- *Corresponding Author:
- S. Y. Chaudhari
Department of Rasashastra and Bhaishajya Kalpana, Institute for Post Graduate Teaching and Research in Ayurveda
Gujarat Ayurved University, Jamnagar-361 008, India
E-mail: drswapnilyc13@gmail.com
| Date of Submission | 29 November 2016 |
| Date of Revision | 26 June 2017 |
| Date of Acceptance | 13 February 2018 |
| Indian J Pharm Sci 2018;80(2):325-333 |
This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms
Abstract
Rasamanikya (an arsenical formulation of Ayurveda) contains Haratala (arsenic trisulphide) as an integral component. Concerns are being raised on such Ayurveda formulations with heavy metals in their composition for safety aspects. Though, these have been used safely in Ayurvedic clinical practice since ages without any noticeable untoward effects; there is need to generate scientific evidence that these are safe and nontoxic. In the present study, safety profile of Rasamanikya prepared from Kushmanda swarasa shodhita haratala (arsenic trisulphide processed in juice of Benincasa hispida) was evaluated through acute and chronic toxicity studies. In acute toxicity, Rasamanikya was administered at a maximal dose of 2000 mg/kg to overnight fasted rats and observed closely for behavioral changes, signs of toxicity and mortality if any, continuously for the first six hours and thereafter periodically up to 14 days. In the chronic toxicity evaluation, the drug was administered daily at the doses of 22.5, 112.5 and 225 mg/kg along with honey and ghee as an adjuvant to rats for 90 days followed by a 30-day recovery period. Animals were sacrificed on the 91st day and hematological, serum biochemical parameters and histopathology of organs were studied. In acute toxicity, Rasamanikya at the dose of 2000 mg/kg did not produced any observable toxic effects or mortality. Safety of Rasamanikya at therapeutic and five-fold therapeutic dose level has been revealed in the chronic toxicity study. Mild to moderate pathological changes on different haematological, serum biochemical and cytoarchitecture of different organs were observed at ten-fold therapeutic dose level. Based on these observations, it can be concluded that Rasamanikya is safe at therapeutic dose levels when used judiciously along with specified adjuvants.
Keywords
Arsenic, Haratala, Rasamanikya, safety, Shodhana, toxicity
Ayurveda utilizes natural resources of plant, animal, metal and mineral origin in therapeutics of different pathologies. These resources are converted into formulations based upon the need by following specified classical guidelines. Herbo-mineral and metallic formulations are an important part of Ayurveda that are attributed to be safe and efficacious when manufactured and used judiciously. Rasamanikya, one such metallic formulations, attracted controversies in scientific community due to the presence of arsenic as an integral component. It is being used by Ayurveda physicians to treat fever (Jwara), cough (Kasa), asthma (Shwasa), fistulous tracts (Nadi Vrana) and skin diseases (Kushtha) [1]. In general, arsenicals are toxic and produce untoward effects on administration [2]. Anticipating such untoward effects; seers of Ayurveda have explained meticulous handling procedures and administration modalities for all such metals and minerals including arsenicals [3]. Importance of following traditional pharmaceutical procedures in preparation of Ayurvedic formulations have been wellestablished [4,5]. Ayurveda emphasizes on administration of metallic formulations orally in specified quantities with great caution along with requisite anupana (vehicle) that is anticipated to play a key role in safety aspects of Rasaushadhies. Anupana facilitates drug administration, improves palatability and also reduces toxic nature of the drug [6,7]. Though, there are many evidences that indicate the safe nature of metallic formulations; there is a need to generate scientific data-based evidences for all such formulations that contain heavy metals. As, Rasamanikya contains Haratala (arsenic trisulphide) as a component; it became necessary to evaluate its safety profile. Earlier studies established safety of Rasamanikya prepared by Haratala processed in Churnodaka (lime water) [8]. But, whether the safety profiles will be the same, if processing media is changed is not known. Considering this, safety profiles of Rasamanikya prepared with Kushmanda Swarasa Shodhita Haratala (arsenic trisulphide processed in fruit juice of Benincasa hispida) was evaluated in the current study.
Materials and Methods
Haratala Shodhana (processing of arsenic trisulphide)
Raw Haratala was procured from the Pharmacy, Gujarat Ayurved University, Jamnagar, Gujarat; made into small pieces (40#), bundled in a cotton cloth (pottali), suspended in a stainless steel vessel taking care not to touch the bottom and walls of the vessel assuring free movement. Quantity sufficient amount of Kushmanda Swarasa (fruit juice of Benincasa hispida) to completely immerse the Pottali was added into the vessel and subjected to heat at 100° for three hours. Care was taken to immerse Pottali completely in Kushmanda swarasa throughout the boiling process. At the end of three hours boiling, Haratala was removed carefully from the Pottali, washed with hot water and dried to obtain Shuddha (processed) Haratala [9] (Figure 1a-e).
