- *Corresponding Author:
- Bhagyashree Patil
Department of Biology, College of Science, Jazan University, Jazan 82817, Saudi Arabia
E-mail: bpatil@jazanu.edu.sa
| Date of Received | 15 November 2024 |
| Date of Revision | 30 December 2024 |
| Date of Acceptance | 06 January 2025 |
| Indian J Pharm Sci 2025;87(1):1-13 |
This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms
Abstract
Leptin hormone is very well known to play a multifarious role in body. Although it is secreted by adipocytes, it has been observed to exert its effects beyond scientists’ initial speculation periphery of obesity. The neuroendocrine pathway of leptin is known and its role in obesity is understood considerably. Yet the role of leptin like undercurrents of the sea in case of cardiovascular health and initiation of cancer has started to uncover itself recently. The aim of the review is to encompass the recent scientific studies related with leptin levels and its direct effect or an indirect metabolic effect on the initiation and/or propagation of cardiac health and cancer. Many investigations are still underway to recognize the underlying mechanisms, it seems the complete uncovering of the so far unknown leptin function pathways may take some time. The effects on cardiac health have been found to have multiple metabolic pathways. Some of which are discovered up to molecular mechanisms. However, cancer related studies are still unclear and often ambiguous while finding the exact cascade of events. This may be partly because cancer originates in multiple organs. Moreover, numerous signalling, molecular and metabolic as well as other functional pathways are involved in cancer commencement and propagation. Last part of review summarises is interplay between the leptin gene with some other crucially important genes which regulate the energy balance, and are expressed on variety of cellular sites.
Keywords
Leptin gene, Leptin, Obesity, Heart, Cardiac, Cancer, Gene interactions
In year 2014, already 20 y later of its discovery in the year 1994, it was realized that the hormone leptin is responsible for more than obesity. Hence, the Lancet article by Norra MacReady ended with speculation that next 20 y may uncover a whole data of disorders commencing from either resistance or deficiency associated with leptin. This review aims at reviewing the discoveries associated with leptin during the last decade[1].
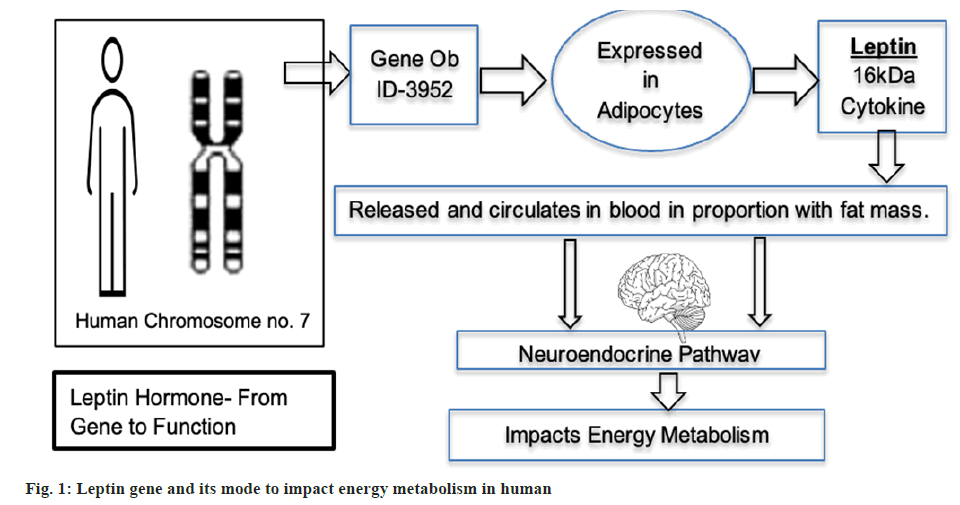
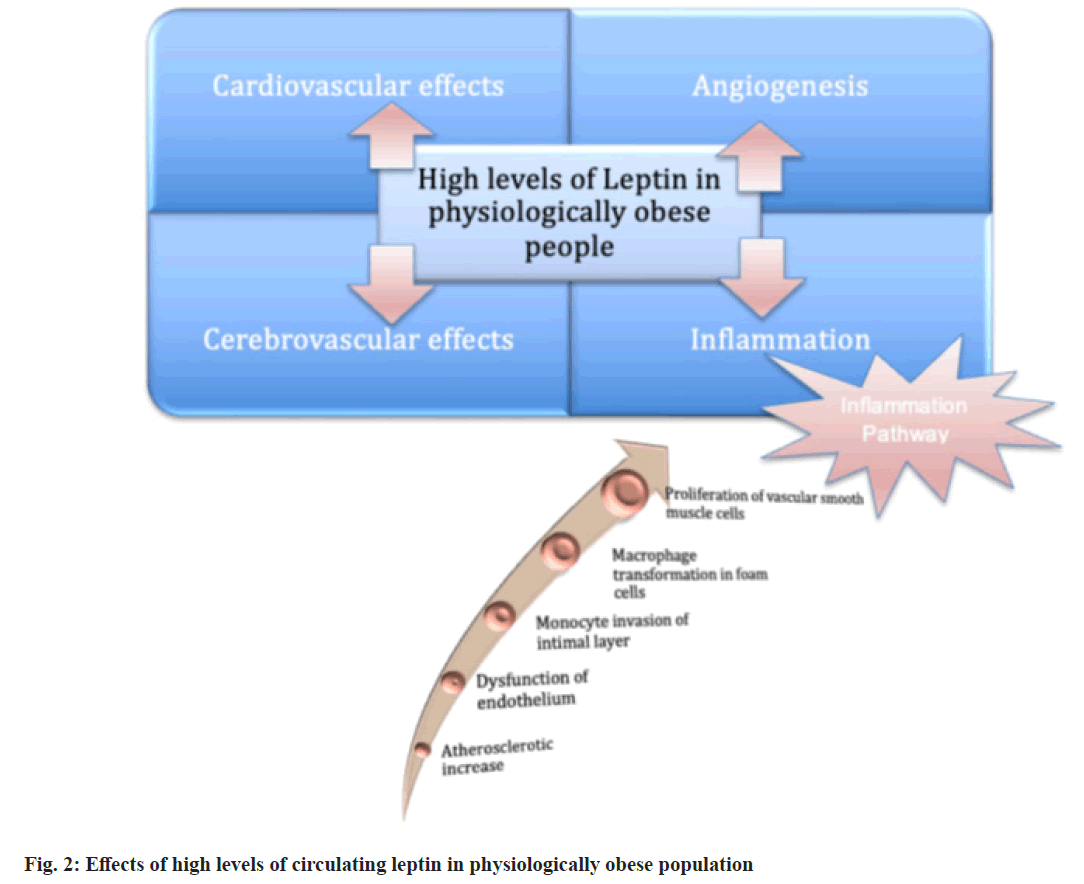
Previously considered as an inert mass, the adipocytes (WAT) entered the cascade of endocrinology and neuroendocrinology once they were identified as being responsible for the expression of obese or leptin gene and their product of expression was a hormone leptin[2,3]. Although variability in plasma leptin concentration was found associated with Body Mass Index (BMI)[3]. In humans it is present on chromosome no. 7 and is given the ID 3952[4]. Basically a cytokine, leptin with its 16-kDa or 16 000 g per mole of molecular weight is released into blood circulation by adipocytes, in proportion with their mass. Leptin is mainly concerned with energy metabolism which also involves the neuro-signalling pathway[2,3]. High levels of leptin have some biased role in physiology of obese people which is observed in cardiovascular (hypertension, atherosclerosis, myocardial infarction) cerebrovascular and other ailments like inflammation and angiogenesis. Leptin works both autocrine and paracrine manner on some occasions, even considered as beneficial factor post myocardial infarction (fig. 1)[5-9].
Among undesired effects of leptin to mention- prominent one is atherosclerosis increase, the end result of which is secretion of a pro-atherogenic cytokine protein which occurs via (1) dysfunction of the endothelium, intimal layers invaded by monocyte; macrophages-to-foam-cell transformation and proliferation of the vascular smooth muscle cell[6]. Nevertheless, leptin is also confirmed to have anti-apoptotic effects on cardiomyocytes induced by ischemia-reperfusion injury, hydrogen peroxide induced apoptosis, hypoxia reoxygenation (fig. 2)[7-9].
