury (TBI)
Schofield et al. [77] suggested that individuals with TBI history are at a higher risk of dementia as compared to those with no such history. Franz et al. [78] reported that the extent of Aβ and tau pathologies increase in the tissues of human brain after injury and levels of cerebrospinal fluid Aβ and APP are also increased.
Prevalence of AD in Pakistan
As it has described that AD is the most common kind of dementia around the world with the increasing life span and changing population demographics. It has become the cause of significant and increasing proportion of morbidity and fatality. The proportion of the disease is also increasing with the passage of time in Pakistan. A short analysis of this increasing number from 2012 to 2015 has given by graphical representation. This data was collected randomly from all government hospitals of Lahore, Pakistan.
The total number of patients from 2012 to 2015 was 128, 130, 250 and 205 approximately in the respective years. Out of these patients, female patients were 80, 90, 204 and 170, while the number of male patients was 48, 40, 46 and 35 in the respective years. These female and male patients were grouped in two categories according to their age and these groups were, number of patients of age more than 71; number of patients of age more than 65. The number of patients of these age groups by gender is given in Table 4.
| Age | Age group by gender | Years | |||
|---|---|---|---|---|---|
| 2012 | 2013 | 2014 | 2015 | ||
| Patients of age more than 71 | Females | 18 | 23 | 40 | 47 |
| Males | 12 | 22 | 55 | 45 | |
| Patients of age more than 65 | Females | 58 | 45 | 80 | 43 |
| Males | 40 | 40 | 75 | 70 | |
Table 4: Patients according to age group
It can be seen from Table 4 that occurrence of disease is more common in females due to expectancy of longer life and postmenopausal oestrogen deficiency, which leads to suppress the plasticity of axons [79]. In Pakistan, the patients of AD were treated with Exelon but now memantine is used for the treatment. Moreover, patients need care giving tasks that include social support, nursing care and medical care.
Mechanism of Action of Nonsteriodal Antiinflammatory Drugs (NSAIDs)
To reduce the prevalence and risk of AD or to delay the progression of disease NSAIDs have been found very effective. Peroxisome proliferator-activated receptor γ (PPARγ) a ligand-activated nuclear receptor has been explored as an appreciated target of NSAIDs. PPARγ acts as a DNA-binding transcription factor and its transcriptional regulatory effects get activated after the binding of agonist. The drugs of thiazolidinedione class and natural ligand prostaglandin J2 have been explored as PPARγ agonists and are involved in the inhibition of Aβ-stimulated secretion of proinflammatory products through microglia and monocytes, which are involved in the activation of neurotoxicity and astrocyte [80]. As these agents inhibit a wide range of inflammatory responses therefore, PPARγ agonists have been considered as novel approach towards AD cure.
Concluding Remarks
The toxic roles of various polypeptides have been suggested in AD pathogenesis but Aβ has appeared as a chief neurotoxic peptide. Many studies have demonstrated that Aβ-aggregates are due to the structural shift (α- to β-sheet conformation) and this leads to the hyper phosphorylation of tau. These are the two key steps in developing the AD. Another factor is the abnormal chaperones or defective proteosomal pathway that aids the disease to progress towards the severity. The aggregation of tau proteins in filamentous insertions is a common property of various neurodegenerative diseases including AD. In AD, tau becomes hyperphosphorylated, which eventually results in the formation of NFTs. Decreasing kinase activities e.g. GSK activity or increasing phosphatase activities e.g. serine/threonine-PP2A needs to be translated into clinical trials for the treatment of AD. In AD, the balance between the formation and removal of Aβ peptides has been disturbed and it has resulted in an enhanced concentration of amyloid peptides and development of cytotoxic aggregates. New techniques to restore this balance would offer auspicious treatments of this disease.
New chaperonopathies need to be discovered through applied or clinical investigations and to expand knowledge about those, which have been already identified. Then, elucidation of mechanisms of defective chaperones would be helpful to reveal their contribution towards AD. In future, there is a need for the determination of prevalence and full knowledge about medical consequences regarding chaperonopathies for the optimization of diagnostic tests and to make strategies to prevent and cure the disease. The recent research approaches are to provide the therapeutic agents for treating the AD. The experiments on animal model to find out the effective pharmacological interventions by using the β-sheet breakers, Aβ immunizations and beneficial chaperones are the main focus of research. This will provide a cure for AD in next few years.