Figure 1: Photomicrographs of preparation steps of Rasamanikya (a) Raw Kushmanda (Benincasa hispida), (b) raw Haratala, (c) Pottali of raw Haratala, (d) boiling of raw Haratala, (e) Shodhita Haratala, (f) clipped Haratala powder within mica sheets, (g) heating on LPG gas stove, (h) prepared Rasamanikya powder
Preparation of Rasamanikya
Thin layers of completely dried and powdered Shuddha haratala were placed in between two mica sheets and the boundaries were locked with ‘U’ pins. This was held with the help of tongs and heated over a gas stove until Haratala melted completely. Heating was stopped, the ‘U’ clips were carefully removed to open the mica layers to expose the ruby-colored product, Rasamanikya was collected carefully by avoiding the mica particles. This was coded as RM (Figure 1f-h) [10].
Experimental animals
Charles-Foster rats of either sex weighing 200±20 g were obtained from the animal house attached to the pharmacology laboratory, Institute for Postgraduate Teaching and Research in Ayurveda, Gujarat Ayurved University, Jamnagar. The animals were exposed to natural day and night cycles under ideal laboratory conditions in terms of ambient temperature (23±2°) and humidity (50-60 %). Animals were fed ad libitum with Amrut brand rat pellet feed supplied by Pranav Agro Industries and tap water. The experiment was carried out after obtaining permission from Institutional Animal Ethics Committee (IAEC/15/2013/39) and care of animals was taken as per the Committee for the Purpose of Control and Supervision of Experiments on Animals guidelines.
Dose fixation and schedule
The therapeutic dose (TED) of RM is 250 mg [1]. Rat dose was calculated by referring to table of Paget and Barnes and was found to be 22.5 mg/kg of rat [11]. As classics advocate using RM along with honey and ghee as adjuvant [12]; RM was administered orally along with honey and ghee with the help of oral cannula.
Acute toxicity study
Young, healthy, nulliparous, and non-pregnant Charles-Foster female rats were selected and acclimatized for seven days before the experiment. RM along with adjuvant were orally administered at the limit dose of 2000 mg/kg to overnight fasted rats by following Organization for Economic Cooperation and Development (OECD) 425 guidelines [13]. The rats were observed closely for behavioural changes, signs of toxicity, and mortality, if any continuously for the first six hours and thereafter periodically up to 14 d.
Chronic toxicity study
The study was carried out by following OECD 408 guidelines [14]. Charles-Foster rats were selected and randomly grouped into six, each consisting of six rats comprising three males and three females. Animals of group-I received tap water and normal food and served as normal control (NC), while animals in group-II received vehicle (1 ml/kg of honey and ghee orally) and served as vehicle control (VC). Group-III to V received RM along with adjuvant at TED (22.5 mg/kg orally), TED×5 (112.5 mg/kg orally) and TED×10 (225 mg/kg orally) dose levels, respectively. Animals of group-VI also received test drug at the level of TED×10 (225 mg/kg orally) along with adjuvant and is served as recovery group (Table 1).
| Group | No of animals | Drug | Dose (mg/kg) | |
|---|---|---|---|---|
| I | Normal Control | 6 | NC | -- |
| II | Vehicle Control | 6 | VC | 1 |
| III | Therapeutic Dose | 6 | RM | 22.5 |
| IV | TED x 5 | 6 | RM | 112.5 |
| V | TED x 10 | 6 | RM | 225 |
| VI | Recovery Group | 6 | RM | 225 |
NC: normal control, VC: vehicle control, RM: Rasamanikya, TED: therapeutically equivalent dose
Table 1: Test drug posology for chronic toxicity
Initial body weight of all animals was recorded. General behavioural pattern was observed once a week by exposing each animal to open arena. On 90th d, animals of group I-V were weighed again and anaesthetized with diethyl ether. Supraorbital plexus was punctured and blood was collected using capillaries in two different tubes, one containing anticoagulant fluid for haematological parameters and another plain tube for serum biochemical investigations. Then the rats were sacrificed with overdose of diethyl ether and the abdomen was opened through midline incision to observe the autopsy changes followed by dissecting out the important organs.
Haematological analysis was performed by using an automatic haematological analyser (Swelab, Sweden). Total red blood cell (RBC), hemoglobin, hematocrit, mean corpuscular volume, mean corpuscular hemoglobin, mean corpuscular hemoglobin concentration, white blood cell, neutrophils, percent lymphocytes, eosinophil’s and monocytes, packed cell volume (PCV) and platelet count were measured from the blood samples.
Serum biochemical parameters were carried out by using fully automated biochemical random access analyser (BS-200, Lilac Medicare Pvt. Ltd., Mumbai). The studied parameters were blood glucose, total cholesterol, triglyceride, high density lipoproteins (HDL) cholesterol, very low density lipoprotein cholesterol, serum glutamic oxaloacetic transaminase, serum glutamic pyruvic transaminase (SGPT), alkaline phosphatase, total bilirubin, direct bilirubin, blood urea, creatinine, total protein, albumin, globulin, uric acid and serum calcium [15-29].
Liver, kidney, heart, lungs, trachea, intestine, spleen, thymus, lymph node, ileum, testis, seminal vesicle, prostate, uterus and ovary were dissected carefully. After noting signs of gross lesions and ponderal changes of these organs; all were transferred to 10 % phosphate buffered formalin solution for fixation and later on subjected to dehydrating, wax embedding, sectioning and staining with haematoxylin and eosin for histological evaluation. The slides were viewed under trinocular research Carl Zeiss microscope at various magnifications to note down the changes in the microscopic features of the tissues.