Methodology
This review was prepared using original research articles and reviews available from Saudi digital library. The journals and articles from Wiley Online Library, Clairvita web of science were searched using its customized search engines. Key word used were leptin. After applying exclusion criteria to stay aligned with the aim of the present review, full text articles were selected for this narrative review based upon the content. Following table describes the details Table 1.
| Sr. no. | Criteria | Details |
|---|---|---|
| 1 | Digital access portal for journal databases | Saudi digital library |
| 2 | Databases searched | Wiley online library Web of science, Nature journals |
| 3 | Search durations | August 2023 to December 2024 |
| 4 | Search keyword | By official symbol ‘LEP’ and official full name ‘leptin’ |
| 5 | Article publication interval | No limits applied |
| 6 | Article exclusion criteria | Articles unrelated to Biology and Genetics |
| 7 | Article inclusion criteria | LEP gene studies single or in association with other genes, its polymorphisms, population studies |
| 8 | Flow charts | All flowcharts were prepared using licensed Microsoft word and PowerPoint tools |
| 9 | Ethical compliance | Not applicable as the present article is of narrative review type and previously published research articles are utilized |
Table 1: Review Methodology Details
Leptin and heart:
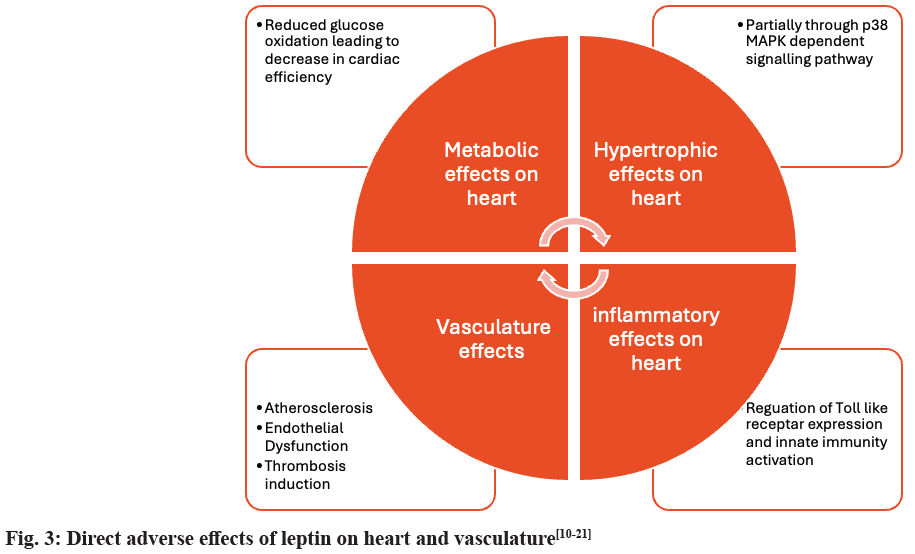
Cardiac health: The metabolic effects exerted by leptin cause some of the undesirable but direct adverse effects on the heart. The metabolic effects can be listed as shrunk oxidation of the primary source of energy that is glucose, secondly amplified fatty acid oxidation.
Consequences of these previous two are observed as an obvious cardiac insufficiency[10,11] second to which comes hypertrophy which is fractionally exerted through a p38 Mitogen-Activated Protein Kinase (MAPK)-dependent signalling pathway[12,13], and an inflammatory effect (through regulating toll like receptor expression and inmate immunity activation) in preadipocytes and adipocytes[14]. Leptin also has direct effects on the vasculature, including atherosclerotic effects on monocytes[15], endothelial dysfunction resulting from the long-term effect of leptin on no synthesis and disturbed bioavailability[16], and thrombosis induction (through platelet aggregation via cyclic Guanosine Monophosphate (cGMP) inhibited 3′,5′-cyclic phosphodiesterase 3a and thrombus formation)[17-19]. Altogether, leptin could result in increased arterial stiffness (fig. 3)[20,21].
Physiological relation of leptin with cancer:
Breast cancer: Multiethnic Cohort studies involving 706 postmenopausal breast cancer patients revealed significantly elevated serum levels of leptin compared to matched controls. Women with the highest prediagnostic levels of leptin, leptin: Adiponectin ratio, and C-Reactive Protein (CRP) exhibited an increased risk of postmenopausal breast cancer[22]. Additionally, leptin was found to be significantly overexpressed in breast cancer tissues compared to non-cancerous tissues[23].
Colorectal cancer: Leptin levels were notably higher in colorectal cancer patients compared to controls[24].
Papillary thyroid cancer: Significant elevations in serum leptin levels were observed in papillary thyroid cancer patients[25].
Prostate cancer: Prostate cancer patients exhibited significantly higher leptin messenger Ribonuclic Acid (mRNA) expression levels compared to healthy controls[26].
Acute lymphoid leukaemia: Serum leptin levels were significantly elevated in acute lymphoid leukemia patients[27,28].
Multiple myeloma: Multiple myeloma patients showed significantly higher leptin levels compared to controls[29].
Glioblastoma: Leptin was overexpressed in glioblastomas relative to normal glial tissues[30].
Hepatocellular carcinoma: Leptin levels were significantly higher in hepatocellular carcinoma patients compared to controls[31].
Ovarian Cancer: High expression of leptin was observed in ovarian cancer patients[32].
Endometrial cancer: Leptin was overexpressed in endometrial cancer specimens compared to normal endometrial tissue samples[33].
Population based studies involving leptin levels or gene and polymorphism as shown in Table 2[34-43].
| Sr. no. | Ethnicity or nationality | Gender | Result/conclusion | Reference |
|---|---|---|---|---|
| 1 | Iranian population with confirmed lung cancer | NS | No association of studied polymorphism of LEP (-2548G/A) with lung cancer | [34] |
| 2 | Caucasian | Male | Obesity is linked to the accumulation of numerous variants of other genes along with lep. | [35] |
| 3 | Saudi population with obesity and T2D with controls | NS | A link is present between common -2548G>A (rs7799039) promoter variant of the human leptin gene (LEP) with leptin and serum glucose leptin levels in obese Saudi patients. This finding is irrespective of blood pressure status of patients. | [36] |
| 4 | CIBERSORT | Female | Lep expression can be diagnostic biomarker of Preeclampsia | [37] |
| 5 | Metanalysis | Female | LEP rs7799039 and leptin receptor rs1137101 polymorphisms were not associated with increased risk of Breast cancer | [38] |
| 6 | Chinese population | Mixed | Leptin and body mass are associated | [39] |
| 7 | Multiethnic postmenopausal 706 patients | Female | Significantly elevated leptin levels in postmenopausal woman as risk factor for breast cancer (including other factors) | [40] |
| 8 | South Indian population | Mixed healthy | Common polymorphisms in the leptin gene are strong predictors of obesity and leptin levels in South Indian population | [41] |
| 9 | Egyptian population | Systemic lupus erythromatosus | No association between leptin levels and gene polymorphism and SLE | [42] |
| 10 | Arabic population of Oman | Male and Female | Gender-specific reference ranges for serum leptin levels reported | [43] |
Table 2: Human Population Studies Related With Leptin
Leptin gene interactions with other significant genes:
The interesting cascade of interactions of leptin gene with other important genes is important to understand role of leptin gene in humans and animals.
Leptin receptor and leptin association: Recent structural studies have provided significant insights into the mechanism of leptin receptor activation. Leptin binding induces a conformational change in leptin receptor that facilitates receptor dimerization and subsequent intracellular signalling. The leptin- leptin receptor complex exhibits structural homology with the Interleukin-6 (IL-6) family cytokine receptor complex exhibits structural homology with the IL-6 family cytokine receptor complexes, particularly in the docking modes of site 2 and site 3 interactions. Notably, the leptin-bound leptin receptor complex forms an asymmetric 2:2 homodimer, resembling the architecture of heterodimeric IL-6 family receptor complexes. This asymmetry suggests that a single leptin molecule can dimerize two leptin receptor chains, initiating downstream signal transduction[44].
