- *Corresponding Author:
- Ahmad Salawi
Department of Pharmaceutics, Faculty of Pharmacy, Sohag University, Sohag 82524, Egypt
E-mail: asalawi@jazanu.edu.sa
| Date of Received | 27 December 2024 |
| Date of Revision | 18 January 2025 |
| Date of Accepted | 27 February 2025 |
| Indian J Pharm Sci 2024;87(1):22-33 |
This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms
Abstract
The current experiment describes hematological restrictions such as blood cells, packed cell volume, and the percentage of mean corpuscles value. It also evaluates the influence of drugs on critical body organs such as the liver, kidney, and heart by analyzing serum biochemical data. Our main goal is to administer weight-dependent doses of anti-inflammatory pharmaceuticals to birds and study the effects of those medications. Five groups of roughly fifty common quails were acquired. Four of the five groups were labeled as experimental, while one was classified as the control group. Subsequently, the experimental groups were divided into four groups, with each group being assigned a trial of 5, 10, 15, or 20 d. Dose of non-steroid anti-inflammatory medications were administrated according to weight of bird. Administrative dose of Famotidine and Naproxen was 0.02 mg/g and 0.55 mg/g respectively, 2X a day. Clinical signs included hypersomnia, reduced food and drink intake, gout, dehydration, accumulation of nitrogen products in the body, and restlessness. There was also significant swelling in the foot. Watery eyes later on, accompanied by swollen eyes, fever, weight loss, and beak cutting in all birds that received the dose, but no similar signs in birds that were not treated. A certain percentage of birds treated with Naproxen died. Administration of Famotidine during summer trial showed significant increase in total white blood cell (p=0.003), uric acid (p=0.02), creatine kinase myocardial band level (p=0.014), mean corpuscular volume level (p=0.006) ASAT (p=0.007), aminotransferase (p=0.029), albumin (p=0.016). While during winter trial uric acid (p=0.000), aminotransferase (p=0.041), mean corpuscular volume (p=0.014), albumin level (p=0.006 ) uric acid (p=0.035) and while administration of naproxen showed cholesterol level (p=0.003), aminotransferase (p=0.008), mean corpuscular volume (p=0.003), total white blood count (p=0.008), creatine kinase myocardial band (p=0.014), albumin (p=0.011) and aminotransferase (p=0.001).While during winter mean corpuscular volume (p=0.004), white blood cell count, (p=0.016), cholesterol (p=0.011), albumin (p=0.007), total protein (p=0.000), total white blood cell count (p=0.002), albumin level (p=0.014) and aminotransferase (p=0.036).
Keywords
Hematology, inflammation, bumble-foot disease, tumor, stomach
The close anatomical and physiological similarity of common quails to domestic chickens, combined with their small size, make them perfect models for poultry research[1]. The primary benefit of utilizing this species as a model animal for birds is its modest size. Males weigh 100-120 g at maturity, while females weigh 120-150 g[2]. Their high egg production rate (up to 300 per year) and short generation interval make them easy to manage in a lab setting. Due to their sensitivity to environmental changes, quails are housed in ideal conditions[3].
The majority of Non-Steroid Anti-Inflammatory Medications (NSAIDs) have analgesic[4], antipyretic[5], and anti-inflammatory properties[6]. To treat inflammation in the musculoskeletal system and other tissues, NSAIDs are frequently utilized in veterinary medicine[7], colitis[8], joint pain[9], endotoxin shock[10], trauma, or surgical pain[11]. In circumstances of trauma or infection, these medications are advised for use in avian medicine[12], heat stress, rapid death syndrome, broiler ascites, growth, or egg production[13], locomotion disturbances, and pain related to beak trimming[14]. Quail NSAIDs are used as an extra label and their dosage is derived from other species because there isn’t an approved NSAID for quails[15].
It is already commonly recognized that NSAIDs poison scavenging birds[16], three Gyps vulture species experienced major population collapses on the Indian subcontinent (about 90 %-98 % in Pakistan, India, and Nepal). This was caused by renal failure and visceral gout that developed from devouring corpses polluted with diclofenac[17]. Among the medications that are most commonly found in marine surface waters are NSAIDs[18], including estuaries, lagoons and coastal areas[19]. NSAIDs, including naproxen, diclofenac, ibuprofen, and nimesulide, have also been found in fish and aquatic invertebrates on a regular basis, which may indicate that they have spread to ichthyophagous and invertivorous animals[20].
More than 99 % of the vultures in the Indian subcontinent have disappeared as a result of veterinary use of the NSAID diclofenac[21]. The Gyps vulture was widely distributed before this medication was used for veterinary purposes in Bangladesh, India, Pakistan, and Nepal. Birds were thought to pose a menace to aircraft in the 1980s due to their enormous numbers[22]. In addition to aspirin, NSAIDs increase the risk of stroke and cardiac damage[23]. There is proof that naproxen has less negative effects than famotidine[24]. NSAIDs have been linked to an aggressive impact on the kidneys[25] and prolonged use results in chronic disease. NSAIDs, or nonsteroidal anti-inflammatory medications, have shown great promise in preventing skin, bladder, and colon cancer[26] colon adenoma development both preclinically and in clinical trials. Nevertheless, several NSAIDs increase the risk of cardiovascular events and induce gastrointestinal ulcers[27]. Other than aspirin, naproxen appears to be the most effective NSAID in reducing cardiovascular events[28,29].
Naproxen is utilized as a source of pain reliever for either head or muscle aches[30], teeth, joints, or as a result of menstruation[30]. It works by preventing our body from releasing certain natural chemicals that lead to inflammation. This medication is used to treat rheumatoid arthritis, tendonitis, kidney stones, migraines, osteoarthritis, gout, and menstrual cramps[31]. Researchers at fox chase cancer centre found that naproxen and no-naproxen reduce tumour formation in a type of mice with mutations that naturally induce colon tumours. These findings are based on animal experiments using the NSAID naproxen. Additionally, the information suggests using naproxen portions as a first step to initiate tumour growth[32].
Famotidine is a medication that is ingested. It reduces the amount of acid in the stomach by blocking the cells involved in acid production[33]. This medication is used to treat stomach and intestinal sores and prevent recurrence of previously healed intestinal ulcers[34,35].
According to a study, some veterinary professionals have been recommending painkillers to their patients for a long time[36]. However, new research has improved our understanding of these patient’s suffering and made it a crucial component of medical animal husbandry care. Our prior knowledge about pain in affected individuals has been compromised by misconceptions about the fundamentals of pain and its consequences, issues with determining its severity, poor decisions regarding crucial medical treatments, and a fundamental lack of scientific information about rare animals[37]. To effectively induce pain in enigmatic veterinary patients. These articles discuss interferences that represent current understanding on the acknowledgement of pain in veterinary patients[38].