Statistical analysis
The data is expressed as mean±standard error (SE) of each experimental group. Statistical comparisons were carried out by both unpaired Student’s t test and oneway analysis of variance to compare the mean values of quantitative variables among the groups followed by Dunnett multiple t - test for unpaired data by using Sigma stat software (version 3.5, Systat Software Inc.) to determine significant difference between groups at p<0.05.
Results and Discussion
The results of acute toxicity showed no changes in gross behaviour in any of the animals. RM along with adjuvant did not showed any signs of toxicity and mortality up to 14 d when given at a dose of 2000 mg/kg. Behavioural changes were not observed in any of the treated groups during the course of chronic toxicity in comparison to NC group. No mortality was observed in any of the experimental groups. Weight gain was observed in all treated groups, but percent change in body weight pattern in treated groups did not differ significantly from the changes observed in control group (Table 2). Administration of RM at different dose levels resulted in insignificant changes in relative weight of nine organs except significant increase in weight of kidney at TED, TED×10 and thymus at TED×5 dose levels. Significant increase in liver, kidney, thymus and uterus was observed in recovery group (Table 3).
| Groups | 0 day | 4th week | 8th week | 12th week |
|---|---|---|---|---|
| NS | 180.00±8.76 | 192.50±7.04 | 197.50±8.3 | 205.83±11.50 |
| VC | 196.67±3.33 | 208.33±4.77 | 223.33±9.54* | 238.33±20.07 |
| RM - TED | 195.00±8.47 | 203.33±9.54 | 230.00±11.83* | 244.00±13.64* |
| RM - TED×5 | 190.00±7.75 | 198.33±10.77 | 221.67±12.22 | 226.67±18.74 |
| RM - TED×10 | 208.33±9.46 | 205.00±7.19 | 235.00±6.71 | 228.33±16.00 |
| Recovery | 198.33±4.014 | 201.67±5.43 | 193.33±3.33 | 228.33±7.49 |
Data presented as mean±SEM, *p<0.05
Table 2: Effect of Rasamanikya on body weight
| Relative weight | NC | VC | Rasamanikya | |||
|---|---|---|---|---|---|---|
| TED | TED×5 | TED×10 | Recovery | |||
| Heart (mg/100 g) |
0.616±0.02 | 0.581±0.04 | 0.597±0.02 | 0.681±0.03 | 0.675±0.02 | 0.656±0.03 |
| Liver (g/100 g) |
6.025±0.19 | 6.162±0.64 | 5.694±0.27 | 7.376±0.67 | 7.277±0.53 | 7.527±0.61 |
| Spleen (mg/100 g) |
0.394±0.01 | 0.337±0.03 | 0.339±0.01 | 0.447±0.03 | 0.441±0.03 | 0.459±0.04 |
| Kidney (mg/100 g) |
1.334±0.04 | 1.495±0.10 | 1.504±0.06 | 1.547±0.10 | 1.604±0.06* | 1.645±0.06* |
| Thymus (mg/100 g) |
0.305±0.02 | 0.340±0.02 | 0.303±0.00 | 0.370±0.01 | 0.375±0.02 | 0.385±0.02 |
| Testis (mg/100 g) |
2.385±0.08 | 2.410±0.04 | 2.104±0.04 | 2.104±0.14 | 2.258±0.17 | 2.374±0.09 |
| Seminal vesical (mg/100 g) | 0.798±0.15 | 0.741±0.11 | 0.650±0.06 | 0.966±0.13 | 1.083±0.09 | 1.108±0.00 |
| Prostate (mg/100 g) | 0.257±0.02 | 0.237±0.04 | 0.218±0.01 | 0.324±0.02 | 0.321±0.02 | 0.387±0.08 |
| Uterus (mg/100 g) | 0.168±0.00 | 0.247±0.05 | 0.269±0.05 | 0.208±0.04 | 0.271±0.03 | 0.327±0.03* |
*P<0.05 when compared with control group (ANOVA followed by Dunnett’s multiple ‘t’ test), NC: normal control, VC: vehicle control
Table 3: Effect of Rasamanikya on relative organ weight
Significant increase in neutrophils and decrease in lymphocytes was observed in group-II and group-III, but they showed insignificant changes at higher dose levels and in recovery group. Significant increase was observed in RBC, PCV and hemoglobin at lower dose level and VC group in comparison to NC group. However, there was insignificant change at higher dose levels in comparison to NC and VC (Table 4). The results of test drugs on serum biochemical parameters showed significant decrease in blood urea at all dose levels including in recovery group. Significant increase in serum creatinine and albumin was observed at lower dose level but was within normal range. However, similar changes were not observed at higher dose levels and in recovery group in comparison to control and VC groups. Significant decrease was observed in serum globulin at TED×5 and in recovery group. Total protein level was not affected by the drug at all doses, while albumin level was increased and globulin level was decreased by test drugs, but, were within normal range, hence are not of serious in nature. The test drug at all dose levels decreased total cholesterol, increased triglycerides and HDL-cholesterol in comparison to control group, but no changes were observed when compared to VC group (Table 5).