Polymorphisms in the leptin receptor gene have been explored for their potential association with obesity and Type 2 Diabetes Mellitus (T2DM). A study focusing on the Korean population identified several Single Nucleotide Polymorphisms (SNPs) in the leptin receptor gene, including non-synonymous SNPs such as Arg109Lys and Arg223Gln. While no significant associations were found between these polymorphisms and the risk of T2DM, the Arg109Lys variant showed a marginal association with BMI, indicating a possible link to obesity[45].
Leptin’s regulatory effects on appetite and energy balance are mediated through specific neuronal populations in the hypothalamus. Traditionally, leptin was known to inhibit orexigenic Agouti-Related Protein (AGRP) neurons and activate anorexigenic Pro- Opiomelanocortin (POMC) neurons. However, recent research has identified a novel population of leptin- responsive neurons expressing Basonuclin 2 (BNC2) in the arcuate nucleus. Activation of BNC2 neurons acutely suppresses food intake by directly inhibiting AGRP neurons, highlighting a new component in the neural circuitry that maintains energy balance[46]. The functional significance of leptin receptor in various neuronal populations has been elucidated using advanced genetic tools. CRISPR-Cas9-mediated deletion of leptin receptor in AGRP neurons results in severe obesity and diabetes, mirroring the phenotype observed in leptin receptor-deficient (leptin receptor^db/ db^) mice. This finding underscores the critical role of leptin receptor in AGRP neurons for the regulation of energy balance and glucose homeostasis. In contrast, deletion of leptin receptor in POMC neurons has minimal effects, suggesting a lesser role in mediating leptin’s primary actions[47]. Activation of leptin receptor in turn drives the phosphorylation and activation of the transcription factor Signal Transducer and Activator of Transcription 3 (STAT3), which drives production of anorexigenic peptides that suppress food intake and increase energy expenditure[48].
Leptin receptor and STAT3 activation: The leptin receptor exists in multiple isoforms, with the long form (Ob-Rb) being chiefly responsible for signal transduction. Upon leptin binding, Ob-Rb undergoes conformational changes that activate associated Janus Kinase 2 (JAK2). Activated JAK2 phosphorylates specific tyrosine residues on Ob-Rb, creating docking sites for STAT3. Subsequent phosphorylation of STAT3 leads to its dimerization and nuclear translocation, where it modulates the transcription of target genes involved in energy homeostasis and appetite control. This mechanism was elucidated through studies demonstrating leptin-induced STAT3 activation in the hypothalamus of wild-type and ob/ob mice, but not in db/ db mice lacking functional Ob-Rb[49]. STAT3 activation is crucial for leptin's regulatory effects on food intake and body weight. Mice with neuron-specific disruptions of STAT3 exhibit hyperplasia, obesity, and impaired glucose tolerance, underscoring STAT3’s essential role in mediating leptin's anorexigenic effects furthermore, studies utilizing cell-permeable phosphopeptides to inhibit STAT3 activation in vivo have demonstrated that leptin's ability to acutely reduce food intake and influence hepatic glucose fluxes is critically dependent on intact STAT3 signalling[50].
STAT3-independent pathways: While STAT3 is vital for many of leptin’s actions, certain physiological processes are regulated via STAT3-independent mechanisms. For instance, research involving mice with a mutated leptin receptor incapable of STAT3 signalling (s/s mice) revealed that, although these mice developed obesity similar to db/db mice, they maintained relatively normal reproductive function. This finding suggests that leptin’s role in reproduction may be mediated through alternative pathways[51]. Dysregulation of the leptin- STAT3 axis has been implicated in various pathological conditions. In obesity, elevated leptin levels often lead to leptin resistance, characterized by impaired STAT3 signalling, which contributes to uncontrolled appetite and further weight gain. Additionally, aberrant leptin-STAT3 signalling has been associated with cancer progression. For example, in breast cancer, leptin-induced STAT3 activation recruits the histone methyltransferase G9a, leading to the repression of tumor suppressor genes and promotion of cancer cell proliferation[52].
Leptin and POMC neurons: POMC neurons, located in the arcuate nucleus of the hypothalamus, are integral to energy homeostasis. They produce the precursor peptide POMC, which is cleaved into several active peptides, including Alpha-Melanocyte-Stimulating Hormone (α-MSH). α-MSH acts on melanocortin receptors to suppress appetite and increase energy expenditure. Leptin receptors are expressed on a subset of POMC neurons, enabling leptin to modulate their activity directly. Approximately 30 % of hypothalamic POMC neurons respond to leptin, influencing metabolic processes[53,54]. Upon binding to leptin receptors on POMC neurons, leptin activates intracellular signalling pathways, including the JAK2 and Phosphoinositide 3-Kinase (PI3K) pathways. This activation leads to increased expression of POMC and subsequent release of α-MSH, promoting satiety and reducing food intake. Additionally, leptin's action on POMC neurons is influenced by glucose levels, with studies indicating that leptin’s effect on Gamma-Aminobutyric Acid (GABA) release to POMC neurons is modulated by glucose[55,56].
The interaction between leptin and POMC neurons is vital for maintaining energy balance. Disruption of leptin receptors in POMC neurons impairs glucose homeostasis and alters leptin secretion during fasting, underscoring the importance of this pathway in metabolic regulation[57]. Furthermore, leptin’s modulation of POMC neurons influences the expression of microRNAs targeting insulin signalling pathways, highlighting a complex network of regulatory mechanisms[58,59]. In obesity, leptin resistance impairs the leptin-POMC signalling pathway, leading to dysregulation of appetite and energy expenditure. Understanding the precise mechanisms of leptin’s action on POMC neurons offers potential therapeutic targets for obesity treatment. Modulating this pathway could restore leptin sensitivity and improve metabolic outcomes[2,60]. Leptin's interaction with POMC neurons is a cornerstone of energy homeostasis, influencing appetite suppression and metabolic regulation.
Disruptions in this pathway contribute to metabolic disorders, including obesity. Further research into the leptin-POMC axis holds promise for developing targeted therapies to address metabolic diseases.
Leptin's inhibitory effects on Neuropeptide Y (NPY) and Agouti-Related Peptide (AgRP) neurons: NPY and AgRP are potent orexigenic peptides produced in the arcuate nucleus of the hypothalamus. Leptin exerts inhibitory effects on NPY/AgRP neurons, thereby suppressing appetite and promoting energy expenditure. Studies have demonstrated that leptin administration significantly reduces Npy and Agrp mRNA expression in the hypothalamus, an effect mediated through the PI3K signalling pathway[61]. Inhibition of PI3K signalling impairs leptin's ability to suppress these orexigenic genes, underscoring the pathway’s crucial role in mediating leptin’s effects. Leptin modulates the intrinsic excitability of NPY/AgRP neurons. In diet-induced obese mice, persistent activation of NPY neurons is observed, and leptin’s efficacy in reducing this activity is diminished[62]. This suggests that leptin resistance in these neurons may contribute to the maintenance of obesity. Additionally, fasting induces a leptin-dependent increase in the intrinsic excitability of NPY/AgRP neurons, further illustrating leptin's role in modulating neuronal activity in response to energy status[63]. Recent research has identified Interferon Regulatory Factor 3 (IRF3) as a key mediator of leptin's acute hunger-suppressing effects in AgRP neurons[64]. Activation of IRF3 within these neurons contributes to the rapid suppression of hunger, indicating a complex intracellular network through which leptin exerts its anorexigenic effects.
Leptin's influence on Peroxisome Proliferator- Activated Receptor Gamma (PPARγ) activity: Leptin has been shown to counteract PPARγ’s inhibitory effects on chondrogenic differentiation and chondrocyte hypertrophy. In growth plate chondrocytes, leptin mitigates the suppressive actions of PPARγ, suggesting a modulatory role in skeletal development[65]. PPARγ directly influences leptin gene expression. A study identified a no canonical PPARγ/Retinoid X Receptor Alpha (RXRα)-binding sequence that regulates leptin expression in adipocytes, indicating that PPARγ can modulate leptin levels through direct interaction with its promoter region[66].