The effects of injecting various histamine subtypes that are hostile to one another on food consumption were investigated in an experiment[39]. In the initial study, the animals received injections of famotidine, thioperamide, and subsequently chlorpheniramine. However, following the administration of each shot the pre-treatment of birds with chlorpheniramine maleate resulted in a considerably lower amount of food consumption (p< or =0.05). However, no effect was observed with famotidine or thioperamide[40]. Researchers have found that naproxen and nonaproxen are both useful treatments for colon and urinary bladder cancer, but not for the development of breast cancer in rats raised on methyl nitrosoureainduced mammary cancer. Between 550 and 400 ppm, no-naproxen and naproxen exhibited non-significant inhibitions (12 % and 24 %), respectively[41].
In an experiment, it was proposed that Japanese quails are utilized to produce meat and eggs for both humans and experimental animals[40]. Our goal is to distinguish between male and female quills based on a variety of serum biochemistry markers. In order to verify biochemical parameters, 16 w old quails-125 mature males and 151 females-were kept under observation. A statistical analysis revealed a negative impact on both sexes’ livers. No significant changes were observed in the levels of creatinine or uric acid[42].
Experiments have shown that many medications have a negative impact on the heartbeat, which is followed by an irregular heart rate. These patients experience vertigo; in some cases, unconsciousness may even be deadly. A case study demonstrated that famotidine is an effective medication for managing this condition[43].
Mahdavi et al.[44] evaluated the effects of peptide consumption after consuming meals and liquids under more extreme circumstances. For a month, the chickens were split up into experimental groups. There were sixteen groups in all. Pepcid was injected into them in half a millilitre based on their body weight. Throughout the day, food and water were provided to the chicks every hour. These findings suggest that the histamine receptor H2 is ineffective when it comes to the feed and water that broiler chickens consume in the consumption of feed. However, famotidine had no discernible impact on feed consumption when in hunger mode.
Van Dingenen, et al.[45] considered the antiinflammatory advantages of Hydrogen Sulphide (H2S) injection to rats with synovitis, researchers examined the influence of the H2S-releasing naproxen derivative ATB-346 in this experimental framework, in addition to the gastrointestinal safety of H2S-releasing NSAIDs. When measured three and 5 h after CGN (p<0.001), both naproxen and ATB-346 decreased pain and edema scores. Severe stomach damage was only produced by Naproxen. When rats with CGN-induced synovitis are given the ATB-346 structure, the H2S-releasing moiety does not diminish the anti-inflammatory properties of the parent drug[46]. The release of H2S was crucial in mitigating the adverse effects on the stomach, indicating that ATB-346 may be a viable therapeutic alternative to naproxen for the treatment of arthritis[47].
Downie and colleagues conducted studies on 41 arthritis patients by administering indomethacin and naproxen[48]. Both medications were administered twice daily: Once in the morning (250 mg of naproxen) and once at night (50 mg of indomethacin).
Twenty patients received naproxen, while twentyone received indomethacin injections. The course of treatment lasted for 1 mo. Both medications were successful in lessening shoulder pain, although movement was somewhat restricted because of muscular stiffness. Fourteen individuals from the naproxen group and sixteen unwell individuals from the indomethacin group reported headache and nausea as side effects[49].
The effectiveness of famotidine 20 mg, 40 mg, and zantac 150 mg was found in an experiment. This multimodal study included 224 patients diagnosed with duodenal ulcers by endoscopy from fifteen Italian organizations, and they were split into four treatment groups. For 8 w, the outcomes remained the same. However, famotidine treatment for patient’s pain later yielded a higher pain recovery rate than Zantac[50].
Materials and Methods
Approximately fifty female common quail were procured from Pakistani Chowk, D. G. Khan, with weights ranging from 50 g to 70 g. Upon acquisition, they were transported to the animal housing facilities of the zoology laboratory. The birds were accommodated in designated cages within a wellventilated and hygienic room. A diet comprising millet and fresh water was provided twice daily, and meticulous cage maintenance, including daily sheet replacement, was upheld throughout the study period. The avian subjects were subjected to laboratory conditions for a duration of 21 d, with five experimental sets, each comprising ten birds. Among these sets, four were designated as experimental groups denoted as A, while one served as the control group designated as B. The administration of NSAIDs was tailored to the weight of the birds. Each experimental group was further subdivided into four subgroups based on the duration of treatment: Subgroups treated for 5 d; subgroups treated for 10 d; subgroups treated for 15 d and subgroups treated for 20 d. The experiment was conducted twice, once during the summer (Trial A) and once during the winter (Trial B) seasons.
Dosage administration:
In this study, two NSAIDs were provided to birds based on their weight.
Famotidine is given at a dose of 0.02 mg/g body weight and Naproxen is given at a dose of 0.55 mg/g of body weight.
Each bird was divided into subgroups, with one group given famotidine and the others naproxen. Both medicines were given orally twice daily. Every 5 d, one subgroup of each medicine was euthanised for analysis.
Collection of blood:
I took blood samples by killing birds and immediately collecting the blood in glass tubes using Ethylenediaminetetraacetic Acid (EDTA), an anticoagulant agent. Blood divided into two test tubes.
For investigation of hematological variables, to research avian serology and calculation of Hematological variables.
In our investigation, hemolodical measures such as packed cell volume, leukocyte measurement, total leukocyte count, and corpuscular value were monitored to determine the effects of the NSAIDs utilized.
Identifying the serological characteristics:
Using sterile Eppendorf tubes, we centrifuged the mixture at 13 000 revolutions per minute for around 60 s in order to separate the blood cells from the serum. To be processed further, the extracted serum was divided into tubes with labels. In serology, cholesterol, Aminotransferase (AST), Alanine Aminotransferase (ALT), total proteins, albumin, creatinine, creatinine kinase, urea, and uric acid levels are measured.
Statistical analysis:
All data is presented in the form of mean and standard error of mean. The data were analysed using the statistical software package Statistical Package for the Social Sciences (SPSS) (version 23x86), and various parameters of blood and serum were compared between treated and untreated groups of birds using a two-sample t-test and one-way Analysis of Variance (ANOVA). The birds were maintained on trial for 5, 10, 15, and 20 d.
Results and Discussion
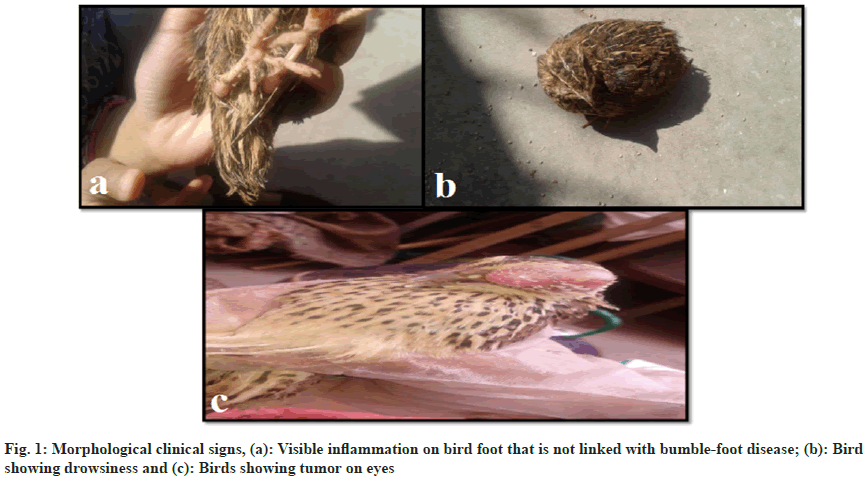
Random medical issues were observed after using two anti-inflammatory medicines, famtotidine 20 mg and naproxen 550 mg. The birds treated with Famotidine demonstrated restlessness, significant swelling in their feet (fig. 1a), decreased food and water intake, and hypersomnia (fig. 1b).