| Parameters | NC | VC | Rasamanikya | |||
|---|---|---|---|---|---|---|
| TED | TED×5 | TED×10 | Recovery | |||
| TWBC (103/ml) | 7700.00±699.05 | 6766.67±725.10 | 6980.00±563.38 | 8750.00±834.96 | 7366.67±1006.53 | 8600.00±610.46 |
| Neutrophil (%) | 14.17±2.59 | 29.17±4.44* | 35.20±2.94 | 20.17±4.08 | 22.67±2.79* | 13.67±3.76 |
| Lymphocyte (%) | 81.83±2.53 | 67.00±4.70* | 63.00±1.64 | 76.50±4.37 | 73.17±2.63 | 82.50±4.07 |
| Eosinophil (%) | 2.33±0.21 | 2.17±0.31 | 2.40±0.24 | 1.83±0.17 | 2.33±0.33 | 2.17±0.31 |
| Monocyte (%) | 1.68±0.21 | 1.67±0.21 | 1.40±0.24 | 1.50±0.22 | 1.83±0.17 | 1.68±0.21 |
| Hb (g/dl) | 14.40±0.36 | 17.67±0.56 | 17.20±0.62 | 14.70±0.18 | 14.73±0.22 | 15.33±0.25 |
| PCV (%) | 43.97±0.62 | 52.75±1.91 | 53.26±2.50 | 45.58±0.81 | 46.12±0.85 | 46.90±0.96 |
| TRBC (106/ml) | 7.69±0.14 | 9.26±0.34 | 9.53±0.47 | 8.12±0.22 | 8.33±0.19 | 8.36±0.18 |
| MCV (fl) | 57.22±0.63 | 56.97±0.30 | 55.84±0.47 | 56.23±0.78 | 55.38±0.51 | 56.10±0.58 |
| MCH (pg/red cell) | 18.72±0.30 | 18.67±0.17 | 18.12±0.31 | 18.13±0.32 | 17.77±0.28 | 18.35±0.29 |
| MCHC (g/dl) | 32.73±0.44 | 32.75±0.21 | 32.46±0.39 | 32.25±0.21 | 32.05±0.23 | 32.70±0.68 |
| Platelets (103/ml) | 1201.83±56.13 | 1284.17±68.67 | 1254.60±74.64 | 1217.50±137.16 | 1291.33±79.78 | 1218.50±37.49 |
Data: Mean±SEM, *p<0.05, when compared with control group; NC: normal control, VC: vehicle control
Table 4: Effect of Rasamanikya on hematological parameters
| Parameters | NC | VC | Rasamanikya | |||
|---|---|---|---|---|---|---|
| TED | TED×5 | TED×10 | Recovery | |||
| FBS (mg/dl) |
62.20±3.72 | 51.00±6.21 | 59.20±4.19 | 66.50±4.31 | 70.00±3.53 | 71.33±1.02 |
| S. cholesterol (mg/dl) |
46.83±2.97 | 41.50±1.43 | 37.00±3.18 | 41.00±3.03 | 43.00±0.73 | 39.17±3.31 |
| Triglycerides (mg/dl) | 67.50±4.31 | 78.50±13.55 | 69.60±20.05 | 85.00±11.48 | 90.33±13.57 | 87.50±9.94 |