Mouse models have provided significant insights into the physiological relevance of leptin and PPARγ interactions. In leptin-deficient (ob/ob) mice with liver- specific disruption of PPARγ, a significant improvement in fatty liver is observed; however, these mice exhibit worsened hyperglycemia and insulin resistance, highlighting the tissue-specific roles of PPARγ in leptin-deficient states[67].
Leptin-Melanocortin-4 Receptor (MC4R) signalling Pathway:
Leptin exerts its effects by binding to receptors in the hypothalamus, leading to the activation of POMC neurons. These neurons produce α-MSH, which subsequently activates MC4R. Activation of MC4R results in reduced food intake and increased energy expenditure, underscoring its critical role in maintaining energy balance[68]. Mutations in either the leptin gene or the MC4R gene can disrupt this signalling pathway, leading to obesity. Studies have demonstrated that individuals with mutations in both genes exhibit an additive effect on fat mass, resulting in severe obesity. Furthermore, these mutations are associated with reduced efficacy of leptin in promoting weight loss and suppressing appetite, indicating a synergistic interaction between leptin and MC4R in energy homeostasis[69]. Leptin resistance, a common feature in obesity, is characterized by diminished sensitivity to leptin's effects. Research suggests that impaired MC4R signalling may contribute to leptin resistance. For instance, the absence of MC4R has been linked to a reduction in leptin's ability to decrease food intake and body weight, highlighting the receptor's role in mediating leptin's actions[70].
Leptin-induced modulation of Uncoupling Protein 2 (UCP2) expression: Leptin has been shown to influence UCP2 expression in various tissues. In neuronal cultures, leptin treatment induces UCP2 expression, which is associated with neuroprotective effects against toxic insults such as 1-Methyl-4-Phenylpyridinium (MPP) toxicity. This upregulation of UCP2 contributes to the maintenance of mitochondrial membrane potential and Adenosine Triphosphate (ATP) levels, thereby enhancing cell survival[71]. Similarly, in peripheral tissues, leptin administration has been observed to modulate UCP2 mRNA expression, suggesting a role in substrate metabolism and energy dissipation[72].
Tissue-specific effects of leptin on UCP2: The effect of leptin on UCP2 expression appears to be tissue- specific. For instance, chronic leptin administration decreases UCP2 protein abundance in the lung, while other mitochondrial proteins remain unaffected[73]. In skeletal muscle, central leptin administration increases UCP2 and UCP3 levels, which is consistent with enhanced mitochondrial function and thermogenesis[74]. These findings highlight the complex regulatory role of leptin on UCP2 expression across different tissues.
The interaction between leptin and UCP2 has significant implications for energy balance and metabolic health. UCP2 is known to uncouple oxidative phosphorylation, leading to reduced ATP production and increased heat generation. By modulating UCP2 expression, leptin can influence mitochondrial efficiency and Reactive Oxygen Species (ROS) production, thereby affecting metabolic rate and insulin sensitivity. Additionally, during states of altered energy demand, such as lactation, inhibition of leptin secretion is associated with downregulation of UCP expression in brown adipose tissue and skeletal muscle, indicating a coordinated regulation of energy expenditure[75].
Genetic associations between leptin and Glucokinase Regulator (GCKRL):
Genome-Wide Association Studies (GWAS) have uncovered significant associations between genetic variants near the leptin gene and the GCKR gene. Variants in these regions have been linked to circulating leptin levels, suggesting a genetic interplay that influences leptin concentrations and thereby affects energy balance and glucose metabolism[76,77].
GCKR harbors several polymorphisms that impact metabolic traits. The rs780094 variant, for example, has been associated with elevated fasting serum triglycerides and reduced fasting insulin levels, highlighting its role in lipid and glucose metabolism[78,79]. These metabolic alterations can indirectly affect leptin secretion and action, given leptin’s sensitivity to changes in energy storage and insulin signalling.
Pleiotropic effects and metabolic syndrome: Research demonstrates that both common and rare exonic mutations in GCKR exhibit pleiotropic effects on various metabolic parameters, including serum triglyceride and albumin levels, as well as the risk of metabolic syndrome[80,81]. Given leptin's role in energy homeostasis and fat storage, alterations in GCKR function may influence leptin dynamics, contributing to the development of metabolic syndrome. While direct molecular interactions between leptin and GCKR require further elucidation, several potential mechanisms have been proposed:
Regulation of glucose metabolism:
Leptin influences hepatic glucose production and insulin sensitivity. GCKR, by modulating glucokinase activity, plays a critical role in hepatic glucose utilization. Alterations in GCKR function could affect glucose levels, subsequently impacting leptin secretion and action[82,83].
Lipid metabolism: GCKR variants associated with dyslipidaemia may alter adipose tissue function, influencing leptin production. Conversely, leptin's role in lipid oxidation and storage could modulate hepatic lipid metabolism, potentially affecting GCKR activity[84,85].
Insulin signalling: Both leptin and GCKR are involved in insulin signalling pathways. Disruptions in GCKR function may impair insulin sensitivity, leading to compensatory changes in leptin levels, given leptin's role in modulating insulin action[86,87].
Genetic associations between LEP and Fat Mass and Obesity (FTO) associated gene:
FTO is known to influence energy intake and expenditure, with its variants affecting body weight regulation[88]. Research has shown that individuals carrying risk alleles of FTO have altered leptin sensitivity, which may contribute to impaired satiety signalling and increased food intake[89,90]. The interaction between leptin and FTO appears to play a significant role in energy balance. Leptin acts on the hypothalamus to regulate appetite and energy expenditure, while FTO influences these processes through its effects on RNA demethylation and metabolic regulation. Studies have shown that FTO variants can affect leptin signalling pathways, potentially leading to leptin resistance, a condition commonly observed in obesity[91,92]. Several mechanisms have been proposed to explain the interaction between leptin and FTO viz.
Regulation of energy intake: FTO variants are associated with increased energy intake, which may influence leptin production and action. Elevated energy intake can lead to increased adiposity, resulting in higher leptin levels[93,94].
Leptin resistance: FTO variants may contribute to leptin resistance by altering the leptin signalling pathway. This resistance impairs leptin's ability to regulate appetite and energy expenditure, promoting weight gain[95,96].
Metabolic regulation: FTO influences metabolic processes through its role in RNA demethylation. Alterations in these processes may affect leptin's metabolic effects, including glucose homeostasis and lipid metabolism [97,98].
Table 3 highlights some important genes which interact with leptin gene and influence obesity, energy balance and interfere with other important parameters[99-111].
