- *Corresponding Author:
- K. M. Manjunatha
N. G. S. M. Institute of Pharmaceutical Sciences, Mangalore - 575 005, India
E-mail: manju_kat2000@yahoo.com
| Date of Submission | 13 February 2006 |
| Date of Revision | 15 February 2007 |
| Date of Acceptance | 19 May 2007 |
| Indian J Pharm Sci, 2007, 69 (3): 384-389 |
Abstract
Sustained release dosage form of diclofenac sodium containing immediate and controlled release components was designed. Solid dispersion of immediate release component was prepared using polyvinyl pyrrolidone and mannitol carriers by common solvent method. Controlled release component was prepared in form of spherical beads by ionotropic gelation technique. The beads were prepared based on dispersing drug in solutions of ionic polysaccharides such as chitosan and sodium alginate. These dispersions were dropped into solutions of counter ions such as tetrasodium pyrophosphate and calcium chloride, respectively. The beads were also prepared using agar by dropping agar-drug hot solution into a mixture of chilled liquid paraffin and water. Then, diclofenac sodium controlled release drug delivery systems were prepared by combining the immediate release and controlled release components in different ratios. The formulations were found to be effective in providing controlled release of drug for a longer period of time. The beads were characterized by scanning electron microscopy and X-ray diffraction studies.
Keywords
Diclofanac, solid dispersion, SEM, XRD, controlled release and in vitro dissolution
Diclofenac sodium is a non-steroidal antiinflammatory agent, which is widely used in the long-term therapy for rheumatoid arthritis. The biological half-life of diclofenac sodium is about 1-2 h, therefore it requires multiple dosing to maintain therapeutic drug blood level. The most frequent adverse side effects of diclofenac sodium on long-term administration are gastrointestinal disturbances, peptic ulceration and gastrointestinal bleeding. Diclofenac sodium is poorly soluble in water and acidic pH (1-3) but is rapidly soluble in alkaline pH (5-8) [1]. Hence an attempt was made to formulate a sustained release dosage form containing solid dispersion of diclofenac sodium for immediate release and beads of diclofenac sodium for controlled release, which eliminates the need for multiple dosing there by increasing patient compliance and decreasing the occurrence of adverse effects [2].
The methods of preparation of solid dispersions as well as beads were simple and reproducible. The carriers and polymers used were non-toxic, relatively less expensive and easily available. They were found to be effective in providing a constant release of drug from the formulations for a longer period of time.
Materials and Methods
Diclofenac sodium IP was received as a gift sample from BAL Pharma Ltd., Bangalore, agarose and sodium alginate were purchased from Loba Chemie, Mumbai and chitosan was received from Central Institute of Fisheries Technology, Kochi, and polyvinyl pyrrolidone and D-mannitol were supplied by Central Drug House (P) Ltd., Mumbai. All other chemicals were of either reagent or analytical grade as received.
Preparation of solid dispersion
Solid dispersions of diclofenac sodium were prepared by common solvent method using PVP and mannitol as carriers. Accurately weighed quantities of diclofenac sodium and carriers (PVP and mannitol) were taken in a beaker containing methanol and mixed until they were dissolved. Then the solvent was completely evaporated by heating on a water bath with constant stirring. The resulting mass was stored in a desiccator for two days to harden. Finally the mass was pulverized carefully by passing through 60-mesh sieve [3]. The various ratios of drug and carriers used for preparing solid dispersions are represented in Table 1.