| HDL (mg/dl) | 31.00±1.53 | 33.17±1.83 | 29.60±2.84 | 34.17±2.07 | 36.50±1.15 | 29.67±1.91 |
| VLDL (mg/dl) | 13.50±0.86 | 15.70±2.71 | 13.92±4.01 | 17.00±2.29 | 18.07±2.71 | 17.50±1.99 |
| SGPT (IU/L) | 43.50±3.57 | 49.83±3.46 | 46.20±2.75 | 54.33±4.88 | 43.33±2.85 | 43.00±3.71 |
| SGOT (IU/l) | 135.50±9.63 | 157.67±12.95 | 134.40±8.15 | 157.17±7.97 | 128.00±6.72 | 117.83±9.25 |
| ALP (IU/l) | 166.00±24.59 | 134.50±23.11 | 106.40±19.00 | 163.33±24.21 | 187.50±23.32 | 134.83±13.81 |
| T. bilirubin (mg/dl) | 0.60±0.73 | 0.40±0.04 | 0.46±0.07 | 0.57±0.08 | 0.48±0.05 | 0.47±0.05 |
| D. bilirubin (mg/dl) | 0.22±0.02 | 0.13±0.02 | 0.16±0.02 | 0.20±0.02 | 0.17±0.02 | 0.13±0.02 |
| Blood urea (mg/dl) |
56.33±2.08 | 37.33±2.73* | 35.80±1.11* | 30.33±1.43* | 30.00±1.39* | 28.00±1.21* |
| Creatinin (mg/dl) |
0.63±0.02 | 0.83±0.02* | 0.80±0.00* | 0.58±0.05 | 0.63±0.04 | 0.65±0.03 |
| Protein (g/dl) | 6.88±0.21 | 7.43±0.16 | 7.14±0.16 | 6.72±0.20 | 6.98±0.22 | 6.62±0.11 |
| Albumin (g/dl) | 3.40±0.086 | 4.00±0.15 | 4.02±0.15 | 3.87±0.09 | 3.90±0.12* | 3.88±0.17* |
| Globulin (g/dl) | 3.48±0.15 | 3.43±0.12 | 3.12±0.04 | 2.85±0.16*@ | 3.15±0.24 | 2.73±0.11@ |
| Uric acid (mg/dl) | 1.10±0.15 | 1.50±0.19 | 1.66±0.20 | 1.00±0.17 | 0.87±0.11 | 0.82±0.08 |
| S. calcium (mg/dl) | 10.05±0.60 | 9.83±0.37 | 9.92±0.33 | 10.12±0.16 | 10.45±0.19 | 10.57±0.31 |
Data: mean±SEM, *p<0.05, when compared with control group, @p<0.05 compared to control group (ANOVA followed by Dunnett’s multiple ‘t’ test), NC: normal control, VC: vehicle control
Table 5: Effect of Rasamanikya on biochemical parameters
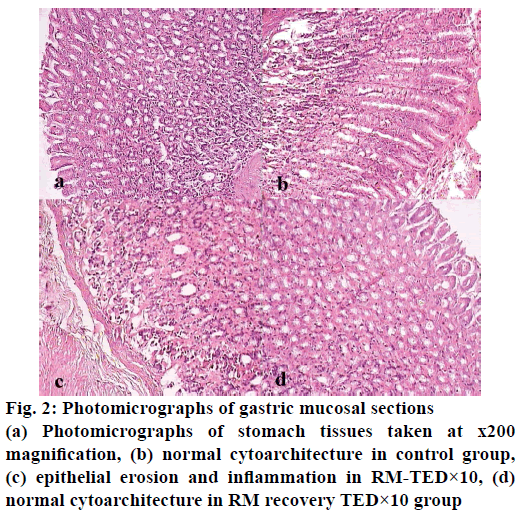
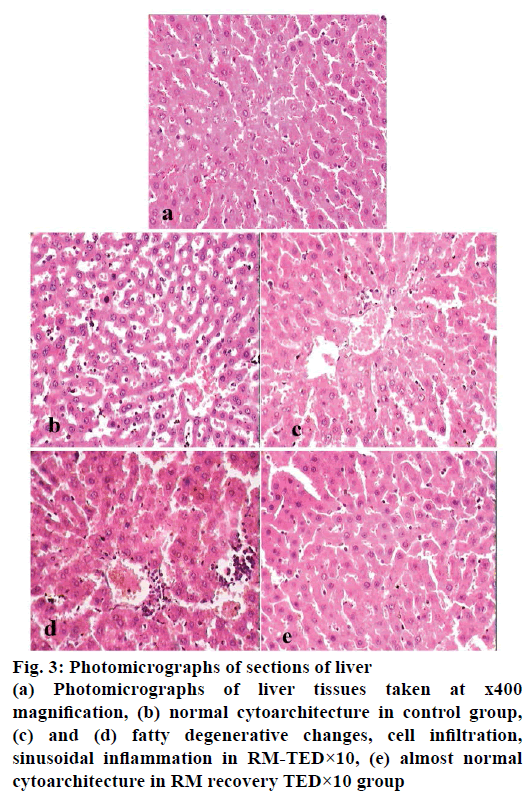
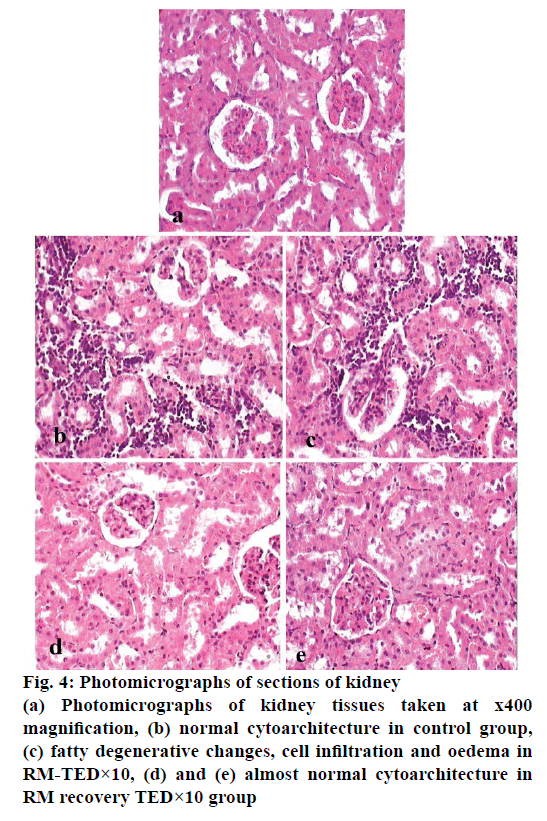
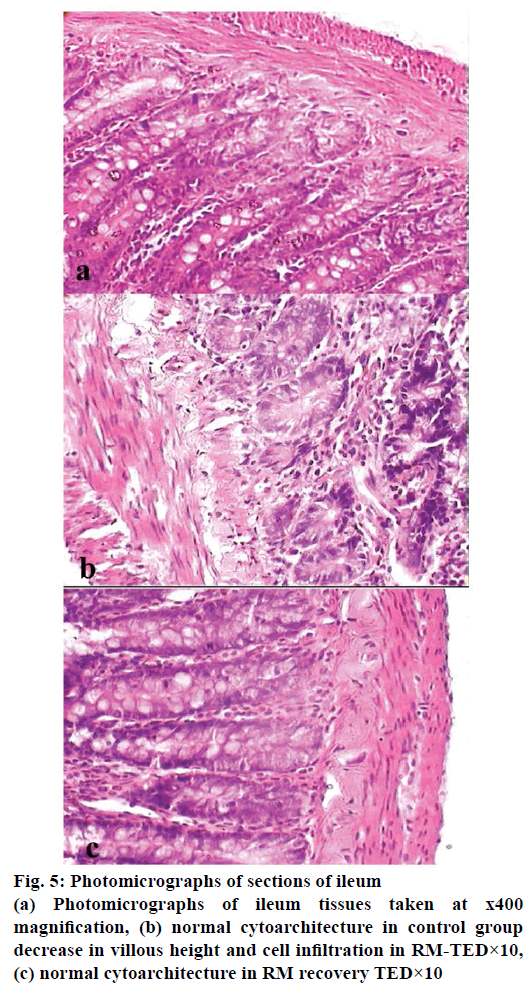
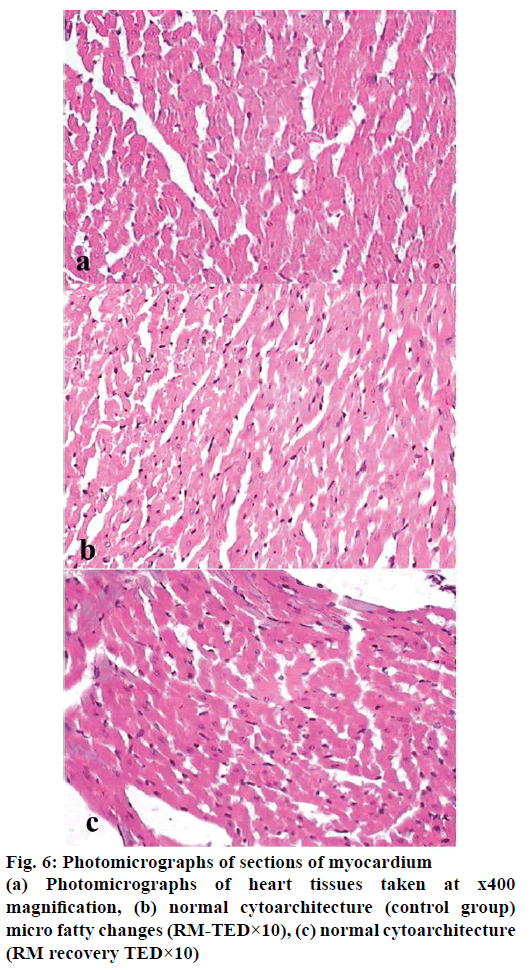