References
- Matthews GG. Neurobiology: molecules, cells and systems. Hoboken, New Jersey: Wiley-Blackwell; 2000.
- Corder EH, Ghebremedhin E, Taylar MG, Thal DR, Ohm TG, Braak H. The biphasic relationship b/w regional brain senile plaque and neurofibrillary Tangle distribution: modification by age, sex and APOE polymorphism. Ann N Y Acad Sci 2004;1019:24-8.
- Hoozemans JJM, Veerhuis R, Rozemuller JM, Eikelenboom P. Neuroinflamation and regeneration in the early stages of Alzheimer’s disease pathology. Int J Dev Neurosci 2006;24:157-65.
- Hamilton RL. Lewy bodies in Alzheimer's disease: a neuropathological review of 145 cases using α‐synuclein Immunohistochemistry. Brain Pathol 2000:10:378-84.
- Pei JJ, Khatoon S, An WL, Nordlinder M, Tanak T, Braak H, et al. Role of protein kisase B in Alzheimer’s neurofibrillary pathology. Acta Neuropathol 2003;105:381-92.
- Mendez MF, Lee AS, Joshi A, Shapira JS. Nonamnestic presentations of early-onset Alzheimer’s disease. Am J Alzheimers Dis Other Demen 2012;27:413-20.
- Farhan S, Fahiem MA, Tahir F, Tauseef H. A comparative study of neuroimaging and pattern recognition techniques for estimation of Alzheimer’s. Life Sci J 2013;10:7s.
- Hamdane M, Delobel P, Sambo AV, Smet C, Begard S, Violleau A, et al. Neurofibrillary degeneration of the Alzheimer-type: an alternate pathway to neuronal apoptosis. Biochem Pharmacol 2003;66:1619-25.
- Samii A, Nutt JG, Ranson BR. Parkinson’s disease. Lancet 2004;363:1783-93.
- Hebert LE, Scherr PA, Bienias JL, Bennett DA, Evans DA. Alzheimer’s disease in the US population: prevalence estimates using the 2000 census. Arch Neurol 2003;60:1119-22.
- Hebert LE, Beckett LA, Schere PA, Evans DA. Annual incidence of Alzheimer’s disease in the United States projected to the years 2000 through 2050. Alzheimer Dis Assoc Discord 2001;15:169-73.
- Liebscher S, Meyer-Luehmann M. A peephole into the brain: Neuropathological features of Alzheimer’s disease revealed by in vivo two-photon imaging. Front Psychiatry 2012;3:26.
- Kamagata E, Kudo T, Kimura R, Tanimukai H, Morihara T, Sadik MDG, et al. Decrease of dynamin 2 levels in LOAD alters Aβ-metabolism. Biochem Biophys Res Commun 2009;379:691-95.
- Suh YH, Checler F. Amyloid precursor protein, presenilins, and α-synuclein: molecular pathogenesis and pharmacological applications in Alzheimer's disease. Erratum in Pharmacol Rev 2002;54:469-25.
- Vilatela MEA, Lopez-Lopez M, Yescas-Gomez P. Genetics of Alzheimer’s disease. Arch Med Res 2012;43:622-31.
- Berezovska O, Lleo A, Herl LD, Frosch MP, Stern EA, Bacskai BJ, et al. Familial Alzheimer's disease presenilin 1 mutations cause alterations in the conformation of presenilin and interactions with amyloid precursor protein. J Neurosci 2005;25:3009-17.
- Pigino G, Morfini G, Pelsman A, Mattson MP, Brady ST, Busciglio J. Alzheimer's presenilin 1 mutations impair kinesin-based axonal transport. J Neurosci 2003;23:4499-08.
- Bell RD, Winkler EA, Singh I, Sagare AP, Deane R. Apolipoprotein E controls cerebrovascular integrity via cyclophilin A. Nature 2012;485:512-16.