| Combinationfor solid dispersion | Drug | Polymer | ||||
|---|---|---|---|---|---|---|
| PVP | Mannitol | Agar | Alginate | Chitosan | ||
| l | 1 | 1 | - | - | - | - |
| ll | 1 | 2 | - | - | - | - |
| lll | 1 | 3 | - | - | - | - |
| lV | 1 | - | 1 | - | - | - |
| V | 1 | - | 2 | - | - | - |
| Vl | 1 | - | 3 | - | - | - |
| Vll | 1 | - | - | 1 | - | - |
| Vlll | 1 | - | - | 2 | - | - |
| lX | 1 | - | - | 3 | - | - |
| X | 1 | - | - | - | 1 | - |
| Xl | 1 | - | - | - | 2 | - |
| Xll | 1 | - | - | - | 3 | - |
| Xlll | 1 | - | - | - | - | 1 |
| XlV | 1 | - | - | - | - | 2 |
| XV | 1 | - | - | - | - | 3 |
Table 1: Ratios of Drug, Carriers And Polymers Used In Preparation Of Solid Dispersions (I To Vi) And Beads (Vii To Xv)
Preparation of beads
Alginate, chitosan and agar beads containing diclofenac sodium in ratios of 1:1, 1:2 and 1:3 were prepared based upon the methods followed by Fattah et al.4 and Bert et al.[5] with some modifications. The drug dispersions were prepared by dispersing diclofenac sodium in 2% sodium alginate solution in water and 1% chitosan solution in 1% v/v of acetic acid. Diclofenac sodium beads of sodium alginate and chitosan were prepared by dropping drugpolymer dispersions through a 20-guage disposable syringe needle into agitated 5% aqueous counter ion solutions (100 ml) of calcium chloride and tetra sodium pyrophosphate, respectively. Agar beads were prepared by dropping hot aqueous 2% solution of agar containing drug into a mixture of chilled liquid paraffin and water (3:1 ratio) through a 20-guage disposable syringe needle. Formulation design of the beads is given in Table 2. Similarly placebo beads were prepared.
| Types of beads | Drug:Polymer | Yield of beads* (%) |
Drug entrapment* (percentage) |
Mean diameter* (mm) |
|---|---|---|---|---|
| Diclofenac sodium: alginate |
1:01 | 78.75 | 74.44 | 0.9 |
| 1:02 | 76.6 | 67.74 | 0.94 | |
| 1:03 | 73.33 | 59.88 | 1.02 | |
| Diclofenac sodium: agar |
1:01 | 75 | 84.2 | 0.89 |
| 1:02 | 71.66 | 79.38 | 0.95 | |
| 1:03 | 68.75 | 74.24 | 0.98 | |
| Diclofenac sodium: chitosan |
1:01 | 68.33 | 47.16 | 0.86 |
| 1:02 | 65.55 | 38.46 | 0.91 | |
| 1:03 | 62.5 | 34.64 | 0.96 |
*Average of three determinations
Table 2: Formulation and physico-chemical characteristics of diclofenac sodium beads.
Formulation of controlled release system
Five formulations were prepared by mixing solid dispersion and beads of drug in different ratios. Beads containing 1:3 drug:agar were selected for the formulation of controlled release. Solid dispersion and beads were mixed in different proportions such that 100 mg of drug is present in the mixture, which was filled in hard gelatin capsule for further studies. Five ratios of solid dispersion:beads chosen were represented in Table 3.
| Formulations | Solid dispersion of III | Beads of IX |
|---|---|---|
| F1 | 3 | 1 |
| F2 | 2 | 1 |
| F3 | 1 | 1 |
| F4 | 1 | 2 |
| F5 | 1 | 3 |
Table 3: Ratio of solid dispersion and beads used in formulation of controlled release formulations of diclofenac sodium.
Granulometric study
As the particle size is very significant on the release profile of drug from the dosage form, granulometric study was conducted to determine average particle size by sieve analysis using standard sieves [6] (#12, #16 and #20).
Drug entrapment efficiency
Accurately weighed quantity of beads containing 100 mg of drug was suspended in 100 ml of 0.1 N NaOH and was kept for 24 h. Next day the mixture was stirred for 15 min. The mixture was subjected to filtration and proper dilution. Diclofenac sodium content in the filtrate was analyzed spectrophotometrically at 276 nm [7].