Histopathological studies revealed pathological changes in stomach, heart, liver, kidney and ileum on administration of RM at TED×10 dose levels. Mild to moderate epithelial erosions were observed in stomach (Figure 2), while liver showed micro fatty changes (Figure 3). Mild to moderate inflammation was observed in kidney (Figure 4). Villous shortening at ileum (Figure 5) and fatty changes in heart was observed (Figure 6). These changes were recovered and no pathological changes were observed in any of the organs in the animals of group-VI.
OECD 425 guidelines for oral acute toxicity study was employed to record immediate adverse signs and symptoms after administration of single dose of drug at 2000 mg/kg dose level that is several folds higher than the actual therapeutic equivalent dose. Female rats were used for acute toxicity study to reduce variability. This is because there is little difference in sensitivity in LD50 between the sexes; however, in those cases where differences were observed; females were generally slightly more sensitive [30]. Oral administration of RM at the dose level of 2000 mg/kg did not produced any observable toxic effects and all female rats were survived for 14 d of observation suggesting that LD50 value of Rasamanikya may be higher than 2000 mg/kg.
Significant increase was observed in RBC, PCV and haemoglobin in group-II and group-III in comparison to NC group. The observed changes cannot be considered as toxicity of test drugs as these changes were not observed at higher dose levels. Arsenic likely causes both direct cytotoxic effect on blood cells and suppression of erythropoiesis through bone marrow toxicity and may inhibit haem synthesis [31]. Test drug did not showed any such toxic effects on cellular constituents in present study. Test drug did not hamper the erythropoiesis rather enhanced the formation of RBC's suggesting possible therapeutic role of Rasamanikya along with honey and ghee in conditions like anaemia. Role of arsenical compound in blood disorders was also reported earlier [32].