A bird developed an eye tumor (fig. 1c), whereas the untreated group did not exhibit any such symptoms. The birds treated with Naproxen exhibited tiredness, sadness, and fits at early doses. Birds also died at a rapid pace during the 5 d testing. On the 3rd d, two birds died, and on the fifth, another died. The death rate gradually decreased, and on the 18th d, two birds died.
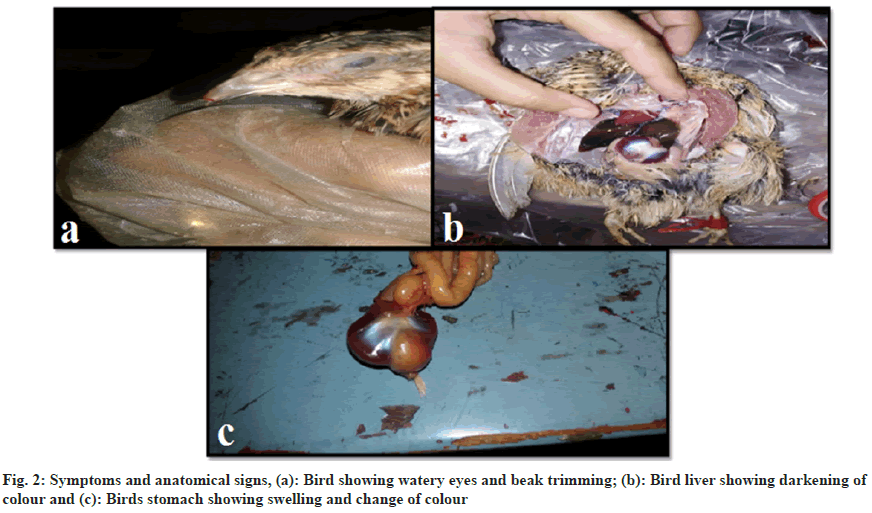
Some common observations were found in Famotidine and Naproxen-treated birds, including an increase in temperature, weight loss, beak trimming (fig. 2a), and decreased food and water intake. Jaundice was evident due to liver enlargement and yellowish coloration (fig. 2b). Swelling and colour change were also noted in one lobe of the stomach (fig. 2c).
The results of hematological analysis are presented in Table 1. Serological parameters are explained in Table 2.
| Trial | Parameters | Untreated group (summer) | Famotidine (summer) | p Value (summer) | Untreated group (winter) | Famotidine (winter) | p value (winter) |
|---|---|---|---|---|---|---|---|
| 5 d | PCV % | 37.1±3.05 | 43.9±7.0 | 0.51 | 38.80±1.35 | 43.85±5.48 | 0.41 |
| 5 d | MCV (fb) | 131.2±2.30 | 134.6±2.19 | 0.32 | 131.8±1.60 | 134.25±1.36 | 0.28 |
| 5 d | TRBC count | 2.78±0.000 | 2.68±0.21 | 0.73 | 2.94±0.08 | 3.47±0.38 | 0.30 |
| 5 d | TWBC count | 183000±2000 | 380983±110112 | 0.258 | 183000±1154 | 419000±91330 | 0.081 |
| 10 d | PCV % | 38.166±2.033 | 37.53±0.881 | 0.789 | 41.06±4.50 | 39.24±1.67 | 0.715 |
| 10 d | MCV (fb) | 130.96±1.348 | 145.20±0.585 | 0.001 | 133.32±1.54 | 147.74±6.35 | 0.059 |
| 10 d | TRBC count | 2.746±0.033 | 3.22±0.373 | 0.269 | 3.022±0.263 | 3.088±0.081 | 0.817 |
| 10 d | TWBC count | 175600±7489 | 447833±80947 | 0.029 | 254120±78889 | 474240±74035 | 0.076 |
| 15 d | PCV % | 38.80 ± 1.35 | 38.13±2.65 | 0.834 | 38.68±0.75 | 36.58±3.27 | 0.550 |
| 15 d | MCV (fb) | 131.93±1.97 | 148.73±2.49 | 0.006 | 130.30±1.47 | 151.90±6.73 | 0.014 |
| 15 d | TRBC count | 2.94±0.080 | 3.07±0.096 | 0.350 | 2.80±0.155 | 3.00±0.17 | 0.427 |
| 15 d | TWBC count | 268000±9539 | 770300±85692 | 0.079 | 294800±22341 | 453920±18482 | 0.001 |
| 20 d | PCV % | 48.30±12.56 | 33.43±3.67 | 0.320 | 46.07±9.15 | 42.05±0.705 | 0.677 |
| 20 d | MCV (fb) | 146.56±3.09 | 151.46±3.38 | 0.345 | 143.40±3.84 | 133.92±3.69 | 0.126 |
| 20 d | TRBC count | 3.09±0.761 | 2.77±0.39 | 0.728 | 3.05±0.53 | 3.317±0.004 | 0.647 |
| 20 d | TWBC count | 183000±1154 | 458166±7218 | 0.000 | 183000±1154 | 530633±25845 | 0.000 |
Note: Mean±Standard Deviation Error (SEM), with p values indicating statistical significance
Table 1: Hematological Parameters of Famotidine-Treated and Untreated Quails Over 5, 10, 15, and 20 D In Summer and Winter Trials
| Trial | Parameters | Untreated group (summer) | Famotidine (summer) | p value (summer) | Untreated group (winter) | Famotidine (Winter) | p value (winter) |
|---|---|---|---|---|---|---|---|
| 5 d | Creatinine (mg/dl) | 0.3±0.10 | 0.267±0.067 | 0.79 | 0.33±0.066 | 0.37±0.02 | 0.51 |
| 5 d | Cholesterol (mg/dl) | 195.00±2.30 | 194.33±22.83 | 0.98 | 191.66±4.05 | 197.50±17.30 | 0.78 |
| 5 d | CKMB (U/L) | 2670±140.0 | 2695.6±191.9 | 0.1 | 1441.5±832.2 | 409.1±204.5 | 0.22 |
| 5 d | Total Protein (g/dl) | 3.85±0.05 | 3.33±0.20 | 0.91 | 3.10±0.05 | 4.33±0.54 | 0.13 |
| 5 d | Albumin (g/dl) | 2.0±0.2 | 1.59±0.49 | 0.32 | 1.93±0.13 | 1.91±0.54 | 0.93 |
| 10 d | Creatinine (mg/dl) | 0.266±0.066 | 0.366±0.033 | 0.251 | 0.340±0.400 | 0.28±0.0374 | 0.305 |
| 10 d | Cholesterol (mg/dl) | 205.33±10.58 | 192.00±3.605 | 0.299 | 201.80±7.059 | 184.80±4.140 | 0.71 |