| Sr. no. | Gene | Expression site | Functional pathway | Activation effects | Deletion effects | Reference |
|---|---|---|---|---|---|---|
| 1 | Leptin receptor | Hypothalamic neurons, various peripheral tissues | JAK2/STAT3 signaling pathway | Mediates leptin signaling, regulating appetite and energy expenditure | Obesity, hyperphagia, reduced energy expenditure | [99] |
| 2 | STAT3 | Widely expressed, including in leptin receptor-expressing neurons | JAK2/STAT3 signaling pathway | Transduces leptin signals to regulate gene expression related to energy balance | Obesity, normal fertility, increased linear growth | [100] |
| 3 | POMC | Hypothalamic neurons | Melanocortin pathway | Produces α-MSH, promoting satiety and reducing food intake | Hyperphagia, obesity | [101] |
| 4 | NPY | Hypothalamic neurons | NPY signaling pathway | Stimulates appetite and food intake | Reduced feeding behavior, leanness | [102] |
| 5 | SOCS3 | Various tissues, including hypothalamic neurons | Negative regulator of JAK2/STAT3 pathway | Inhibits leptin signaling, modulating energy balance | Enhanced leptin sensitivity, resistance to diet-induced obesity | [103] |
| 6 | AMPK | Widely expressed, including in hypothalamic neurons | AMPK signaling pathway | Inhibits food intake and regulates energy expenditure in response to leptin | Increased food intake, decreased energy expenditure | [104] |
| 7 | AgRP | Hypothalamic neurons | Melanocortin pathway antagonist | Increases food intake by inhibiting melanocortin receptors | Reduced food intake, leanness | [105] |
| 8 | PI3K | Widely expressed, including in hypothalamic neurons | PI3K/Akt signaling pathway | Mediates leptin's effects on glucose homeostasis and energy balance | Impaired glucose metabolism, altered energy homeostasis | [106] |
| 9 | PPARγ | Adipocytes, macrophages, muscle cells | Lipid metabolism and glucose homeostasis pathway | Promotes adipogenesis and improves insulin sensitivity | Insulin resistance, reduced fat storage, increased inflammation | [107] |
| 10 | MC4R | Hypothalamic neurons | Melanocortin signaling pathway | Reduces food intake, increases energy expenditure | Hyperphagia, obesity | [108] |
| 11 | UCP2 | Mitochondria in various tissues | Mitochondrial uncoupling and thermogenesis pathway | Regulates energy expenditure, reduces ROS | Reduced thermogenesis, increased oxidative stress | [109] |
| 12 | GCKR | Liver, pancreas | Glucose metabolism pathway | Modulates glucokinase activity, impacting glucose and lipid metabolism | Impaired glucose homeostasis, altered lipid metabolism | [110] |
| 13 | FTO | Various tissues including brain | RNA demethylation and energy homeostasis pathway | Affects energy intake and metabolism, linked to obesity | Reduced growth, lean phenotype | [111] |
Table 3: Summary of Some Important Genes Which Interact With Leptin
Conclusion
As is evident from above narrations, that role of leptin in human is varied however no specific trend can be established. Often population studies have seen contrasting results. It is possible that the interplay of genes may be playing a significant role. As gene-gene interaction at various levels from epistasis to higher order complex QTNs are well known to exist, there is still lack of established connections on genetic levels. Similarly, the molecular cascades leading to a particular effect are still being studied. This review can conclude that the role of leptin gene the gene product hormone and cytokine leptin and its effect in human body is very important. However clinical and non-clinical studies are required for establishing the clear signalling, molecular and physiological mechanisms to thoroughly understand the leptin cascade. The interplay of various molecules, neurons and leptin gene/hormone will still remain an interesting research topic in near future[112].
Acknowledgement:
This work is not funded or sponsored by any agency.
Conflict of interests:
The authors declared no conflict of interests.
References
- MacReady N. Leptin: 20 years later. Lancet Diabetes Endocrinol 2014;2(11):864.
[Crossref] [Google Scholar] [PubMed]
- Friedman JM, Halaas JL. Leptin and the regulation of body weight in mammals. Nature 1998;395(6704):763-70.
[Crossref] [Google Scholar] [PubMed]
- Maffei Á, Halaas J, Ravussin E, Pratley RE, Lee GH, Zhang Y, et al. Leptin levels in human and rodent: Measurement of plasma leptin and ob RNA in obese and weight-reduced subjects. Nat Med 1995;1(11):1155-61.
[Crossref] [Google Scholar] [PubMed]
- Gene. Bethesda (MD): National Library of Medicine (US), National Center for Biotechnology Information; 2004.
- Sweeney G. Cardiovascular effects of leptin. Nat Rev Cardiol 2010;7(1):22-9.
[Crossref] [Google Scholar] [PubMed]
- Beltowski J. Leptin and atherosclerosis. Atherosclerosis 2006;189(1):47-60.
[Crossref] [Google Scholar] [PubMed]
- Eguchi M, Liu Y, Shin EJ, Sweeney G. Leptin protects H9c2 rat cardiomyocytes from H2O2‐induced apoptosis. FEBS J 2008;275(12):3136-44.
[Crossref] [Google Scholar] [PubMed]
- Shin EJ, Schram K, Zheng XL, Sweeney G. Leptin attenuates hypoxia/reoxygenation‐induced activation of the intrinsic pathway of apoptosis in rat H9c2 cells. J Cell Physiol 2009;221(2):490-7.
[Crossref] [Google Scholar] [PubMed]
- Wu TJ, Wu DA, Hsu BG. Serum leptin level is positively correlated with aortic stiffness in patients with type 2 diabetes mellitus. Front Biosci (Landmark Ed) 2023;28(6):128.
[Crossref] [Google Scholar] [PubMed]
- Palanivel R, Eguchi M, Shuralyova I, Coe I, Sweeney G. Distinct effects of short-and long-term leptin treatment on glucose and fatty acid uptake and metabolism in HL-1 cardiomyocytes. Metabolism 2006;55(8):1067-75.
[Crossref] [Google Scholar] [PubMed]
- Lopaschuk GD, Folmes CD, Stanley WC. Cardiac energy metabolism in obesity. Circ Res 2007;101(4):335-47.
[Crossref] [Google Scholar] [PubMed]
- Xu FP, Chen MS, Wang YZ, Yi Q, Lin SB, Chen AF, et al. Leptin induces hypertrophy via endothelin-1–reactive oxygen species pathway in cultured neonatal rat cardiomyocytes. Circulation 2004;110(10):1269-75.
[Crossref] [Google Scholar] [PubMed]
- Zeidan A, Javadov S, Chakrabarti S, Karmazyn M. Leptin-induced cardiomyocyte hypertrophy involves selective caveolae and RhoA/ROCK-dependent p38 MAPK translocation to nuclei. Cardiovasc Res 2008;77(1):64-72.
[Crossref] [Google Scholar] [PubMed]
- Batra A, Pietsch J, Fedke I, Glauben R, Okur B, Stroh T, et al. Leptin-dependent toll-like receptor expression and responsiveness in preadipocytes and adipocytes. Am J Pathol 2007;170(6):1931-41.
[Crossref] [Google Scholar] [PubMed]
- Konstantinidis D, Paletas K, Koliakos G, Kaloyianni M. Signaling components involved in leptin-induced amplification of the atherosclerosis-related properties of human monocytes. J Vasc Res 2009;46(3):199-208.
[Crossref] [Google Scholar] [PubMed]
- Korda M, Kubant R, Patton S, Malinski T. Leptin-induced endothelial dysfunction in obesity. Am J Physiol Heart Circ Physiol 2008;295(4):1514-21.
[Crossref] [Google Scholar] [PubMed]
- Nakata M, Yada T, Soejima N, Maruyama I. Leptin promotes aggregation of human platelets via the long form of its receptor. Diabetes 1999;48(2):426-9.
[Crossref] [Google Scholar] [PubMed]
- Dellas C, Schafer K, Rohm I, Lankeit M, Ellrott T, Faustin V, et al. Absence of leptin resistance in platelets from morbidly obese individuals may contribute to the increased thrombosis risk in obesity. Thromb Haemost 2008;100(12):1123-9.
[Google Scholar] [PubMed]
- Elbatarny HS, Maurice DH. Leptin-mediated activation of human platelets: Involvement of a leptin receptor and phosphodiesterase 3A-containing cellular signaling complex. Am J Physiol Endocrinol Metab 2005;289(4):E695-702.
[Crossref] [Google Scholar] [PubMed]
- Gairolla J, Kler R, Modi M, Khurana D. Leptin and adiponectin: Pathophysiological role and possible therapeutic target of inflammation in ischemic stroke. Rev Neurosci 2017;28(3):295-306.
[Crossref] [Google Scholar] [PubMed]
- Stanek A, Grygiel-Górniak B, Brożyna-Tkaczyk K, Myśliński W, Cholewka A, Zolghadri S. The influence of dietary interventions on arterial stiffness in overweight and obese subjects. Nutrients 2023;15(6):1440.
[Crossref] [Google Scholar] [PubMed]
- Ollberding NJ, Kim Y, Shvetsov YB, Wilkens LR, Franke AA, Cooney RV, et al. Prediagnostic leptin, adiponectin, C-reactive protein, and the risk of postmenopausal breast cancer. Cancer Prev Res 2013;6(3):188-95.
[Crossref] [Google Scholar] [PubMed]
- Garofalo C, Koda M, Cascio S, Sulkowska M, Kanczuga-Koda L, Golaszewska J, et al. Increased expression of leptin and the leptin receptor as a marker of breast cancer progression: Possible role of obesity-related stimuli. Clin Cancer Res 2006;12(5):1447-53.
[Crossref] [Google Scholar] [PubMed]
- Mhaidat NM, Alzoubi KH, Kubas MA, Banihani MN, Hamdan N, Al-Jaberi TM. High levels of leptin and non-high molecular weight-adiponectin in patients with colorectal cancer: Association with chemotherapy and common genetic polymorphisms. Biomed Rep 2021;14(1):13.