In vitro release studies for solid dispersion
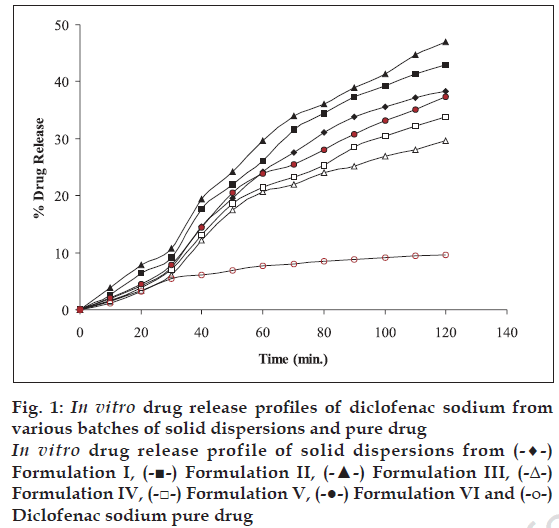
The dissolution profile of the solid dispersion was determined using a six panel USP XXIII dissolution apparatus- taking 900 ml of HCl (pH 1.2) solution for 2 h. The dissolution medium was maintained at a temperature of 37±1°. The speed of rotation of basket was 50 rpm. The basket was covered with 100- mesh nylon cloth to prevent the escape of the solid dispersion powder. The samples were withdrawn at 10 min intervals. The samples were filtered and suitably diluted to determine the absorbance at 276 nm in UVspectrophotometer Chemito-2500. Dissolution medium was replaced into the vessel after each withdrawal [10]. Cumulative percent drug release from the beads is shown in fig. 1.
Figure 1: In vitro drug release profiles of diclofenac sodium from various batches of solid dispersions and pure drug In vitro drug release profile of solid dispersions from (-♦-) Formulation I, (-■-) Formulation II, (-▲-) Formulation III, (-Δ-) Formulation IV, (-□-) Formulation V, (-●-) Formulation VI and (-ο-) Diclofenac sodium pure drug
In vitro release studies for beads
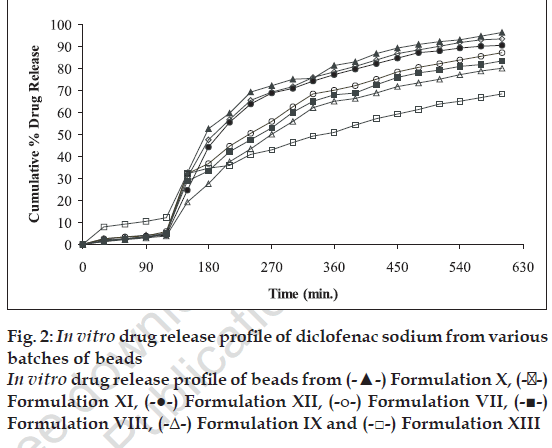
The dissolution profile of the beads was determined using a six-panel USP XXIII dissolution apparatus taking 900 ml of HCl (pH 1.2) solution for first 2 h and phosphate buffer (pH 7.4) for next 8 h. The dissolution media were maintained at a temperature of 37±1°. The speed of rotation of basket maintained was 50 rpm. The basket was covered with 100-mesh nylon cloth to prevent the escape of the beads. The samples were withdrawn at 30 min intervals. The samples were filtered and suitably diluted to determine the absorbance at 276 nm in UV- spectrophotometer Chemito-2500. Corresponding dissolution medium was replaced into the vessel after each withdrawl [9]. Cumulative percent drug release from the beads is shown in fig. 2.
In vitro release studies for diclofenac sodium controlled drug delivery system
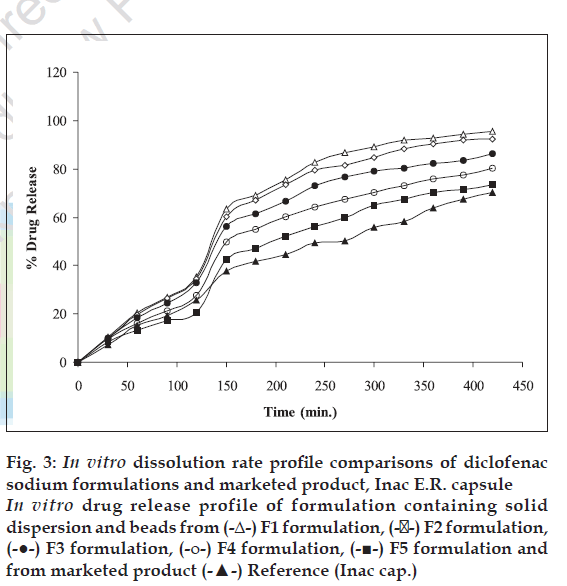
The dissolution profile of developed controlled drug delivery system (mixture of solid dispersion and beads enclosed in hard gelatin capsule) was determined in a similar manner as that of beads. Cumulative percent drug release from dosage form is shown in fig. 3.