| 10 d | CKMB (U/L) | 2733.33±96.83 | 2625.33±329.94 | 0.786 | 2884.0±125.76 | 2274.00±313.86 | 0.109 |
| 15 d | Creatinine (mg/dl) | 0.33±0.033 | 0.366±0.033 | 0.347 | 0.320±0.037 | 0.400±0.000 | 0.065 |
| 15 d | Cholesterol (mg/dl) | 174.00±1.154 | 151.66±16.91 | 0.258 | 175.60±1.208 | 137.80±6.135 | 0.000 |
| 15 d | CKMB (U/L) | 3783.00±76.79 | 2601.33±44.66 | 0.000 | 3715±64.12 | 2631.60±502.53 | 0.065 |
| 20 d | Creatinine (mg/dl) | 0.366±0.066 | 0.333±0.033 | 0.678 | 0.375±0.047 | 0.300±0.040 | 0.278 |
| 20 d | Cholesterol (mg/dl) | 160.00±3.51 | 165.00±2.51 | 0.312 | 147.50±12.74 | 167.07±0.942 | 0.176 |
| 20 d | CKMB (U/L) | 2883.33±228.13 | 2598±49.81 | 0.289 | 3013.75±207.43 | 2637.75±19.81 | 0.121 |
Note: Mean±SEM, with p values indicating statistical significance
Table 2: Serological Parameters of Famotidine-Treated and Untreated Quails Over 5, 10, 15, and 20 D In Summer and Winter Trials
During the 5 d summer trial, hematological parameters did not show a significant increase in the Famotidinetreated groups. However, among serological markers, cholesterol and ALAT levels exhibited notable increases (p=0.003 and p=0.008, respectively), which are smaller than the threshold of significance (p=0.05). In the 10 d trial, the Famotidine-treated quails had significantly higher Mean Corpuscular Volume (MCV) and White Blood Cell (WBC) counts (p=0.001 and p=0.029, respectively). Among serological parameters, Creatine Kinase Myocardial Band (CKMB) showed a significant increase (p=0.014). During the 15 d trial, the MCV and WBC count remained elevated (p=0.006 and p=0.079, respectively). Serological markers CKMB and ASAT were significantly higher than normal, with a p-value below 0.05. By the 20th d, among hematological indicators, WBC count was significantly increased (p=0.000). In serological parameters, albumin and ALAT levels were statistically significant at p=0.011 and p=0.022, respectively.
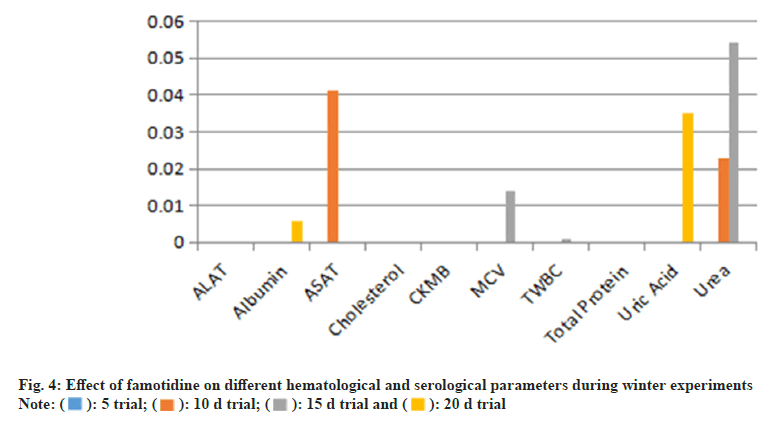
In the 5 d winter trial, the Famotidine-treated groups exhibited a significant increase in MCV and WBC count (p=0.059 and p=0.000, respectively). Among serological markers, cholesterol (p=0.011), albumin (p=0.007), and uric acid (p=0.03) were significantly higher. In the 10 d trial, quails treated with Famotidine demonstrated a significant decrease in MCV (p=0.059), while ALAT exhibited a substantial increase (p=0.001). The 15 d study showed significant increases in WBC count (p=0.001) and total protein (p=0.000). In the 20 d study, only WBC count exhibited a significant rise (p=0.002), whereas albumin (p=0.014) and ALAT (p=0.036) remained statistically elevated. Additionally, CKMB and ASAT levels were significantly higher than normal. Regarding hematological markers, WBC count remained elevated beyond the 20th d.
NSAIDs such as Famotidine and Naproxen are administered to animals and birds to regulate body temperature, reduce inflammation, alleviate pain, and prevent blood clotting. Birds receiving weightdependent doses of NSAIDs displayed health issues such as hypersomnia, restlessness, and significant foot swelling. Symptoms progressed to fever, puffy eyes, and weight reduction, followed by watery eyes. The experiment was conducted in both summer and winter, revealing similar health concerns across seasons. However, the irritation and watery eyes were more pronounced in the summer-treated birds.
The findings from hematological and serological analyses highlight significant variations due to Famotidine treatment, indicating potential physiological impacts that warrant further investigation.
Table 3 explain the blood cell variables. The serological parameters are explained in Table 4.