[Google Scholar] [PubMed]
- Akinci M, Kosova F, Cetin B, Aslan S, Ari Z, Cetin A. Leptin levels in thyroid cancer. Asian J Surg 2009;32(4):216-23.
[Crossref] [Google Scholar] [PubMed]
- Kamel HF, Nassir AM, Al Refai AA. Assessment of expression levels of leptin and leptin receptor as potential biomarkers for risk of prostate cancer development and aggressiveness. Cancer Med 2020;9(15):5687-96.
[Crossref] [Google Scholar] [PubMed]
- Arisan ED, Arisan S, Atis G, Palavan-Unsal N, Ergenekon E. Serum adipocytokine levels in prostate cancer patients. Urol Int 2009;82(2):203-8.
[Crossref] [Google Scholar] [PubMed]
- Aref S, Ibrahim L, Azmy E, Al Ashary R. Impact of serum adiponectin and leptin levels in acute leukemia. Hematology 2013;18(4):198-203.
[Crossref] [Google Scholar] [PubMed]
- Yu W, Cao DD, Li QB, Mei HL, Hu Y, Guo T. Adipocytes secreted leptin is a pro-tumor factor for survival of multiple myeloma under chemotherapy. Oncotarget. 2016;7(52):86075.
[Crossref] [Google Scholar] [PubMed]
- Riolfi M, Ferla R, Valle LD, Piña‐Oviedo S, Scolaro L, Micciolo R, et al. Leptin and its receptor are overexpressed in brain tumors and correlate with the degree of malignancy. Brain Pathol 2010;20(2):481-9.
[Crossref] [Google Scholar] [PubMed]
- Zhang L, Yuan Q, Li M, Chai D, Deng W, Wang W. The association of leptin and adiponectin with hepatocellular carcinoma risk and prognosis: A combination of traditional, survival, and dose-response meta-analysis. BMC Cancer 2020;20:1-20.
[Crossref] [Google Scholar] [PubMed]
- Wei X, Liu Y, Gong C, Ji T, Zhou X, Zhang T, et al. Targeting leptin as a therapeutic strategy against ovarian cancer peritoneal metastasis. Anticancer Agents Med Chem 2017;17(8):1093-1101.
[Crossref] [Google Scholar] [PubMed]
- Zhang Y, Liu L, Li C, Ai H. Correlation analysis between the expressions of leptin and its receptor (ObR) and clinicopathology in endometrial cancer. Cancer Biomarkers 2014;14(5):353-9.
[Crossref] [Google Scholar] [PubMed]
- Pezeshki M, Hosseini SM, Ansari J, Ahmadi A. Investigating the importance of EGFR (-216G/T), Exo1 (K589E) and LEP (-2548G/A) gene polymorphisms with risk of lung cancer as potential diagnostic biomarker in Iranian population. Egypt J Med Hum Genet 2023;23;24(1):55.
- Maculewicz E, Leonska Duniec A, Mastalerz A, Szarska E, Garbacz A, Lepionka T, Lakomy R, Anyzewska A, Bertrandt J. The influence of FTO, FABP2, LEP, leptin receptor, and MC4R genes on obesity parameters in physically active Caucasian men. Int J Environ Res Public Health 2022;19(10):6030.
[Crossref] [Google Scholar] [PubMed]
- Sabi EM, Bin Dahman LS, Mohammed AK, Sumaily KM, Al-Daghri NM. -2548G> A LEP polymorphism is positively associated with increased leptin and glucose levels in obese Saudi patients irrespective of blood pressure status. Medicina 2022;58(3):346.
- Chen S, Ke Y, Chen W, Wu S, Zhuang X, Lin Q, et al. Association of the LEP gene with immune infiltration as a diagnostic biomarker in preeclampsia. Front Mol Biosci 2023;10:1209144.
[Crossref] [Google Scholar] [PubMed]
- Sayad S, Dastgheib SA, Farbod M, Asadian F, Karimi-Zarchi M, Salari S, et al. Association of PON1, LEP and leptin receptor polymorphisms with susceptibility to breast cancer: A meta-analysis. Asian Pac J Cancer Prev 2021;22(8):2323.
[Crossref] [Google Scholar] [PubMed]
- Yu D, Yu Z, Sun Q, Sun L, Li H, Song J, et al. Effects of body fat on the associations of high-molecular-weight adiponectin, leptin and soluble leptin receptor with metabolic syndrome in Chinese. PloS One 2011;6(2):e16818.
[Crossref] [Google Scholar] [PubMed]
- Dasgupta S, Salman M, Siddalingaiah LB, Lakshmi GL, Xaviour D, Sreenath J. Genetic variants in leptin: Determinants of obesity and leptin levels in South Indian population. Adipocyte 2015;4(2):135-40.
[Crossref] [Google Scholar] [PubMed]
- Afifi AE, Shaat RM, Gharbia OM, Elhanafy M, Hasan AS. Role of serum leptin levels and leptin receptor gene polymorphisms in systemic lupus erythematosus. Clin Rheumatol 2020;39:3465-72.
[Crossref] [Google Scholar] [PubMed]
- Zadjali F, Al Farsi B, Zadjali R, Bayoumi RA, Al Barwani S, Al-Yahyaee S. Gender-specific reference range for serum leptin in Omani population. Oman Med J 2023;38(5):e545.
[Google Scholar] [PubMed]
- Saxton RA, Caveney NA, Moya-Garzon MD, Householder KD, Rodriguez GE, Burdsall KA, et al. Structural insights into the mechanism of leptin receptor activation. Nat Commun 2023;14(1):1797.
- Park KS, Shin HD, Park BL, Cheong HS, Cho YM, Lee HK, et al. Polymorphisms in the leptin receptor-putative association with obesity and T2DM. J Hum Genet 2006;51(2):85-91.
[Crossref] [Google Scholar] [PubMed]
- Tan HL, Yin L, Tan Y, Ivanov J, Plucinska K, Ilanges A, et al. Leptin-activated hypothalamic BNC2 neurons acutely suppress food intake. Nature 2024;636(8041):198-205.
[Crossref] [Google Scholar] [PubMed]
- Xu J, Bartolome CL, Low CS, Yi X, Chien CH, Wang P, Kong D. Genetic identification of leptin neural circuits in energy and glucose homeostases. Nature 2018;556(7702):505-9.
[Crossref] [Google Scholar] [PubMed]
- Vaisse C, Halaas JL, Horvath CM, Darnell Jr JE, Stoffel M, Friedman JM. Leptin activation of Stat3 in the hypothalamus of wild–type and ob/ob mice but not db/db mice. Nat Genet 1996;14(1):95-7.
[Crossref] [Google Scholar] [PubMed]
- Buettner C, Pocai A, Muse ED, Etgen AM, Myers MG, Rossetti L. Critical role of STAT3 in leptin's metabolic actions. Cell Metabol 2006;4(1):49-60.
[Crossref] [Google Scholar] [PubMed]
- Bates SH, Stearns WH, Dundon TA, Schubert M, Tso AW, Wang Y, et al. STAT3 signalling is required for leptin regulation of energy balance but not reproduction. Nature 2003;421(6925):856-9.
[Crossref] [Google Scholar] [PubMed]
- Chang CC, Wu MJ, Yang JY, Camarillo IG, Chang CJ. Leptin–STAT3–G9a signalling promotes obesity-mediated breast cancer progression. Cancer Res 2015;75(11):2375-86.
[Crossref] [Google Scholar] [PubMed]
- Williams KW, Scott MM, Elmquist JK. Modulation of the central melanocortin system by leptin, insulin, and serotonin: Co-ordinated actions in a dispersed neuronal network. Eur J Pharmacol 2011;660(1):2-12.
[Crossref] [Google Scholar] [PubMed]
- Müller TD, Blüher M, Tschöp MH, DiMarchi RD. Anti-obesity drug discovery: Advances and challenges. Nat Rev Drug Discov 2022;21(3):201-23.