Figure 3: In vitro dissolution rate profile comparisons of diclofenac sodium formulations and marketed product, Inac E.R. capsule In vitro drug release profile of formulation containing solid dispersion and beads from (-Δ-) F1 formulation, (-��-) F2 formulation, (-●-) F3 formulation, (-ο-) F4 formulation, (-■-) F5 formulation and from marketed product (-▲-) Reference (Inac cap.)
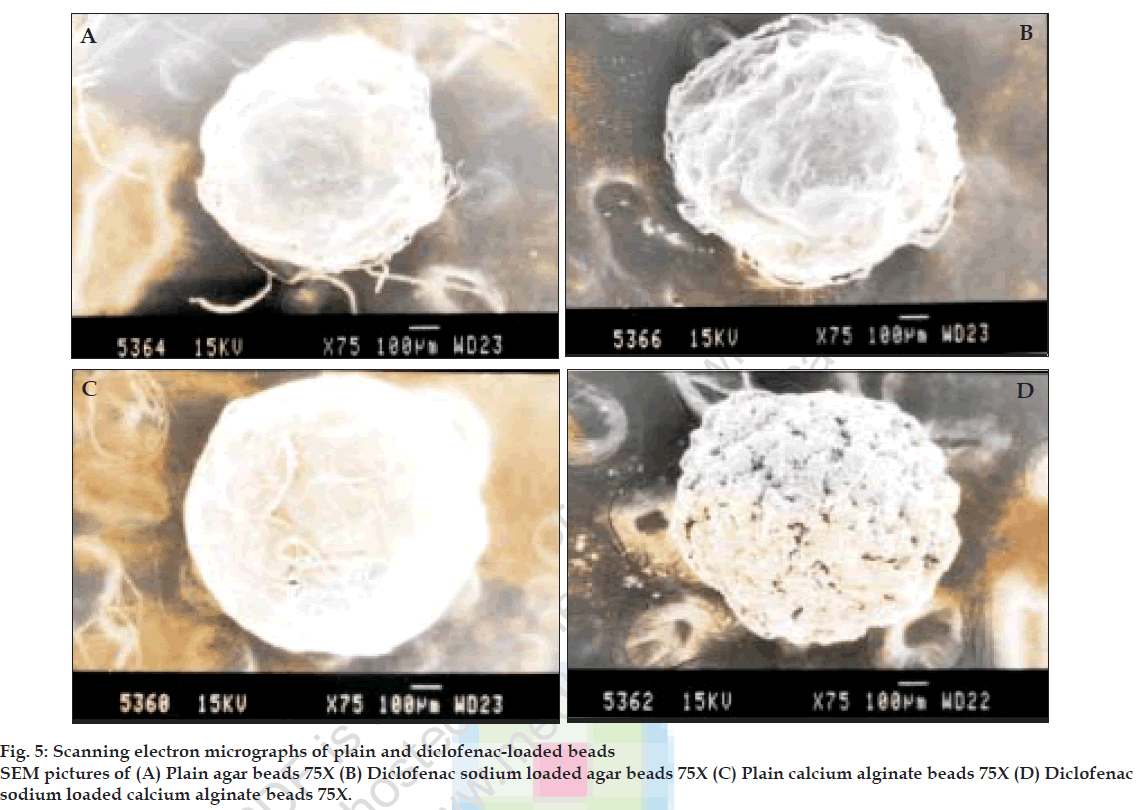
X-ray diffraction and scanning electron microscopy (SEM)
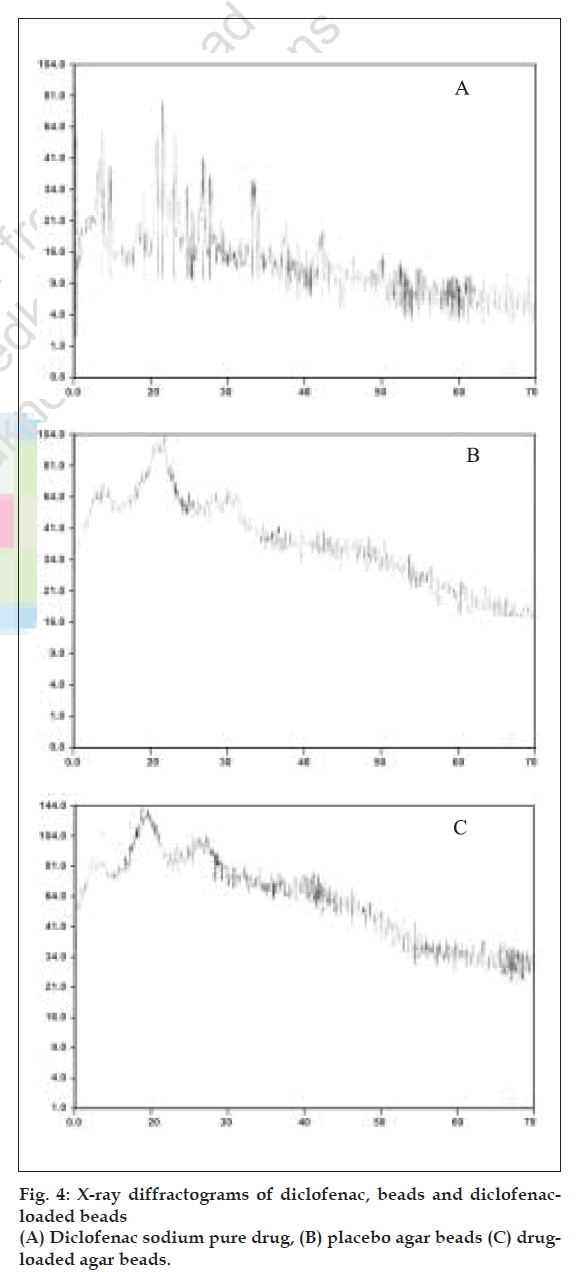
X-ray diffraction studies were performed on pure drug, calcium alginate and agar placebo beads and drug loaded calcium alginate and agar beads. Scanning electron microscopic studies were performed on calcium alginate and agar placebo beads and beads with drug to characterize the surface topography of beads. The samples were coated for 70s under an argon atmosphere with gold palladium and observed with a Jeol JSM-840 A, Japan, scanning electron microscope [8].
Results and Discussion
Diclofenac sodium is poorly soluble in water and gastric medium. As a result it possesses absorption problems in gastric medium. Hence an attempt was made to increase the solubility of drug by preparing it in solid dispersion form. PVP and mannitol were reported to be used as carriers of drug in preparing solid dispersions. Therefore, solid dispersions of diclofenac sodium were prepared using these carriers in different ratios by common solvent method. Solid dispersions were moderately fine powders with a size ranging from 194 to 246 µ (by sieve analysis method) and were free flowing.
Diclofenac sodium has high solubility in alkaline medium and may not show prolonged effect. Therefore to control the drug release and thereby absorption, beads of drug were prepared. Controlled release beads of diclofenac sodium were prepared using natural polymers such as agar, alginate and chitosan. The formed beads were sufficiently hard and spherical in shape. Beads were characterized for their particle size (by sieve analysis method), drug entrapment, X-ray diffraction and SEM.
The release pattern of solid dispersions of I to VI and pure drug were studied in HCl (pH 1.2) medium and are represented in fig. 1. These solid dispersions showed an increase in rate of release of drug in HCl (pH 1.2) medium compared to that of pure drug. PVP solid dispersions showed higher rate of release of drug than mannitol solid dispersions. Beads prepared from agar, alginate and chitosan were sufficiently hard and spherical in shape. Physico-chemical properties of beads are represented in Table 2.