| Trial | Parameters | Untreated group (summer) | Naproxen (summer) | P-value (summer) | Untreated group (winter) | Naproxen (winter) | p value (winter) |
| 5 d | PCV % | 37.1±3.05 | 43.9 ± 7.0 | 0.24 | 38.80±1.35 | 35.16±0.54 | 0.06 |
| 5 d | MCV (fl) | 131.2±2.30 | 134.6±2.19 | 0.12 | 131.8±1.60 | 142.10±0.66 | 0.004 |
| 5 d | TRBC count | 2.78±0.000 | 2.68±0.21 | 0.337 | 2.94±0.08 | 2.81±0.081 | 0.34 |
| 5 d | TWBC count | 183000±2000 | 380983±110112 | 0.461 | 183000±1154 | 18966±1201 | 0.016 |
| 10 d | PCV % | 38.166±2.033 | 34.46±1.008 | 0.178 | 41.06±4.50 | 34.44±3.06 | 0.259 |
| 10 d | MCV (fl) | 130.96±1.348 | 142.13±1.066 | 0.003 | 133.32±1.54 | 146.92±2.62 | 0.002 |
| 10 d | TRBC count | 2.746±0.033 | 2.950±0.143 | 0.24 | 3.022±0.263 | 2.682±0.222 | 0.352 |
| 10 d | TWBC count | 175600±7489 | 414266±64688 | 0.021 | 254120±78889 | 452360±43649 | 0.059 |
| 15 d | PCV % | 38.80±1.35 | 32.30±5.21 | 0.328 | 38.68±0.75 | 38.24±4.11 | 0.919 |
| 15 d | MCV (fl) | 131.93±1.97 | 144.10±4.21 | 0.059 | 130.30±1.47 | 146.78±6.49 | 0.038 |
| 15 d | TRBC count | 2.94±0.080 | 2.740±0.115 | 0.229 | 2.80±0.155 | 3.02±0.23 | 0.463 |
| 15 d | TWBC count | 268000±9539 | 558933±256653 | 0.000 | 294800±22341 | 44080±19669 | 0.001 |
| 20 d | PCV % | 48.30±12.56 | 42.33±4.09 | 0.675 | 46.07±9.15 | 53.12±0.704 | 0.472 |
| 20 d | MCV (fl) | 146.56±3.09 | 152.50±11.81 | 0.653 | 143.40±3.84 | 152.6±1.139 | 0.061 |
| 20 d | TRBC count | 3.09±0.761 | 2.88±0.247 | 0.800 | 3.05±0.53 | 4.23±0.221 | 0.089 |
| 20 d | TWBC count | 183000±1154 | 51735±1350 | 0.000 | 530633±25845 | 478233±39359 | 0.002 |
Note: Mean±SEM, with p values indicating statistical significance
Table 3: Hematological Parameters of Naproxen-Treated and Untreated Quails Over 5, 10, 15, and 20 D in Summer and Winter Trials Data
| Trial | Parameters | Untreated group (summer) | Naproxen (summer) | p value (summer) | Untreated group (winter) | Naproxen (winter) | p value (winter) |
|---|---|---|---|---|---|---|---|
| 5 d | Creatinine (mg/dl) | 0.3±0.10 | 0.367±0.33 | 0.495 | 0.33±0.066 | 0.33±0.33 | 1 |
| 5 d | Cholesterol (mg/dl) | 195.00±2.309 | 174.33±2.027 | 0.003 | 191.66±4.05 | 172.66±1.20 | 0.011 |
| 5 d | CKMB (U/L) | 2670±140.0 | 2579.0±35.6 | 0.412 | 1441.5±832.2 | 180.7±104.3 | 0.34 |
| 5 d | Total Protein (g/dl) | 3.85±0.05 | 3.50±0.26 | 0.21 | 3.10±0.05 | 3.10±0.05 | 1 |
| 10 d | Creatinine (mg/dl) | 0.266±0.066 | 0.333±0.0333 | 0.422 | 0.3400±0.400 | 0.3200±0.0374 | 0.724 |
| 10 d | Cholesterol (mg/dl) | 205.33±10.58 | 198.00±9.867 | 0.639 | 201.80±7.059 | 196.40±5.065 | 0.552 |
| 10 d | CKMB (U/L) | 2733.33±96.83 | 2210.32±75.09 | 0.014 | 2884.0±125.76 | 2455.40±197.46 | 0.105 |
| 15 d | Creatinine (mg/dl) | 0.33±0.033 | 0.300±0.057 | 0.374 | 0.320±0.037 | 0.320±0.0200 | 0.1 |
| 15 d | Cholesterol (mg/dl) | 174.00±1.154 | 183.33±19.22 | 0.653 | 175.60±1.208 | 173.8±13.05 | 0.89 |
| 15 d | CKMB (U/L) | 3783.00±76.79 | 2031.33±262.66 | 0.003 | 3715±64.12 | 2200.60±199.21 | 0.000 |
| 20 d | Creatinine (mg/dl) | 0.366±0.066 | 0.533±0.033 | 0.089 | 0.375±0.047 | 0.425±0.075 | 0.595 |
| 20 d | Cholesterol (mg/dl) | 160.00±3.51 | 160.66±3.43 | 0.899 | 147.50±12.74 | 156.50±1.190 | 0.508 |
| 20 d | CKMB (U/L) | 2883.33±228.13 | 2472.00±24.87 | 0.148 | 3013.75±207.43 | 2527.00±11.82 | 0.058 |
Note: Mean±SEM, with p values indicating statistical significance
Table 4: Serological Parameters of Naproxen-Treated and Untreated Quails Over 5, 10, 15, and 20 D in Summer and Winter Trials
Over the 5 d period, no statistically significant changes were observed in hematological parameters following Naproxen administration. However, serological markers exhibited notable alterations, with cholesterol and ALAT levels significantly increasing (p=0.003 and p=0.008, respectively). By the 10th d, treated quails displayed a significant increase in MCV (p=0.003) and Total WBC (TWBC) count (p=0.021). Furthermore, creatine kinase MB (CKMB) levels showed a significant rise (p=0.014).
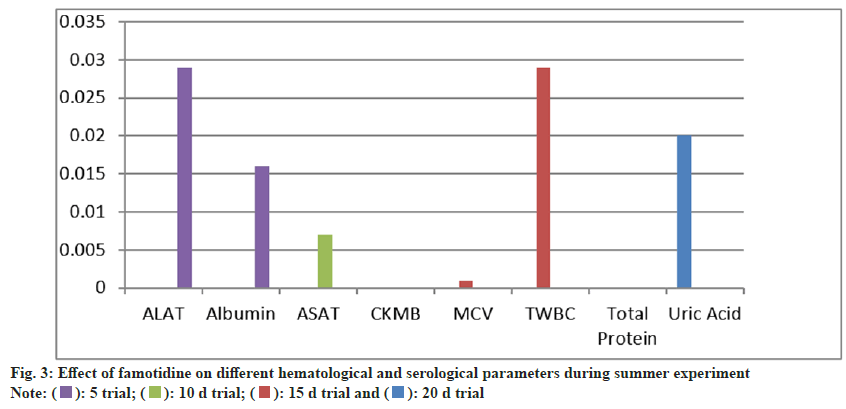
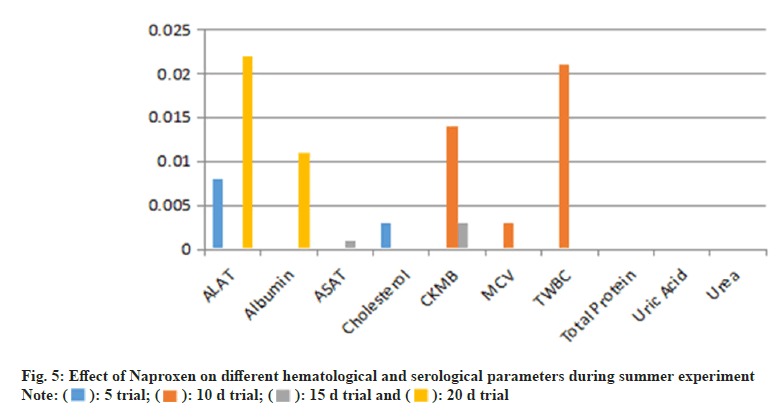
During the 15 d trial, both MCV (p=0.059) and TWBC count (p=0.000) were elevated, with serological markers CKMB and Aspartate Aminotransferase (ASAT) significantly increased. At the 20 d mark, TWBC count remained significantly higher than in the control group, while albumin and ALAT levels were also statistically elevated (p=0.011 and p=0.022, respectively). These findings suggest a progressive impact of Naproxen on hematological and serological parameters over prolonged exposure (fig. 3).
In the 5 d winter experiment, a significant increase was observed in MCV (p=0.004) and TWBC count (p=0.016). Cholesterol (p=0.011), albumin (p=0.007), and uric acid levels were significantly elevated among the serological markers. By the 10th d, MCV significantly decreased (p<0.05), while ALAT exhibited a marked increase (p=0.001).