[Crossref] [Google Scholar] [PubMed]
- Lee DK, Jeong JH, Chun SK, Chua Jr S, Jo YH. Interplay between glucose and leptin signalling determines the strength of GABAergic synapses at POMC neurons. Nat Commun 2015;6(1):6618.
[Crossref] [Google Scholar] [PubMed]
- Belgardt BF, Okamura T, Brüning JC. Hormone and glucose signalling in POMC and AgRP neurons. J Physiol 2009;587(22):5305-14.
- Caron A, Dungan Lemko HM, Castorena CM, Fujikawa T, Lee S, Lord CC, et al. POMC neurons expressing leptin receptors coordinate metabolic responses to fasting via suppression of leptin levels. Elife 2018;7:e33710.
[Crossref] [Google Scholar] [PubMed]
- Ebert MS, Sharp PA. Roles for microRNAs in conferring robustness to biological processes. Cell 2012;149(3):515-24.
[Crossref] [Google Scholar] [PubMed]
- Schneeberger M, Gomis R, Claret M. Hypothalamic and brainstem neuronal circuits controlling homeostatic energy balance. J Endocrinol 2014;220(2):T25-46.
[Crossref] [Google Scholar] [PubMed]
- Myers MG, Leibel RL, Seeley RJ, Schwartz MW. Obesity and leptin resistance: Distinguishing cause from effect. Trends Endocrinol Metabol 2010;21(11):643-51.
[Crossref] [Google Scholar] [PubMed]
- Morrison CD, Morton GJ, Niswender KD, Gelling RW, Schwartz MW. Leptin inhibits hypothalamic Npy and Agrp gene expression via a mechanism that requires phosphatidylinositol 3-OH-kinase signaling. Am J Physiol Endocrinol Metabol 2005;289(6):E1051-7.
[Crossref] [Google Scholar] [PubMed]
- Baver SB, Hope K, Guyot S, Bjørbaek C, Kaczorowski C, O'Connell KM. Leptin modulates the intrinsic excitability of AgRP/NPY neurons in the arcuate nucleus of the hypothalamus. J Neurosci 2014;34(16):5486-96.
[Crossref] [Google Scholar] [PubMed]
- Murphy BA, Fioramonti X, Jochnowitz N, Fakira K, Gagen K, Contie S, et al. Fasting enhances the response of arcuate neuropeptide Y-glucose-inhibited neurons to decreased extracellular glucose. Am J Physiol Cell Physiol 2009;296(4):C746-56.
[Crossref] [Google Scholar] [PubMed]
- Heyward FD, Liu N, Jacobs C, Machado NL, Ivison R, Uner A, et al. AgRP neuron cis-regulatory analysis across hunger states reveals that IRF3 mediates leptin’s acute effects. Nat Commun 2024;15(1):4646.
- Wang L, Shao YY, Ballock RT. Leptin antagonizes peroxisome proliferator‐activated receptor‐γ signaling in growth plate chondrocytes. PPAR Res 2012;2012(1):756198.
[Crossref] [Google Scholar] [PubMed]
- Zhang Y, Dallner OS, Nakadai T, Fayzikhodjaeva G, Lu YH, Lazar MA, et al. A noncanonical PPARγ/RXRα-binding sequence regulates leptin expression in response to changes in adipose tissue mass. Proc Natl Acad Sci 2018;115(26):E6039-47.
[Crossref] [Google Scholar] [PubMed]
- Matsusue K, Haluzik M, Lambert G, Yim SH, Gavrilova O, Ward JM, et al. Liver-specific disruption of PPARγ in leptin-deficient mice improves fatty liver but aggravates diabetic phenotypes. J Clin Invest 2003;111(5):737-47.
[Crossref] [Google Scholar] [PubMed]
- Schwartz MW, Woods SC, Porte Jr D, Seeley RJ, Baskin DG. Central nervous system control of food intake. Nature 2000;404(6778):661-71.
[Crossref] [Google Scholar] [PubMed]
- Farooqi IS, Keogh JM, Yeo GS, Lank EJ, Cheetham T, O'Rahilly S. Clinical spectrum of obesity and mutations in the melanocortin 4 receptor gene. New Engl J Med 2003;348(12):1085-95.
[Crossref] [Google Scholar] [PubMed]
- Balthasar N, Dalgaard LT, Lee CE, Yu J, Funahashi H, Williams T, et al. Divergence of melanocortin pathways in the control of food intake and energy expenditure. Cell 2005;123(3):493-505.
[Crossref] [Google Scholar] [PubMed]
- Ho PW, Liu HF, Ho JW, Zhang WY, Chu AC, Kwok KH, et al. Mitochondrial uncoupling protein-2 (UCP2) mediates leptin protection against MPP+ toxicity in neuronal cells. Neurotox Res 2010;17:332-43.
[Crossref] [Google Scholar] [PubMed]
- Ceddia RB, William Jr WN, Lima FB, Flandin P, Curi R, Giacobino JP. Leptin stimulates uncoupling protein‐2 mRNA expression and Krebs cycle activity and inhibits lipid synthesis in isolated rat white adipocytes. Eur J Biochem 2000;267(19):5952-8.
[Crossref] [Google Scholar] [PubMed]
- Gnanalingham MG, Mostyn A, Webb R, Keisler DH, Raver N, Alves-Guerra MC, et al. Differential effects of leptin administration on the abundance of UCP2 and glucocorticoid action during neonatal development. Am J Physiol Endocrinol Metabol 2005;289(6):E1093-100.
[Crossref] [Google Scholar] [PubMed]
- Henry BA, Andrews ZB, Rao A, Clarke IJ. Central leptin activates mitochondrial function and increases heat production in skeletal muscle. Endocrinology 2011;152(7):2609-18.
[Crossref] [Google Scholar] [PubMed]
- Xiao XQ, Grove KL, Grayson BE, Smith MS. Inhibition of uncoupling protein expression during lactation: Role of leptin. Endocrinology 2004;145(2):830-8.
[Crossref] [Google Scholar] [PubMed]
- Loos RJ, Lindgren CM, Li S, Wheeler E, Zhao JH, Prokopenko I, et al. Common variants near MC4R are associated with fat mass, weight and risk of obesity. Nat Genet 2008;40(6):768-75.
[Crossref] [Google Scholar] [PubMed]
- Mahajan A, Taliun D, Thurner M, Robertson NR, Torres JM, Rayner NW, et al. Fine-mapping type 2 diabetes loci to single-variant resolution using high-density imputation and islet-specific epigenome maps. Nat Genet 2018;50(11):1505-13.
[Crossref] [Google Scholar] [PubMed]
- Saxena R, Hivert MF, Langenberg C, Tanaka T, Pankow JS, Vollenweider P, et al. Genetic variation in GIPR influences the glucose and insulin responses to an oral glucose challenge. Nat Genet 2010;42(2):142-8.
[Crossref] [Google Scholar] [PubMed]
- Scott RA, Lagou V, Welch RP, Wheeler E, Montasser ME, Luan JA, et al. Large-scale association analyses identify new loci influencing glycemic traits and provide insight into the underlying biological pathways. Nat Genet 2012;44(9):991-1005.
[Crossref] [Google Scholar] [PubMed]
- Rippe C, Leshan RL, Petersen N. Severe obesity and a homozygous loss-of-function mutation in the human leptin receptor gene. Eur J Endocrinol 2015;172(5):E199-E201.
- Leong A, Porneala B, Dupuis J, Florez JC, Meigs JB. Type 2 diabetes genetic predisposition, obesity, and all-cause mortality risk in the US: A multiethnic analysis. Diabetes Care 2016;39(4):539-46.
[Crossref] [Google Scholar] [PubMed]
- Boden G, Chen XI, Ruiz J, White JV, Rossetti L. Mechanisms of fatty acid-induced inhibition of glucose uptake. J Clin Invest 1997;100(1):258-63.
[Crossref] [Google Scholar] [PubMed]
- Beale EG, Harvey BJ, Forest C. PCK1 and PCK2 as candidate diabetes and obesity genes. Cell Biochem Biophys 2007;48(2-3):89-95.
[Crossref] [Google Scholar] [PubMed]
- Unger RH, Orci L. Diseases of liporegulation: New perspective on obesity and related disorders. FASEB J 2001;15(2):312-21.