In vitro dissolution studies were carried out for all the beads (except chitosan 1:2 and 1:3 ratio because of less drug entrapment efficiency) in HCl medium (pH 1.2) for first 2 h and in phosphate buffer (pH 7.4) for next 8 h. Agar beads 1:3 ratio showed more sustained and uniform release. The drug release profiles are as shown in fig. 2.
An attempt was made to formulate constant release formulations of diclofenac sodium by mfixing solid dispersions and beads in suitable proportions as given in Table 3. Solid dispersion of III and beads of IX were selected to formulate constant release formulations because these were showed desired loading and maintenance drug release patterns. The in vitro release profiles of F1 to F5 formulations are shown in fig. 3.
Marketed product (Inac TR 100 mg) was subjected to dissolution studies similar to the conditions followed for developed formulations. Comparisons of drug release profiles are represented in fig. 3. The f1 (dissimilarity factor) and f2 (similarity factor) values
were calculated using MS Excel software as described by Huey lin ju and Shu-jean liaw [11]. The f1 and f2 values are given in Table 4. Perusal of Table 4 and fig. 3 indicates that formulation F5 is more similar to the marketed product.
| Formulations | f1 (dissimilarity factor) | f2 (similarity factor) |
|---|---|---|
| F1 | 61.01 | 51.63 |
| F2 | 54.58 | 54.6 |
| F3 | 41.71 | 59.36 |
| F4 | 26.26 | 69.34 |
| F5 | 14.36 | 78.42 |
Table 4: Similarity And Dissimilarity Factors of Various Formulations In Comparison With Marketed Product
Perusal of the X-ray diffractograms (fig. 4) indicated that there is no interaction between drug and polymer. The drug could be present in amorphous form in the beads. Scanning electron micrographs of beads are shown in fig. 5. The placebo beads were nearly spherical in shape where as the loaded beads were irregular and partially shrunken.
The methods of preparation of solid dispersion as well as beads were found to be simple and reproducible. The carriers and polymers used were non-toxic, relatively less expensive and are easily available. The developed formulations were found to be effective in providing a prolonged release of drug.
References
- Tripathi KD. Essential of medical pharmacology. 4th ed. Delhi: Jaypee Brothers Medical Publishers (p) Ltd; 1998.
- .Chien YW. Controlled and modulated-release drug delivery systems. In: Swarbrick J, Boylan JC, editors. Encyclopedia of pharmaceutical technology. Vol. 3. New York; Marcel Decker Inc: 1991. p. 286-9.
- Chowdary KPR, Ramana KV, Prasad DS. Solid dispersions of nimodipine: Physico-chemical and dissolution rate studies. Indian Drugs 1994;32:543-7.
- Fatah EA, Grant, Gabr KE. Physical characteristics and release behavior of salbutamol sulfate beads prepared with different ionic polysaccharides. Drug Develop Ind Pharm 1998;24:541-7.
- Bert O, Haglund, Satyanaranayana M, Steven HN, Michael A.Preparation and evaluation of sustained release agarose beads containing ibuprofen and indomethacin. Control Drug Develop Ind Pharm 1994;20:947-57.
- Subrahmanyam CVS. Physical pharmaceutics. 2nd ed. Delhi; Vallabh Prakashan: 2000.
- Mana A, Ghosh I, Goswami N, Ghosh LK, Gupta BK. Design and evaluation of an oral controlled release microparticulate drug delivery system of nimesulide by ionotropic gelation technique and statistical optimization by factorial analysis. J Sci Ind Res 1999;58:712-22.
- Ramarao P, Prakash VD. Formulation and evaluation of solid dispersion systems of indomethacin. Eastern Pharmacist 2000;43:97-9.
- Gohel MC, Sheth MN, Jani GK, Patel. Design of chitosan microspheres containing diclofenac sodium. Indian J Pharm Sci 1994;56:210-4.
- Roland B, Ornlaksana P. Spherical agglomerates of water-insoluble drugs. J Pharm Sci 1989;78:964-7.
- Huey LJ, Shu JL. On the assessment of similarity of drug dissolution proÞles- A simulation study. Drug Inform J 1997;31:1273-89.