- Combs B, Kneynsberg A, Kanaan NM. Gene therapy models of Alzheimer’s disease and other dementias. Methods Mol Biol 2015;339-66.
- Arriagada PV, Growdon TH, Hedley-whyte ET, Hyman BT. Neurofibrillary Tangles but not senile plaques parallel duration and severity of Alzheimer’s disease. Neurology 1992;42:631-39.
- Evin G, Weidemann A. Biogenesis and metabolism of Alzheimer’s disease Aβ-amyloid peptides. Peptides 2002;23:1285-97.
- Jellinger KA. Neuropathology of multiple system atrophy: new thoughts about pathogenesis. Mov Disord 2014;29:1720-41.
- Skovronsky DM, Lee VMY, Trojanowski JQ. Neurodegenerative disease: New concepts of pathogenesis and their therapeutic implication. Ann Rev Pathol 2006;1:151-70.
- Golde ET, Eckmann BC, Younkin SG. Biochemical detection of Aβ isoforms: implications for pathogenesis, diagnosis and treatment of Alzheimer’s disease. Biochim Biophys Acta 2000;1502:172-87.
- Kirkitadze MD, Bitan G, Teplow DB. Paradigm shifts in Alzheimer’s disease and other neurodegenerative disorders: The emerging role of oligomeric assemblies. J Neurusci Res 2002;69:567-77.
- Kayed R, Head E, Thompson JL, McIntire TM, Milton SC. Common structure of soluble amyloid oligomers implies common mechanism of pathogenesis. Science 2003;300:486-89.
- Sanchez-Varo R, Trujillo-Estrada L, Sanchez-Mejias E, Torres M, Baglietto-Vargas D, Moreno-Gonzalez I, et al. Abnormal accumulation of autophagic vesicles correlates with axonal and synaptic pathology in young Alzheimer’s mice hippocampus. Acta Neuropathol 2012;123:53-70.
- Suh YH, Checler F. Amyloid precursor protein, presenilins, and α-synuclein: molecular pathogenesis and pharmacological applications in Alzheimer's disease. Pharmacol Rev 2002;54:469-25.
- Kang J, Lemaire HG, Unterbeck A, Salbaum JM, Masters CL, Grzeschik KH, et al. The precursor of Alzheimer’s disease amyloid Aβ protein resembles a cell surface receptor. Nature 1987;325:733-36.
- Small DH, McLean CA. Alzheimer's disease and the amyloid β protein. J Neurochem 1999;73:443-49.
- Pasternak JJ. An introduction to human molecular genetics: mechanisms of inherited diseases. Hoboken, New Jersey: John Wiley and Sons; 2005.
- Lichtenthaler FS. Alpha-secretase cleavage of the amyloid precursor protein: proteolysis regulated by signaling pathways and protein trafficking. Curr Alzheimer Res 2012;9:165-77.
- Devi L, Ohno M. Mitochondrial dysfunction and accumulation of the β-secretase-cleaved C-terminal fragment of APP in Alzheimer's disease transgenic mice. Neurobiol Dis 2012;45:417-24.
- Buee L, Bussiere T, Buee-Scherrer V, Delacourte A, Hof PR. Tau protein isoforms, phosphorylation and role in neurodegenerative disorders. Brain Res Rev 2000;33:95-30.
- Neselius S, Zetterberg H, Blennow K, Randall J, Wilson D, Marcusson J, et al. Olympic boxing is associated with elevated levels of the neuronal protein tau in plasma. Brain Injury 2013;27:425-33.
- Blennow K, Zetterberg H. The application of cerebrospinal fluid biomarkers in early diagnosis of Alzheimer disease. Med Clin North Am 2013;97:369-76.
- Rademakers R, Cruts M, van Broeckhoven C. The role of tau (MAPT) in frontotemporal dementia and related tauopathies. Hum Mutat 2004;24(4):277-95.
- Neve RL, Harris P, Kosik KS, Kurnit DM, Donlon TA. Identification of cDNA clones for the human microtubule-associated protein tau and chromosomal localization of the genes for tau and microtubule-associated protein 2. Brain Res 1986;387:271-80.