[Crossref] [Google Scholar] [PubMed]
- Kovacs P, Stumvoll M. Fatty acids and insulin resistance in muscle and liver. Best Pract Res Clin Endocrinol Metab 2005;19(4):625-35.
[Crossref] [Google Scholar] [PubMed]
- Porte Jr D, Baskin DG, Schwartz MW. Leptin and insulin action in the central nervous system. Nutr Rev 2002;60(suppl_10):S20-9.
[Crossref] [Google Scholar] [PubMed]
- Cote-Sierra J, Foucras G, Guo L, Chiodetti L, Young HA, Hu-Li J, et al. Interleukin 2 plays a central role in Th2 differentiation. Proc Natl Acad Sci U S A 2004;101(11):3880-5.
[Crossref] [Google Scholar] [PubMed]
- Frayling TM, Timpson NJ, Weedon MN, Zeggini E, Freathy RM, Lindgren CM, et al. A common variant in the FTO gene is associated with body mass index and predisposes to childhood and adult obesity. Science 2007;316(5826):889-94.
[Crossref] [Google Scholar] [PubMed]
- Scuteri A, Sanna S, Chen WM, Uda M, Albai G, Strait J, et al. Genome-wide association scan shows genetic variants in the FTO gene are associated with obesity-related traits. PLoS Genet 2007;3(7):e115.
[Crossref] [Google Scholar] [PubMed]
- Wardle J, Carnell S, Haworth CM, Farooqi IS, O'Rahilly S, Plomin R. Obesity associated genetic variation in FTO is associated with diminished satiety. J Clin Endocrinol Metab 2008;93(9):3640-3.
[Crossref] [Google Scholar] [PubMed]
- Sanna S, Jackson AU, Nagaraja R, Willer CJ, Chen WM, Bonnycastle LL, et al. Common variants in the GDF5-UQCC region are associated with variation in human height. Nat Genet 2008;40(2):198-203.
[Crossref] [Google Scholar] [PubMed]
- Church C, Moir L, McMurray F, Girard C, Banks GT, Teboul L, et al. Overexpression of Fto leads to increased food intake and results in obesity. Nat Genet 2010;42(12):1086-92.
[Crossref] [Google Scholar] [PubMed]
- Seed M, Agius R. Further corroboration of the asthmagenicity of 5-aminosalicylic acid. Occup Environ Med 2011;68(5):386-95. [Crossref]
[Google Scholar] [PubMed]
- Melhorn SJ, Askren MK, Chung WK, Kratz M, Bosch TA, Tyagi V, et al. FTO genotype impacts food intake and corticolimbic activation. Am J Clin Nutr 2018;107(2):145-54.
[Crossref] [Google Scholar] [PubMed]
- Zhao J, Bradfield JP, Zhang H, Sleiman PM, Kim CE, Glessner JT, et al. Role of BMI‐associated loci identified in GWAS meta‐analyses in the context of common childhood obesity in European Americans. Obesity 2011;19(12):2436-9.
[Crossref] [Google Scholar] [PubMed]
- Do R, Bailey SD, Desbiens K, Belisle A, Montpetit A, Bouchard C, et al. Genetic variants of FTO influence adiposity, insulin sensitivity, leptin levels, and resting metabolic rate in the Quebec family study. Diabetes 2008;57(4):1147-50.
[Crossref] [Google Scholar] [PubMed]
- Speakman JR, Rance KA, Johnstone AM. Polymorphisms of the FTO gene are associated with variation in energy intake, but not energy expenditure. Obesity 2008;16(8):1961-5.
[Crossref] [Google Scholar] [PubMed]
- Zhao X, Yang Y, Sun BF, Shi Y, Yang X, Xiao W, et al. FTO-dependent demethylation of N6-methyladenosine regulates mRNA splicing and is required for adipogenesis. Cell Res 2017;27(6):616-32.
[Crossref] [Google Scholar] [PubMed]
- Fischer J, Koch L, Emmerling C, Vierkotten J, Peters T, Brüning JC, et al. Inactivation of the Fto gene protects from obesity. Nature 2009;458(7240):894-8.
[Crossref] [Google Scholar] [PubMed]
- Tartaglia LA, Dembski M, Weng X, Deng N, Culpepper J, Devos R, et al. Identification and expression cloning of a leptin receptor, OB-R. Cell 1995;83(7):1263-71.
[Crossref] [Google Scholar] [PubMed]
- Piper ML, Unger EK, Myers Jr MG, Xu AW. Specific physiological roles for signal transducer and activator of transcription 3 in leptin receptor-expressing neurons. Mol Endocrinol 2008;22(3):751-9.
[Crossref] [Google Scholar] [PubMed]
- Elmquist JK, Elias CF, Saper CB. From lesions to leptin: Hypothalamic control of food intake and body weight. Neuron 1999;22(2):221-32.
[Crossref] [Google Scholar] [PubMed]
- Elias CF, Aschkenasi C, Lee C, Kelly J, Ahima RS, Bjorbæk C, et al. Leptin differentially regulates NPY and POMC neurons projecting to the lateral hypothalamic area. Neuron 1999;23(4):775-86.
[Crossref] [Google Scholar] [PubMed]
- Bjorbæk C, Lavery HJ, Bates SH, Olson RK, Davis SM, Flier JS, et al. SOCS3 mediates feedback inhibition of the leptin receptor via Tyr985. J Biol Chem 1998;273(49):32686-90.
[Crossref] [Google Scholar] [PubMed]
- Minokoshi Y, Alquier T, Furukawa N, Kim YB, Lee A, Xue B, et al. AMP-kinase regulates food intake by responding to hormonal and nutrient signals in the hypothalamus. Nature 2004;428(6982):569-74.
[Crossref] [Google Scholar] [PubMed]
- Fekete C, Légrádi G, Mihály E, Huang QH, Tatro JB, Rand WM, et al. α-Melanocyte-stimulating hormone is contained in nerve terminals innervating thyrotropin-releasing hormone-synthesizing neurons in the hypothalamic paraventricular nucleus and prevents fasting-induced suppression of prothyrotropin-releasing hormone gene expression. J Neurosci 2000;20(4):1550-8.
[Crossref] [Google Scholar] [PubMed]
- Niswender KD, Schwartz MW. Insulin and leptin revisited: Adiposity signals with overlapping physiological and intracellular signaling capabilities. Front Neuroendocrinol 2003;24(1):1-10.
[Crossref] [Google Scholar] [PubMed]
- Rosen ED, Sarraf P, Troy AE, Bradwin G, Moore K, Milstone DS, et al. PPAR gamma is required for the differentiation of adipose tissue in vivo and in vitro. Genes Dev 2002;16(17):2159-71.
[Crossref] [Google Scholar] [PubMed]
- Huszar D, Lynch CA, Fairchild-Huntress V, Dunmore JH, Fang Q, Berkemeier LR, et al. Targeted disruption of the melanocortin-4 receptor results in obesity in mice. Cell 1997;88(1):131-41.
[Crossref] [Google Scholar] [PubMed]
- Fleury C, Neverova M, Collins S, Raimbault S, Champigny O, Levi-Meyrueis C, et al. Uncoupling protein-2: A novel gene linked to obesity and hyperinsulinemia. Nat Genet 1997;15(3):269-72.
[Crossref] [Google Scholar] [PubMed]
- Beer NL. The role of GCKR in metabolic traits. Diabetes 2009;58(10):2441-9.
- Church C, Moir L, McMurray F, Girard C, Banks GT, Teboul L, et al. Overexpression of Fto leads to increased food intake and results in obesity. Nat Genet 2010;42(12):1086-92.
[Crossref] [Google Scholar] [PubMed]
- Phillips PC. Epistasis-the essential role of gene interactions in the structure and evolution of genetic systems. Nat Rev Genet 2008;9(11):855-67.
[Crossref] [Google Scholar] [PubMed]
- Tan HL, Yin L, Tan Y, Ivanov J, Plucinska K, Ilanges A, et al. Leptin-activated hypothalamic BNC2 neurons acutely suppress food intake. Nature 2024;636(8041):198-205.
[Crossref] [Google Scholar] [PubMed]