During the 15 d period, the average number of Red Blood Cells (RBCs) was significantly affected (p=0.038), while TWBC count remained significantly higher (p=0.001). A highly significant rise in total protein (p=0.000) was also noted in serology. By the 20th d, TWBC count showed a continued increase (p=0.002), with albumin (p=0.014) and ALAT (p=0.036) levels also elevated. Additionally, CKMB and ASAT levels were significantly raised, indicating potential hepatic stress (fig. 4).
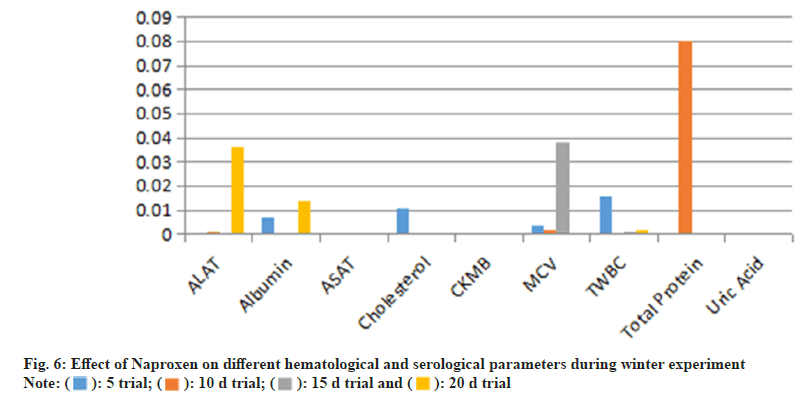
Overall, Naproxen administration resulted in hematological and biochemical alterations, with more pronounced effects on WBC proliferation and liver enzyme activity.Serological analysis (Table 4) indicated that Naproxen administration resulted in significant alterations in biochemical markers, particularly CKMB, ALAT, and albumin. CKMB levels were significantly higher on day 10 (p=0.014) and d 15 (p=0.003) in the summer trial, whereas in the winter trial, CKMB remained elevated at d 15 (p=0.000). The increase in CKMB levels suggests possible myocardial stress or tissue damage. Similarly, ALAT levels showed a significant increase (p=0.000) in the summer trial, while albumin and ASAT levels were elevated beyond normal limits in the 20 d trials, indicating potential hepatic dysfunction (fig. 5 and fig. 6).
Table 5 and Table 6 present the biochemical parameters of Naproxen and Famotidine-treated groups during summer and winter trials. Naproxen administration led to a significant decrease in cholesterol levels in both seasons (p=0.003 in summer, p=0.011 in winter), which may indicate altered lipid metabolism or liver function impairment. Additionally, uric acid levels exhibited a substantial reduction (p=0.03) in the summer trial, whereas in the winter trial, albumin levels were significantly lower (p=0.01), suggesting possible protein synthesis disturbances.
| Parameters | Untreated group | Famotidine | Naproxen | p value | Range |
|---|---|---|---|---|---|
| Creatinine (mg/dl) | 0.3±0.10 | 0.267±0.067 | 0.367±0.33 | 0.53 | (0.020-1.5706) |
| Cholesterol (mg/dl) | 195.00±2.309 | 194.33±2.83 | 174.33±2.027 | 0.58 | (96.08-292.5) |
| CKMB (U/L) | 2670±140.0 | 2695.6±191.9 | 2579.0±35.6 | 0.82 | (891.1-4448.8) |
| Total Protein (g/dl) | 3.85±0.05 | 3.33±0.20 | 3.50±0.26 | 0.29 | (2.46-4.48) |
| Albumin (g/dl) | 2.0±0.2 | 1.59±0.49 | 1.59±0.04 | 0.05 | (0.54-4.54) |
| ASAT (U/L) | 13.5±1.50 | 9.3±1.45 | 11.6±1.86 | 0.32 | (3.08-32.5) |
| ALAT (U/L) | 11.0±1.00 | 8.00±0.57 | 1.52±0.88 | 0.000 | (1.70-23.7) |
| Urea (mg/dl) | 55.5±0.50 | 59.3±3.38 | 59.66±4.05 | 0.72 | (42.2-77.1) |
| Uric Acid (mg/dl) | 19.8±0.70 | 13.8±0.95 | 15.1±1.17 | 0.03 | (9.7-28.6) |
Note: Data are presented as Mean±SEM, with p values indicating the statistical significance of the comparisons
Table 5: Blood Biochemical Variables of Famotidine and Naproxen-Treated and Untreated Groups in Summer Trials
| Parameters | Untreated group | Famotidine | Naproxen | p value | Range |
|---|---|---|---|---|---|
| Creatinine (mg/dl) | 0.33±0.066 | 0.37±0.02 | 0.33±0.33 | 0.712 | (0.045-0.62) |
| Cholesterol (mg/dl) | 191.66±4.05 | 197.50±17.30 | 172.66±1.20 | 0.395 | (142.6-252.8) |
| CKMB (U/L) | 1441.5±832.2 | 409.1±204.5 | 180.7±104.3 | 0.323 | (1739.2-500.2) |
| Total Protein (g/dl) | 3.10±0.05 | 4.33±0.54 | 3.10±0.05 | 0.081 | (2.59-6.07) |
| Albumin (g/dl) | 1.93±0.13 | 1.91±0.54 | 1.23±0.03 | 0.01 | (1.03-2.50) |
| ASAT (U/L) | 13.3±0.88 | 12.25±1.54 | 3.51±2.02 | 0.513 | (1.94-19.39) |
| ALAT (U/L) | 11.3±0.66 | 13.2±2.65 | 10.6±0.66 | 0.624 | (4.79-21.7) |
| Urea (mg/dl) | 56.33±0.88 | 57.25±3.17 | 56.66±1.45 | 0.913 | (47.15-67.34) |
| Uric Acid (mg/dl) | 17.90±1.94 | 20.22±2.25 | 8.70±0.32 | 0.712 | (7.31-27.41) |
Note: Data are presented as Mean±SEM, with p values indicating the statistical significance of the comparisons
Table 6: Blood Biochemical Variables of Famotidine and Naproxen-Treated and Untreated Groups in Winter Trials
Famotidine-treated birds displayed a notable decline in albumin levels (p=0.05) in the summer trial and a significant decrease in uric acid levels (p=0.03), which may be indicative of renal stress. Additionally, CKMB levels showed a downward trend, though statistical significance was not reached (p=0.323), suggesting a lesser impact on cardiac biomarkers compared to Naproxen. A noteworthy observation was the significant increase in total protein levels in the winter trial (p=0.081), which may reflect alterations in protein metabolism or immune response.
The variations in hematological and serological parameters highlight the physiological impact of NSAID administration. The elevated WBC count suggests an immune response, potentially due to inflammatory processes induced by Naproxen. The significant rise in ALAT and CKMB levels indicates potential hepatic and myocardial stress, raising concerns about long-term administration of Naproxen in quails. Seasonal variations also played a role, as winter-treated birds exhibited greater disturbances in albumin and uric acid levels, while summer-treated birds showed more pronounced fluctuations in cholesterol and liver enzymes.