Result of test drugs on serum biochemical parameters showed significant decrease in blood urea at all dose levels including recovery group of animals. Increase in urea has significant role in toxicity, while low level represents low turnover of protein and nitrogenous material in rats or may be due serious liver toxicity. In present study, liver function tests didn’t elevated to significant extent but mild to moderate changes were observed in histopathological studies suggesting decrease in urea level may be due to low turnover of protein level and nitrogenous material in rats or low magnitude of liver toxicity. Significant increase in serum creatinine was observed at TED level in group- III, but was in the physiological range. This increase in serum creatinine was not observed at higher dose levels and in animals under recovery study in comparison to control or VC groups. Significant decrease was observed in serum globulin at TED×5 and in recovery group. Total protein level was unaffected at all doses. Albumin level was increased while globulin level was decreased by test drugs. All these values are within normal range.
The test drug at all dose levels decreased total cholesterol, while triglycerides and HDL cholesterol were increased in comparison to control group, but no changes were observed when compared to VC group. This indicates possible interference with the lipid turnover in the experimental animals. HDL plays very important role in preventing the atherogenesis by taking away the cholesterol from the arterial wall and by inhibiting the oxidation of atherogenic lipoproteins [33]. Decrease in total cholesterol level with concomitant increase in HDL cholesterol may suggest therapeutic utility of Rasamanikya in the hyperlipidemic conditions.
Whenever there is injury in organs like liver; SGPT level gets elevated. Serum aminotransferases are elevated in most liver disorders and these are one of the most reliable markers of hepatocellular injury or necrosis [34]. In present study, serum transaminases insignificantly increased at lower dose level including VC, however values are within normal range. All these observed changes are reverted in recovery group and not observed at higher dose level in comparison to control and VC group. The decrease in bilirubin is still within normal range. The observed serum biochemical changes are mild and within normal ranges, hence it is inferred that drug is devoid of any serious toxic effects on serum biochemical parameters in rats.
Mild to moderate pathological changes in heart, liver, kidney and GI tract at higher dose levels were observed in histopathological study. The observed changes were not seen in group-I, where RM was administered at therapeutic dose, suggesting safety nature of the drug. The effects of biochemical parameters also match and support the histopathological findings of liver, kidney, stomach and ileum at higher dose level of the test drug validating safety nature of Ayurvedic arsenical formulations.
As few pathological changes appeared at TED×10 dose levels; physicians should be cautious while using this drug in patients with hepatic, cardiac and renal impairments. Interestingly, these pathological changes were reversed in the recovery group indicating reversal of drug-induced adverse changes. Main treatment of heavy metal toxicity is to prevent or terminate the exposure [35]. Hence, after termination of RM, normal histopathology was appeared in the organs of animals in the recovery group.
The current study demonstrated safety of Rasamanikya processed in fruit juice of Benincasa hispida when administered at therapeutic and at TED×5 dose levels along with honey and ghee as adjuvants. RM may produce mild to moderate pathological changes when administered for longer duration at higher (TED×10) doses. Though the observed pathological changes were reverted after withdrawal of the drug; it is advisable to administer RM in specified TED along with a suitable vehicle (honey and ghee) for shorter periods. Care need to be observed while administering the drug in patients with hepatic, cardiac and renal impairments. It is also inferred that such formulations are not to be administered continuously for a longer period and observing a gap of few days is advisable [36].
Conflicts of interest
There are no conflicts of interest.
Financial support and sponsorship
Nil.
References
- Sharma SN. Rasatarangini. In: Shastri KN, editor. 11th Chapter. Verse 88-89. Reprint. Delhi: Motilal Banarasi Das; 2009. p. 257.
- Mehta M, Hundal SS. Assessment of genotoxic potential of arsenic in female albino rats at permissible dose levels. Toxicol Int 2014;21:24-8.
- Somadeva. Rasendra Chudamani. In: Mishra SN, editor. 11th Chapter. Verse 35. Reprint. Varanasi: Chaukhamba Orientalia; 2009. p. 176.
- Jagtap CY, Ashok BK, Patgiri BJ, Prajapati PK, Ravishankar B. Acute and sub chronic toxicity study of Tamra Bhasma (Incinerated Copper) prepared from Ashodhita (Unpurified) and Shodhita (Purified) Tamra in rats. Indian J Pharm Sci 2013;75:346-52.
- Chaudhari S, Nariya M, Galib R, Prajapati PK. Acute and subchronic toxicity of Tamra Bhasma (incinerated copper) prepared with and without Amritikarana. J Ayurveda Integr Med 2016;7:23-9.
- Kapoor RC. Some observations on the metal-based preparations in the Indian Systems of Medicine. Indian J Tradit Know2010;9:562-75.
- Dwivedi V, Anandan EM, Mony RS, Muraleedharan TS, Valiathan MS, Mutsuddi M, et al. In vivo effects of traditional Ayurvedic formulations in drosophila melanogaster model relate with therapeutic applications. PLoS One 2012;7:e37113.
- Anonymous. Research study profile of Rasamanikya. New Delhi: Central Council for Research in Ayurvedic Sciences; 2009. p. 1-212.
- Vagbhatacharya. Rasaratna Samuchchaya, In: Kulkarni DA, editor. 3rd Chapter. Verse 70. Reprint. New Delhi: Meharchand Laxmandas publication; 2007. p. 54.
- Bhatta K. Siddha Bhaishajya Manimala. 4th Chapter. Verse 66. Varanasi: Choukhambha Krishnadas Academy; 2008. p. 159.