- Lee HG, Perry G, Moreira PI, Garrett MR, Liu Q, Zho X, et al. Tau phosphorylation in Alzheimer’s disease: pathogen or protector? Trends Mol Med 2005;11:164-69.
- Badiola N, Suarez-Calvet M, Lleo A. Tau phosphorylation and aggregation as a therapeutic target in tauopathies. CNS Neurol Disord Drug Targets 2010;9:727-40.
- Morrison RS, Kinoshita Y, Johnson MD, Uo T, Ho JT, McBee JK, et al. Proteomic analysis in the neurosciences. Mol Cell Proteom 2002;1:553-60.
- Bulic B, Pickhardt M, Mandelkow E. Progress and developments in tau aggregation inhibitors for Alzheimer disease. J Med Chem 2013;56:4135-55.
- Serpell LC, Blake CC, Fraser PE. Molecular structure of a fibrillar Alzheimer's Aβ fragment. Biochemistry 2000;39:13269-75.
- Wang Q, Yu X, Li L, Zheng J. Inhibition of amyloid-β aggregation in Alzheimer's disease. Curr Pharm Des 2014;20:1223-43.
- Wilhelmus MM, Otte-Holler I, Wesseling P. Specific association of small heat shock proteins with the pathological hallmarks of Alzheimer’s disease brains. Neuropathol Appl Neurobiol 2006;32:119-30.
- Niu L, Liu L, Xu M, Cramer J, Gothelf KV, Dong M, et al. Transformation of β-sheet structures of the amyloid peptide induced by molecular modulators. Chem Commun 2014;50:8923-26.
- Sparvero LJ, Patz S, Brodsky JL, Coughlan CM. Proteomic analysis of the amyloid precursor protein fragment C99: expression in yeast. Anal Biochem 2007;370:162-70.
- Kauwe JS. Extreme cerebrospinal fluid amyloid beta levels identify family with late-onset Alzheimer’s disease presenilin 1 mutation. Ann Neurol 2007;61:446-53.
- Harrington CR. The molecular pathology of Alzheimer's disease. Neuroimaging Clin N Am 2012;22:11-22.
- Liu Z, Li T, Li P, Wei N, Zhao Z, Liang H, et al. The ambiguous relationship of oxidative stress, tau hyperphosphorylation, and autophagy dysfunction in Alzheimer’s disease. Oxid Med Cell Longev 2015:2015;352723.
- O’Callaghan C, Fanning LJ, Barry OP. p38δ MAPK: emerging roles of a neglected isoform. Int J Cell Biol 2014:2014;272689.
- Wolfe MS. The role of tau in neurodegenerative diseases and its potential as a therapeutic target. Scientifica 2012:2012;796024.
- Yaniv Y, Spurgeon HA, Ziman BD, Lakatta EG. Ca2+/calmodulin-dependent protein kinase II (CaMKII) activity and sinoatrial nodal pacemaker cell energetics. PLoS One 2013;8:e57079.
- Lund H, Cowburn RF, Gustafsson E, Strömberg K, Svensson A. Tau‐tubulin kinase 1 expression, phosphorylation and co‐localization with phospho‐ser422 tau in the Alzheimer's disease brain. Brain Pathol 2013;23:378-89.
- Henriksen K, O’Bryant SE, Hampel H, Trojanowski JQ, Montine TJ, Jeromin A, et al. The future of blood-based biomarkers for Alzheimer's disease. Alzheimer's Dement 2014;10:115-31.
- Rosenmann H. CSF biomarkers for amyloid and tau pathology in Alzheimer's disease. J Mol Neurosci 2012;47:1-14.
- Ellis J. Proteins as molecular chaperones. Nature 1987;328:378-79.
- Gething MJ. Guidebook to molecular chaperones and protein-folding catalyst. Oxford, UK: Oxford University Press; 1997.
- Lindquist S. Heat‐shock proteins and stress tolerance in microorganisms. Curr Opin Genet Dev 1992;2:748-55.
- Ruddon RW, Sherman SA, Bedows E. Protein folding in the endoplasmic reticulum: Lessons from the human chorionic gonadotropin beta subunit. Protein Sci 1996;5:1443-52.