The study provides evidence that Naproxen and Famotidine administration influence key hematological and biochemical markers in quails. Naproxen treatment notably affected WBC count, MCV, CKMB, and ALAT levels, indicating inflammatory and hepatic stress. Famotidine showed moderate effects, primarily influencing albumin and uric acid levels. These findings suggest that prolonged NSAID usage may pose health risks, particularly with seasonal variations affecting physiological responses. Future research should focus on optimizing NSAID dosage and evaluating long-term consequences in Avian species.
Acknowledgments:
The authors gratefully acknowledge the funding of the Deanship of Graduate Studies and Scientific Research, Jazan University, Saudi Arabia, through project number: (RG24-M015).
Conflict of interests:
The authors declared no conflict of interests.
References
- Baer J, Lansford R, Cheng K. Japanese quail as a laboratory animal model. Lab Anim Med 2015;37(11):1087-108.
[Crossref] [Google Scholar] [PubMed]
- Agina OA, Ezema WS, Iwuoha EM. The haematology and serum biochemistry profile of adult Japanese quail (Coturnix japonica). Notulae Sci Biol 2017;9(1):67-72.
- Cheng KM, Bennett DC, Mills AD. The Japanese quail. UFAW Handbook Care Management Lab Other Res Anim 2010:655-73.
- Christopher S, Tadlock BA, Veroneau BJ, Harnish C, Perera NK, Knab AM, et al. Epidemiological profile of pain and non-steroid anti-inflammatory drug use in collegiate athletes in the United States. BMC Musculoskeletal Disord 2020;21(1):561.
[Crossref] [Google Scholar] [PubMed]
- Sohail R, Mathew M, Patel KK, Reddy SA, Haider Z, Naria M, et al. Effects of non-steroidal anti-inflammatory drugs (NSAIDs) and gastroprotective NSAIDs on the gastrointestinal tract: A narrative review. Cureus 2023;15(4):e37080.
[Crossref] [Google Scholar] [PubMed]
- Tyumina EA, Bazhutin GA, Cartagena Gómez AD, Ivshina IB. Nonsteroidal anti-inflammatory drugs as emerging contaminants. Microbiology 2020;89:148-63.
- Jacobs CC, Schnabel LV, McIlwraith CW, Blikslager AT. Non‐steroidal anti‐inflammatory drugs in equine orthopaedics. Equine Vet J 2022;54(4):636-48.
[Crossref] [Google Scholar] [PubMed]
- Xu N, Zhang C, Jing L, Mou S, Cao X, Yu Z. Protective effect and mechanism of rebamipide on NSAIDs associated small bowel injury. Int Immunopharmacol 2021;90:107136.
[Crossref] [Google Scholar] [PubMed]
- Di Salvo A, Chiaradia E, Nannarone S, Della Rocca G. Intra-articular use of analgesic/antinflammatory drugs in dogs and horses. Res Vet Sci 2021;134:159-70.
[Crossref] [Google Scholar] [PubMed]
- Flood J, Stewart AJ. Non-steroidal anti-inflammatory drugs and associated toxicities in horses. Animals 2022;12(21):2939.
[Crossref] [Google Scholar] [PubMed]
- Schug SA. Do NSAIDs really interfere with healing after surgery? J Clin Med 2021;10(11):2359.
[Crossref] [Google Scholar] [PubMed]
- Boatright-Horowitz SL. Avian bornaviral ganglioneuritis: Current debates and unanswered questions. Veterinary Med Int 2020;2020(1):6563723.
- Parolini M. Toxicity of the Non-Steroidal Anti-Inflammatory Drugs (NSAIDs) acetylsalicylic acid, paracetamol, diclofenac, ibuprofen and naproxen towards freshwater invertebrates: A review. Sci Total Environ 2020;740:140043.
[Crossref] [Google Scholar] [PubMed]
- Stafford KJ, Mellor DJ, Vogel K. Painful husbandry procedures in livestock and poultry. Improv Anim Welfare 2015:96-124.
- Turk E, Tekeli IO, Corum O, Durna Corum D, Kirgiz FC, Cetin G, et al. Pharmacokinetics of meloxicam, carprofen, and tolfenamic acid after intramuscular and oral administration in Japanese quails (Coturnix japonica). J Vet Pharmacol Ther 2021;44(3):388-96.
[Crossref] [Google Scholar] [PubMed]
- Cuthbert R, Parry-Jones J, Green RE, Pain DJ. NSAIDs and scavenging birds: Potential impacts beyond Asia's critically endangered vultures. Biol Lett 2007;3(1):91-4.
[Crossref] [Google Scholar] [PubMed]
- Naidoo V, Swan GE. Diclofenac toxicity in Gyps vulture is associated with decreased uric acid excretion and not renal portal vasoconstriction. Comp Biochem Physiol C Toxicol Pharmacol 2009;149(3):269-74.
[Crossref] [Google Scholar] [PubMed]
- Castro-Pastrana LI, Palacios-Rosas E, Toledo-Wall ML, Cerro-López M. Worldwide occurrence, detection, and fate of nonsteroidal anti-inflammatory drugs in water. Springer Int Publish 2020;55-82.
- Ngubane NP, Naicker D, Ncube S, Chimuka L, Madikizela LM. Determination of naproxen, diclofenac and ibuprofen in Umgeni estuary and seawater: A case of northern Durban in KwaZulu-Natal province of South Africa. Reg Stud Marine Sci 2019;29:1-10.
- Distefano GG, Zangrando R, Basso M, Panzarin L, Gambaro A, Ghirardini AV, et al. Assessing the exposure to human and veterinary pharmaceuticals in waterbirds: The use of feathers for monitoring antidepressants and nonsteroidal anti-inflammatory drugs. Sci Total Environ 2022;821:153473.
[Crossref] [Google Scholar] [PubMed]
- Markandya A, Taylor T, Longo A, Murty MN, Murty S, Dhavala K. Counting the cost of vulture declines-economic appraisal of the benefits of the vulture in India. Ecol Eco 2008;67(2):194-204.
- Cuthbert RJ, Taggart MA, Prakash V, Chakraborty SS, Deori P, Galligan T, et al. Avian scavengers and the threat from veterinary pharmaceuticals. Philos Trans R Soc Lond B Biol Sci 2014;369(1656):20130574.
[Crossref] [Google Scholar] [PubMed]
- Varas‐Lorenzo C, Riera‐Guardia N, Calingaert B, Castellsague J, Pariente A, Scotti L, et al. Stroke risk and NSAIDs: A systematic review of observational studies. Pharmacoepidemiol Drug Saf 2011;20(12):1225-36.
[Crossref] [Google Scholar] [PubMed]
- Schaffer D, Florin T, Eagle C, Marschner I, Singh G, Grobler M, et al. Risk of serious NSAID‐related gastrointestinal events during long‐term exposure: A systematic review. Med J Aust 2006;185(9):501-6.