- Paget GE, Barnes JM. Evaluation of drug activities. In: Laurence DR, Bacharach AL, editors. Pharmacometrics. Vol. 1. London: Academic Press; 1964. p. 50.
- Dhundhuknath. Rasendra Chintamani, In: Mishra SN, editor. 9th Chapter. Verse 131. Edition. Varanasi: Chaukhamba Orientalia; 2000. p. 376.
- Organization for Economic Co-operation and Development (OECD) Guideline No. 423. Acute oral toxicity in animals. OECD/OCDE No. 425, adopted 17th December, 2001. Available from: http://www.ntp.niehs.nih.gov/iccvam/suppdocs/feddocs/oecd/oecd_gl423.pdf.
- http://www.keepeek.com/Digital-Asset-Management/oecd/environment/test-no-408-repeated-dose-90-day-oral-toxicity-study-in-rodents_9789264070707-en#.WUuQ3rafIhk.
- Pennock CA, Murphy D, Sellers J, Longdon KJ. A comparison auto analyzer method for the estimation of glucose in blood. Clin Chim Acta 1973;48:193-201.
- Roeschlau P, Bernt E, Gruber WA. Enzymatic determination of total cholesterol in serum. Z Klin Chem Klin Biochem 1974;12:226.
- Fossati P, Prencipe L. Serum triglycerides determined calorimetrically with an enzyme that produces hydrogen peroxide. Clin Chem 1982;28:2077-80.
- Dominiczak M, McNamara J, Nauk M, Wiebe D, Warnick G. Measurement of high density lipoprotein cholesterol. In: Rifai, Warnick, Dominiczak, editors. Handbook of lipoprotein testing. 2nd ed. Washington DC: AACC Press; 2000. p. 819.
- Tietz NW. Clinical guide to laboratory tests. 3rd ed. Philadelphia: WB Saunders; 1995. p. 76.
- Burtis CA, Ashwood ER. Tietz Textbook of Clinical Chemistry. 3rd ed. Philadelphia: WB Saunders; 1999. p. 652.
- Wilkinson JH, Boutwell JH, Winsten S. Evaluation of a new system for kinetic measurement of serum alkaline phosphatase. Clin Chem 1969;15:487-95.
- Pearlman PC, Lee RT. Detection and measurement of total bilirubin in serum with use of surfactants as solubilising agents. Clin Chem 1974;20:447.
- Burtis CA, Ashwood ER, Editors. Tietz Textbook of Clinical Chemistry. 3rd ed. Philadelphia: WB Saunders; 1999. p. 1136.
- Talke H, Schubert GE. Enzymatic urea determination in the blood and serum in Warburg optical test. Klin Wochens Chr 1965;42:174-5.
- Slot C. Plasma creatinine determination: a new and specific Jaffe reaction method. Scand J Clin Lab Invest 1965;17:381-7.
- Tietz NW. Text book of Clinical Chemistry. Philadelphia: WB Saunders; 1986. p. 579.
- Doumas BT, Arends RL, Pinto PC. In standard methods of clinical chemistry. Vol. VII. Chicago: Academic Press; 1972. p. 175-89.
- Kabasakalian P, Kalliney S, Wescott A. Determination of uric acid in serum, with use of uricase and tribromophenol amino antipyrine chromogen. Clin Chem 1973;19:522.
- Biggs HG, Moorehead WR. 2-Amino-2-methyl-1-propanol as the alkalizing agent in an improved continuous-flow cresolphthalein complexone procedure for calcium in serum. Clin Chem 1974;20:1458-60.
- Lipnick RL, Cotruvo JA, Hill RN, Bruce RD, Stitzel KA, Walker AP, et al. Comparison of the up and down, conventional LD50, and fixed dose acute toxicity procedures. Food Chem Toxicol 1995;33:223e31.
- Byrns MC, Penning TM. Environmental Toxicology: Carcinogens and Heavy Metals. In: Brunton L, Chabner B, Knollman B, editors. Goodman and Gilman’s The Pharmacological Basis of Therapeutics. 12th ed. New York: McGraw-Hill Medical Publishing Division; 2011. p. 1870.
- Shen ZX, Chen GQ, Ni JH, Li XS, Xiong SM, Qiu QY, et al. Use of arsenic trioxide (As2O3) in the treatment of acute promyelocytic leukemia (APL): II. Clinical efficacy and pharmacokinetics in relapsed patients. Blood 1997;89:3354-60.
- Barter P. The Role of HDL Cholesterol in prevention of atherosclerotic disease. Eur Heart J Suppl 2005;7:4-8.
- Geboney P. Mildly elevated liver transaminase levels in asymptomatic patient. Am Fam Physician 2005;71:1105-110.
- Satoskar RS, Bhandarkar SD, Rege NN. Pharmacology and Pharmaco therapeutics. Reprint. Mumbai: Popular Prakashan; 2010. p. 1041.
- Charaka. Charaka Samhita, In: Chakrapani, Chikitsasthana. 5th Chapter. Verse 57. Reprint. Varanasi: Chaukhamba Sanskrita Sansthana; 2000. p. 439.