- Thomas PJ, Qu BH, Pedersen PL. Defective protein folding as a basis of human disease. Trends Biochem Sci 1995;20:456-59.
- Fiorelli TN. Proteolytic processing of the amyloid precursor protein during apoptosis and cell cycle: implications for Alzheimer's disease [dissertation]. Tampa, Florida: University of South Florida; 2013.
- Knowles TP, Vendruscolo M, Dobson CM. The amyloid state and its association with protein misfolding diseases. Nat Rev Mol Cell Biol 2014;15:384-96.
- Eckert A, Nisbet R, Grimm A, Götz J. March separate, strike together—Role of phosphorylated TAU in mitochondrial dysfunction in Alzheimer's disease. Biochim Biophys Acta 2011;1842:1258-66.
- Perez N, Sugar J, Charya S. Increased synthesis and accumulation of heat shock 70 proteins in Alzheimer’s disease. Mol Brain Res 1991;11:249-54.
- Renkawek K, Bosman GJ, Gaestel M. Increased expression of heatshock protein 27 kDa in Alzheimer disease: A preliminary study. Neuroreport 1993;5:14-16.
- Hardy J, Selkoe DJ. The amyloid hypothesis of Alzheimer’s disease: progress and problems on the road to therapeutics. Science 2002;297:353-56.
- Ha CE, Bhagavan NV. Essentials of medical biochemistry: with clinical cases. Cambridge, Massachusetts: Academic Press; 2011.
- Keller JN, Hanni KB, Markesbery WR. Impaired proteasome function in Alzheimer’s disease. J Neurochem 2000;75:436-39.
- Yang Y, Turner RS, Gaut JR. The chaperone BiP/GRP78 binds to amyloid precursor protein and decreases Aβ40 and Aβ42 secretion. J Biol Chem 1998;273:25552-55.
- Wilhelmus MM, Boelens WC, Otte-Höller I, Kamps B, Kusters B, Maat-Schieman ML, et al. Small heat shock protein HspB8: its distribution in Alzheimer’s disease brains and its inhibition of amyloid-β protein aggregation and cerebrovascular amyloid-β toxicity. Acta Neuropathol 2006;111:139-49.
- Donev R, Kolev M, Millet B, Thome J. Neuronal death in Alzheimer’s disease and therapeutic opportunities. J Cell Mol Med 2009;13:4329-48.
- Cheung ZH, Gong K, Ip NY. Cyclin-dependent kinase 5 supports neuronal survival through phosphorylation of Bcl-2. J Neurosci 2008;28:4872-77.
- Luchsinger JA, Tang MX, Stern Y, Shea S, Mayeux R. Diabetes mellitus and risk of Alzheimer’s disease and dementia with stroke in a multiethnic cohort. Am J Epidemiol 2001;154:635-41.
- Park CR. Cognitive effects of insulin in the central nervous system. Neurosci Biobehav Rev 2001;25:311-23.
- Yaffe K, Weston AL, Blackwell T, Krueger KA. The metabolic syndrome and development of cognitive impairment among older women. Arch Neurol 2009;66:324-28.
- Schofield PW, Tang M, Marder K, Bell K, Dooneief G, Chun M, et al. Alzheimer’s disease after remote head injury: an incidence study. J Neurol Neurosurg Psychiatry 1997;62:119-24.
- Franz G, Beer R, Kampfl A, Engelhardt K, Schmutzhard E, Ulmer H, et al. Amyloid beta 1-42 and tau in cerebrospinal fluid after severe traumatic brain injury. Neurology 2003;60:1457-61.
- Bhavnani BR. Estrogens and menopause: pharmacology of conjugated equine estrogens and their potential role in the prevention of neurodegenerative diseases such as Alzheimer’s. J Steroid Biochem Mol Biol 2003;85:473-82.
- Combs CK, Johnson DE, Karlo JC, Cannady SB, Landreth GE. Inflammatory mechanisms in Alzheimer's disease: inhibition of β-amyloid-stimulated proinflammatory responses and neurotoxicity by PPARγ agonists. J Neurosci 2000;20:558-67.