[Crossref] [Google Scholar] [PubMed]
- LaForge JM, Urso K, Day JM, Bourgeois CW, Ross MM, Ahmadzadeh S, et al. Non-steroidal anti-inflammatory drugs: Clinical implications, renal impairment risks, and AKI. Adv Ther 2023;40(5):2082-96.
[Crossref] [Google Scholar] [PubMed]
- Thiruchenthooran V, Sánchez-López E, Gliszczyńska A. Perspectives of the application of non-steroidal anti-inflammatory drugs in cancer therapy: Attempts to overcome their unfavorable side effects. Cancers 2023;15(2):475-78.
[Crossref] [Google Scholar] [PubMed]
- Panchal NK, Sabina EP. Non-Steroidal Anti-Inflammatory Drugs (NSAIDs): A current insight into its molecular mechanism eliciting organ toxicities. Food Chem Toxicol 2023;172:113598.
[Crossref] [Google Scholar] [PubMed]
- Steele VE, Rao CV, Zhang Y, Patlolla J, Boring D, Kopelovich L, et al. Chemopreventive efficacy of naproxen and nitric oxide-naproxen in rodent models of colon, urinary bladder, and mammary cancers. Cancer Prev Res 2009;2(11):951-6.
[Crossref] [Google Scholar] [PubMed]
- McGettigan P, Henry D. Cardiovascular risk with non-steroidal anti-inflammatory drugs: Systematic review of population-based controlled observational studies. PLoS Med 2011;8(9):e1001098.
[Crossref] [Google Scholar] [PubMed]
- Abualhasan MN, Al-Masri MY, Manasara R, Yadak L, Abu-Hasan NS. Anti‐inflammatory and anticoagulant activities of synthesized NSAID prodrug esters. Scientifica 2020;2020(1):9817502.
[Crossref] [Google Scholar] [PubMed]
- Khan MF, Zuthi SS, Kayser MS, Islam MS, Asad S, Rashid MA. a simple rp-hplc method development and validation for the simultaneous estimation of naproxen and rabeprazole. J Appl Pharm Sci 2016;6(11):147-52.
- Lubet RA, Clapper ML, McCormick DL, Pereira MA, Chang WC, Steele VE, et al. Chemopreventive efficacy of targretin in rodent models of urinary bladder, colon/intestine, head and neck and mammary cancers. Oncol Rep 2012;27(5):1400-6.
[Crossref] [Google Scholar] [PubMed]
- Bharati S, Gaikwad V, Pawar A, Chellampillai B. Investigation of a biopolymer-based pH-responsive and sustained release raft-gel-forming tablet of famotidine: In-vitro, ex-vivo, bioavailability and anti-ulcer evaluation in New Zealand albino rabbit. J Drug Deliv Sci Tech 2023;86:1-10.
- Şener-Muratoğlu G, Paskaloğlu K, Arbak S, Hürdağ C, Ayanoğlu-Dülger G. Protective effect of famotidine, omeprazole, and melatonin against acetylsalicylic acid-induced gastric damage in rats. Dig Dis Sci 2001;46:318-30.
[Crossref] [Google Scholar] [PubMed]
- Cai L, Zhao C, Cao X, Lu M, Li N, Luo Y, et al. Chinese herb pollen derived micromotors as active oral drug delivery system for gastric ulcer treatment. Bioact Mater 2024;32:28-36.
[Crossref] [Google Scholar] [PubMed]
- Kogan L, Hellyer P, Rishniw M, Schoenfeld-Tacher R. The US opioid epidemic and its impact on US general practice veterinarians. Front Vet Sci 2019;6:1-12.
[Crossref] [Google Scholar] [PubMed]
- Mogil JS. Animal models of pain: Progress and challenges. Nat Rev Neurosci 2009;10(4):283-94.
[Crossref] [Google Scholar] [PubMed]
- Fenwick N, Duffus SE, Griffin G. Pain management for animals used in science: Views of scientists and veterinarians in Canada. Animals 2014;4(3):494-514.
[Crossref] [Google Scholar] [PubMed]
- Kovacova-Hanuskova E, Buday T, Gavliakova S, Plevkova J. Histamine, histamine intoxication and intolerance. Allergol Immunopathol 2015;43(5):498-506.
[Crossref] [Google Scholar] [PubMed]
- Yue JX, Wang RR, Yu J, Tang YY, Hou WW, Lou GD, et al. Histamine upregulates Nav1. 8 expression in primary afferent neurons via H2 receptors: Involvement in neuropathic pain. CNS Neurosci Ther 2014;20(10):883-92.
[Crossref] [Google Scholar] [PubMed]
- Zendehdel M, Hamidi F, Hassanpour S. The effect of histaminergic system on nociceptin/orphanin FQ induced food intake in chicken. Int J Pept Res Ther 2015;21:179-86.
- Scholtz N, Halle I, Flachowsky G, Sauerwein H. Serum chemistry reference values in adult Japanese quail (Coturnix japonica) including sex-related differences. Poult Sci 2009;88(6):1186-90.
[Crossref] [Google Scholar] [PubMed]
- Ng FH, Wong SY, Lam KF, Chu WM, Chan P, Ling YH, et al. Famotidine is inferior to pantoprazole in preventing recurrence of aspirin-related peptic ulcers or erosions. Gastroenterology 2010;138(1):82-8.
[Crossref] [Google Scholar] [PubMed]
- Mahdavi K, Zendehdel M, Zarei H. The role of central neurotransmitters in appetite regulation of broilers and layers: Similarities and differences. Vet Res Commun 2024;48(3):1313-28.
[Crossref] [Google Scholar] [PubMed]
- Van Dingenen J, Pieters L, Vral A, Lefebvre RA. The H2S-releasing naproxen derivative ATB-346 and the slow-release H2S donor GYY4137 reduce intestinal inflammation and restore transit in postoperative ileus. Front Pharmacol 2019;10:1-16.
[Crossref] [Google Scholar] [PubMed]
- Ekundi-Valentim E, Mesquita FP, Santos KT, de Paula MA, Florenzano J, Zanoni CI, et al. A comparative study on the anti-inflammatory effects of single oral doses of naproxen and its hydrogen sulfide (H2S)-releasing derivative ATB-346 in rats with carrageenan-induced synovitis. Med Gas Res 2013;3(1):1-9.
[Crossref] [Google Scholar] [PubMed]
- Song ZJ, Ng MY, Lee ZW, Dai W, Hagen T, Moore PK, et al. Hydrogen sulfide donors in research and drug development. Med Chem Comm 2014;5(5):557-70.
- Krishnamachari B. Medication use and immune responses in Glioma. University of Illinois at Chicago, Health Sciences Center 2011.
- Preston SJ, Arnold MH, Beller EM, Brooks PM, Buchanan WW. Comparative analgesic and anti‐inflammatory properties of sodium salicylate and acetylsalicylic acid (aspirin) in rheumatoid arthritis. Br J Clin Pharmacol 1989;27(5):607-11.
[Crossref] [Google Scholar] [PubMed]
- Scolnick B. Antihistamine agents and pitolisant might be useful for anorexia nervosa. Med Hypotheses 2019;132:1-10.
[Crossref] [Google Scholar] [PubMed]